-
Call Now
1800-102-2727
Second Law of Thermodynamics - Statement, Examples, Practice problems, FAQs
The first law of thermodynamics explains that energy is conserved–however it does not talk about the direction of flow of heat. Energy is capable of being exchanged between systems and surroundings, however it cannot be created or destroyed. For instance, when food is fried in a pan, and a lid is put on top of the pan, the pan would exchange heat with the lid. The quantity of heat transferred can be calculated with the first law–however, it gives no information regarding the direction of heat transfer. The second law of thermodynamics aims to explain the feasibility of a heat transfer process. Additionally, the second law talks about entropy–the degree of disorderliness of a system, and what causes it to increase or decrease. When a reaction has to occur feasibly, then its entropy has to increase. So let us explore the second law of thermodynamics in detail.
What is the second law of thermodynamics?
For any process that occurs spontaneously, the second law of thermodynamics states that the entropy(S) of the universe will always increase. Simply put, for an isolated system, the entropy of the system will never decrease with time.
Example: Melting of ice–in this case, the entropy of the system always continues to increase. Entropy is a measure of the randomness of the system.
The second law explains that it is impossible to convert all the heat energy into mechanical energy with 100 % efficiency. Let us take the example of an engine fitted with a piston. When the gas in the engine is heated, the piston moves up. However, there is some unused heat in the gas that is not used to do any kind of work. This heat is wasted and needs to be discarded. Additionally, the heat generated from friction( movement of piston against the cylinder generates friction) is generally unusable and also needs to be discarded.
Second law of thermodynamics equation
The second law of thermodynamics can be written as,
Suniv indicates the entropy change of the universe.
In a closed system, the mass remains constant while energy is exchanged. This exchange in heat leads to more disturbances in the system, thereby increasing its entropy.
Kelvin Planck statement
It states that: it is impossible to design a heat engine to extract heat from a single reservoir at a single fixed temperature and do work in the process.
Clausius’ statement
It is not possible to construct a device, operating in a cycle capable of transferring heat from a colder body to a hotter body without doing any work. Additionally, energy is not capable of flowing simultaneously from an object of lower temperature to another object of higher temperature. Some form of work needs to be done to transfer heat from colder to hotter bodies. The heat pump and refrigerator work on the principle of second law. The refrigerator needs to be driven by a compressor to make the heat transfer possible.
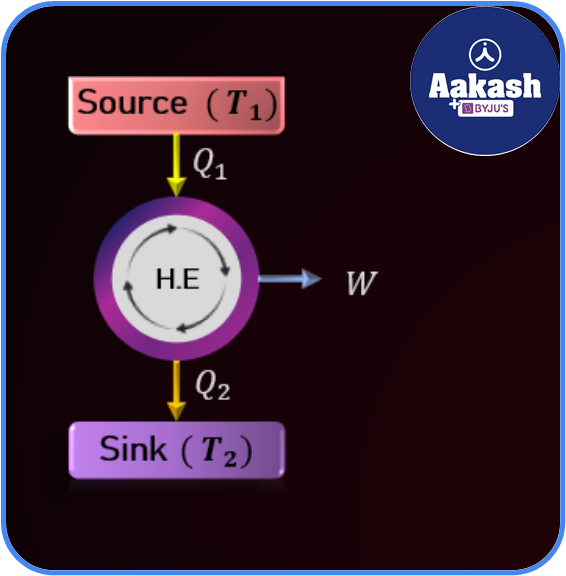
Carnot Engine
French physicist Sadi Carnot, also known as the father of thermodynamics, devised the Carnot cycle in 1824. It consists of a hotter reservoir( source— at temperature T1) and colder reservoir ( sink— at temperature T2). It works on four cycles– isothermal expansion, adiabatic expansion, isothermal compression and adiabatic compression.
If Q1 indicates the heat extracted from the hotter reservoir and Q2is the heat rejected to the colder reservoir, then the amount of work done in transferring heat from hotter to colder reservoir would be
W=Q1-Q2
The efficiency of the carnot engine is defined as the ratio of the work done to the heat extracted from the hotter reservoir.
Efficiency,
Alternately, if Tc and Th indicate the colder body and hotter body temperatures, then the efficiency can also be written as,
Note:
Efficiency of a Carnot engine is always less than 1 , ie less than 100 %. If it were equal to 1 or 100 %, it means that it Tc=0, meaning that it exchanges heat with only one reservoir—thereby violating Kelvin Planck's statement. All reversible heat engines working between the same temperatures are not more efficient than the Carnot engine.
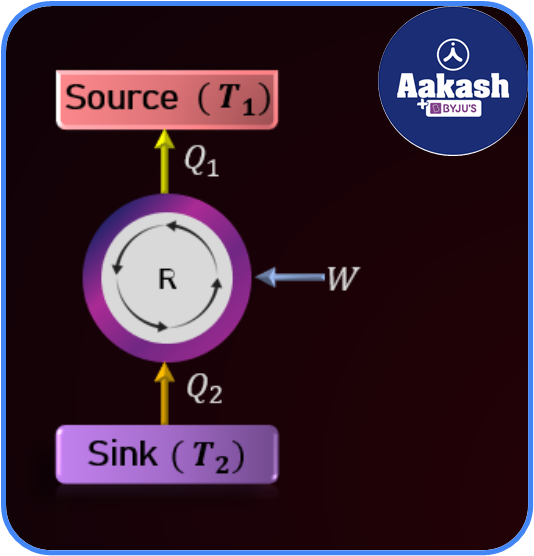
Refrigerator
Refrigerator is the reverse of a Carnot engine. The heat flows from colder body(sink) to hotter body(source) and in the process, some amount of work is done.
Let indicate the coefficient of performance of a refrigerator. Then
It is always greater than one.
Perpetual Motion Machine of the second kind
A device which is capable of producing work when it interacts with only one heat reservoir is known as a perpetual motion machine of the second kind (PMM2).This clearly violates the second law of thermodynamics which takes into account two reservoirs.
Hence, a heat engine has to operate between two reservoirs to execute a cyclic process.
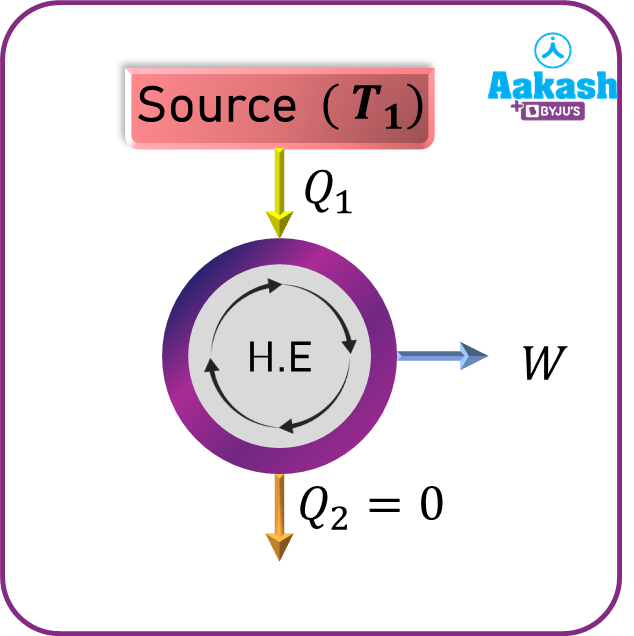
Here,Q2=0 and hence, work done W= Q1-Q2=Q1
Mastering the Second Law of Thermodynamics: Class 11 Physics Guide
Welcome, future scientists! If you have ever wondered why a hot cup of chai always cools down or why your room magically gets messy even when you don’t try, you are already observing the second law of thermodynamics in action. This topic is a cornerstone of second law of thermodynamics class 11 studies, and understanding it will change how you look at the entire universe!
What Exactly Is the Second Law?
To define the second law of thermodynamics simply for a younger student: think of it as the "Law of Messiness". In nature, things love to move from being organised to being a bit chaotic.
While the First Law of Thermodynamics tells us that energy cannot be created or destroyed, the second law tells us which direction that energy prefers to move. For instance, the first law allows a broken cup to spontaneously jump back together because energy would still be conserved, but the second law tells us this is impossible in the real world.
The Core Statements You Need to Know
In your exams, you will likely need to provide a formal second law of thermodynamics statement. Scientists use two main perspectives to explain it, which are actually equivalent to one another:
- Clausius Statement: This focuses on heat transfer. It states that heat cannot move from a cooler object to a hotter one on its own. You always need to do work (like using electricity for a refrigerator) to make that happen.
- Kelvin–Planck Statement: This focuses on work and engines. It states that it is impossible to build a machine that takes heat from a single source and turns 100% of it into useful work without any other changes. Some energy is always wasted as heat.
The Concept of Entropy: The Measure of Disorder
To understand why things get "messy," we use a physical property called Entropy (S). Introduced by Clausius, entropy measures the randomness or disorder in a system.
The Core Formula: The change in entropy (ΔS) is calculated using the heat added (ΔQ) and the absolute temperature (T):
ΔS = ΔQ / T
In any natural process, the total entropy of the universe always increases or remains the same; it never decreases.
- For Reversible Processes: ΔSuniverse = 0
- For Irreversible (Natural) Processes: ΔSuniverse > 0
Common Pitfalls & Pro-Tips
| The Mistake | The Physics Reality | Pro-Tip for Marks |
|---|---|---|
| The Temperature Trap | Using Celsius (°C) in calculations | Always convert temperature to Kelvin (K). Entropy formulas require absolute temperature. |
| Ignoring the Sign of ΔQ | Forgetting that heat flow direction matters | If a system absorbs heat, ΔQ is positive and entropy increases. If it loses heat, ΔQ is negative. |
| The "Ideal" Illusion | Assuming real engines are 100% efficient | No real engine reaches Carnot efficiency due to friction and heat loss. If your math shows 100% efficiency, it's wrong! |
Real-World Second Law of Thermodynamics Examples
Why does this matter in your daily life? Here are a few relatable ways the law works:
- Cooling Coffee: A hot drink loses heat to the cooler room until both reach the same temperature.
- The Broken Egg: You can crack an egg (increasing disorder), but the egg won't un-crack itself because nature forbids a spontaneous decrease in entropy.
- Car Engines: Your engine gets hot because it can't convert all the petrol's energy into movement; some must be released as waste heat.
Reversible vs. Irreversible Processes
In second law of thermodynamics class 11 physics, we distinguish between two types of changes:
- Reversible Processes: Ideal, "perfect" processes that could be undone without leaving any change in the universe. They must occur very slowly (quasi-statically) and involve no friction.
- Irreversible Processes: Real-world processes that cannot be perfectly reversed. Most natural phenomena—such as a plant growing, gas leaking from a cylinder, or heat generated by friction—are irreversible.
Heat Engines and Refrigerators
The second law sets the "speed limit" for technology:
- Heat Engines: Machines that convert heat into work. Due to the second law, some energy is always lost to a cold reservoir as waste heat.
- Refrigerators: These act as heat pumps, moving heat from a colder region to a hotter one. They require external work because heat will not flow backward on its own.
Practice problems
Q. A heat pump needs to do 300 J of work in order to remove 400 J from the reservoir that has lower temperature. Find out how much heat is delivered to the hotter reservoir?
(a)100 J (b)450 J (c) 320 J (d)700 J
A.d
Given, work done W=300 J,
Heat removed from the colder reservoir Q2=400 J
Heat delivered to the hotter reservoir, Q1=?
Q1-Q2=WQ1=Q2+W=400+300=700 J
Hence, heat delivered to the hotter temperature reservoir is 700 J.
Q.A heat engine takes in 4000 kJ of heat from a source which is maintained at a constant temperature of 600 K. If the surrounding temperature is 300 K , calculate
(a) available heat energy (b)unavailable heat
A.
Given, heat extracted from source, Q=4000 kJ
T1=600 K
Temperature of the surroundings, Ts=300 K
Change in entropy, 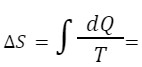
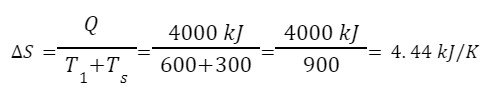
The available heat energy,
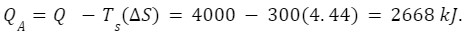
The amount of heat energy unavailable,
Heat available, =2668 kJ
Heat unavailable,=1332 kJ
Q.A Carnot Engine is operated between two reservoirs which are at temperatures 300 K and 600 K. Find the efficiency of the engine.
A.
Given, temperature of the colder reservoir, Tc= 300 K
Temperature of the hotter reservoir, Th= 600 K
Q.The colder reservoir of a refrigerator is at a temperature of 400 K and the hotter reservoir is at a temperature of 600 K. Calculate the coefficient of performance of the refrigerator.
B.
Hotter reservoir temperature, T1= 600 K
Colder reservoir temperature T2= 400 K
Coefficient of performance of the refrigerator,
Frequently Asked Questions (FAQs)
1. What is the basic definition of the second law of thermodynamics?
The second law states that the total entropy or disorder of an isolated system will always increase over time. It determines the direction of natural processes and explains why systems move toward greater chaos.
2. Can heat flow from a cold body to a hot body?
According to the Clausius Statement, heat cannot spontaneously move from a colder object to a hotter one. This transfer is possible only with external work, as in refrigerators, and results in an increase in total entropy.
3. Why can’t we build a 100% efficient engine?
The Kelvin–Planck statement explains that no engine can convert all absorbed heat into useful work. Some energy must always be lost as waste heat, making perfect efficiency physically impossible.
4. What is the difference between reversible and irreversible processes?
A reversible process is an ideal case where both the system and surroundings can return perfectly to their original states. Irreversible processes—like a broken egg—are real-world processes that always increase entropy.
5. What violates Kelvin Planck statement?
A. When the efficiency of a heat engine is greater than one, the heat engine would do work by exchanging heat with only one reservoir. This would violate the Kelvin Planck statement because the temperature of a reservoir needs to change and not be fixed at one value.
6. What violates the second law of thermodynamics?
A. A heat engine operating at 100% efficiency would violate the second law. Because, the heat engine must reject some of the heat it receives from the reservoir to the outside. Hence the heat extracted from the hotter reservoir must be given to the colder reservoir and some heat is always lost in the process.
7. Why is the second law of thermodynamics important in daily life?
A. The second law talks about entropy–a concept which dictates whether a reaction is going to be spontaneous or not. For a reaction to be spontaneous, the entropy of the system should always increase.
8. Where can the second law of thermodynamics be applied?
A. The second law can be applied where the direction of heat flow needs to be specified–heat engine, heat pump and refrigerator. They are in accordance with the second law since they operate between two reservoirs transferring heat.


