-
Call Now
1800-102-2727
Photoelectric effect, definition, experimental study, variation of photocurrent with different parameters, practice problems, FAQs
German physicist Hertz conducted experiments to see if he could pull out the free electrons which were revolving in the outermost shells of an atom. By free electrons, we refer to those electrons inside a metal which are free to move. He observed that upon heating the surface of the metal to high temperatures, they were able to be ejected from the metal, a phenomenon called thermionic emission. Another way is to shine high frequency electromagnetic waves on them. This is what is used in the photoelectric effect. It proved that light has particle nature in addition to wave nature. Let’s see more about this here in this article!
Table of contents
- Photoelectric effect
- Photoelectric effect experimental setup
- Effect of potential, intensity and frequency of incident radiation on photoelectrons
- Einstein’s photoelectric equation
- Practice problems
- FAQs
Photoelectric effect
The phenomenon of emission of photoelectrons from the surface of the metal when radiations of sufficient frequency (like ultraviolet ray) fall on it, is called photoelectric emission. The current produced in the process is called photocurrent. The incident radiation should have a high enough frequency (v) which is greater than the threshold frequency (v0) of the metal
i.e. v > v0
Photoelectric effect experimental setup
It consists of an evacuated glass tube with a photosensitive material as the cathode(C) and a collector plate acting as the anode(A). The incident light shines through a quartz window. A battery is connected to the cathode to facilitate evacuation of photoelectrons. The emitted photoelectrons strike the anode, upon which photocurrent is produced. A microammeter connected in series with the battery measures the photocurrent.

Effect of potential, intensity and frequency of incident radiation on photoelectrons
So now you have an understanding about photoelectrons and how instrumental they are in proving the particle nature of light. The intensity and frequency of the incident radiation play a vital role in photoelectric effect
- Effect of intensity on photoelectrons
Keeping the frequency of incident radiation constant, if its intensity is increased, then the number of photoelectrons striking per second increases; increasing the photocurrent.

- Effect of potential on photocurrent
The collector plate anode is initially kept positive with respect to the emitter plate. As the potential at the anode is increased, the photocurrent measured by the microammeter also increases. Even at zero potential, some photocurrent exists due to the already emitted electrons which move because of their kinetic energy. The photocurrent increases up to a certain level after which it becomes saturated since the number of electrons emitted remains constant. When the polarity is reversed, the photocurrent starts decreasing and becomes zero at a particular value of retarding potential known as stopping potential.
When light rays of different intensities are made to fall on the emitter, it is observed that at high intensity, the value of saturation current increases, as shown in the figure.

- Effect of frequency of incident radiation
With the increase in the frequency of the incident radiation, the value of saturation current remains constant whereas the magnitude of stopping potential increases and becomes more negative because the maximum kinetic energy of the photoelectrons increases.

Einstein's photoelectric equation
- Einstein proposed that there is a certain minimum frequency below which the emission of electrons is not possible. This frequency is known as the threshold frequency or cut-off frequency(vo). So it can be deduced that the generation of photocurrent is only possible when the light of sufficient frequency is made to fall on the metal plate.
- Also there was no time lag between the emission of electrons and incidence of light.
- The electrons ejected can have the maximum kinetic energy given by Einstein's photoelectric equation,
Kmax = hv -
Where
It can be seen that the kinetic energy of the electrons emitted does not depend upon the intensity of incident light but its frequency instead. Also it is to be noted that there is zero chance of two photons hitting a single electron at a time.
Practice problems
Q. The work function
(Use hc=1240 eV nm)
|
Metal |
Work function ( |
|
Li |
2.4 |
|
Cu |
4.8 |
|
Ag |
4.3 |
|
W |
4.75 |
Which of the given metal(s) will show photoelectric emission when illuminated by light of wavelength 300 nm?
A. Photoelectric emission takes place when,
Energyincident radiation > Work function(
Here, the energy of incident radiation (eV) is given by,
Since only
Therefore Li shows photoelectric emission.
Q. A metal surface is illuminated by light of two different wavelengths 250 nm and 300 nm. The maximum speeds of ejected electrons corresponding to these wavelengths are v1 and v2 respectively. If the ratio of v1 : v2 = 2 : 1. Find the work function of the metal.
(Use hc=1240 eV nm)
A.
Given,
From Einstein's photoelectric equation,
Q. The relationship between the maximum kinetic energy of electrons flying off the surface of a metal versus the frequency of light striking the metal surface as part of the photoelectric effect is shown in the graph below. According to this graph, what will happen when light of frequency 61014 Hz strikes the metal surface?

A.
From the figure, the threshold frequency of the metal,
threshold=101014 Hz
Photoelectrons are emitted only when light having a frequency larger than the threshold frequency is allowed to fall on a metal surface.
As the light used has frequency, 61014 Hz which is less than the threshold frequency, no electrons will be emitted.
Q. For photoelectric effect in a metal, the graph of stopping potential Vo (in V) versus frequency (in Hz) of the incident radiation is shown in the figure. From the graph, find the threshold frequency and work function of the metal. (Take h=6.6 10-34 Js)
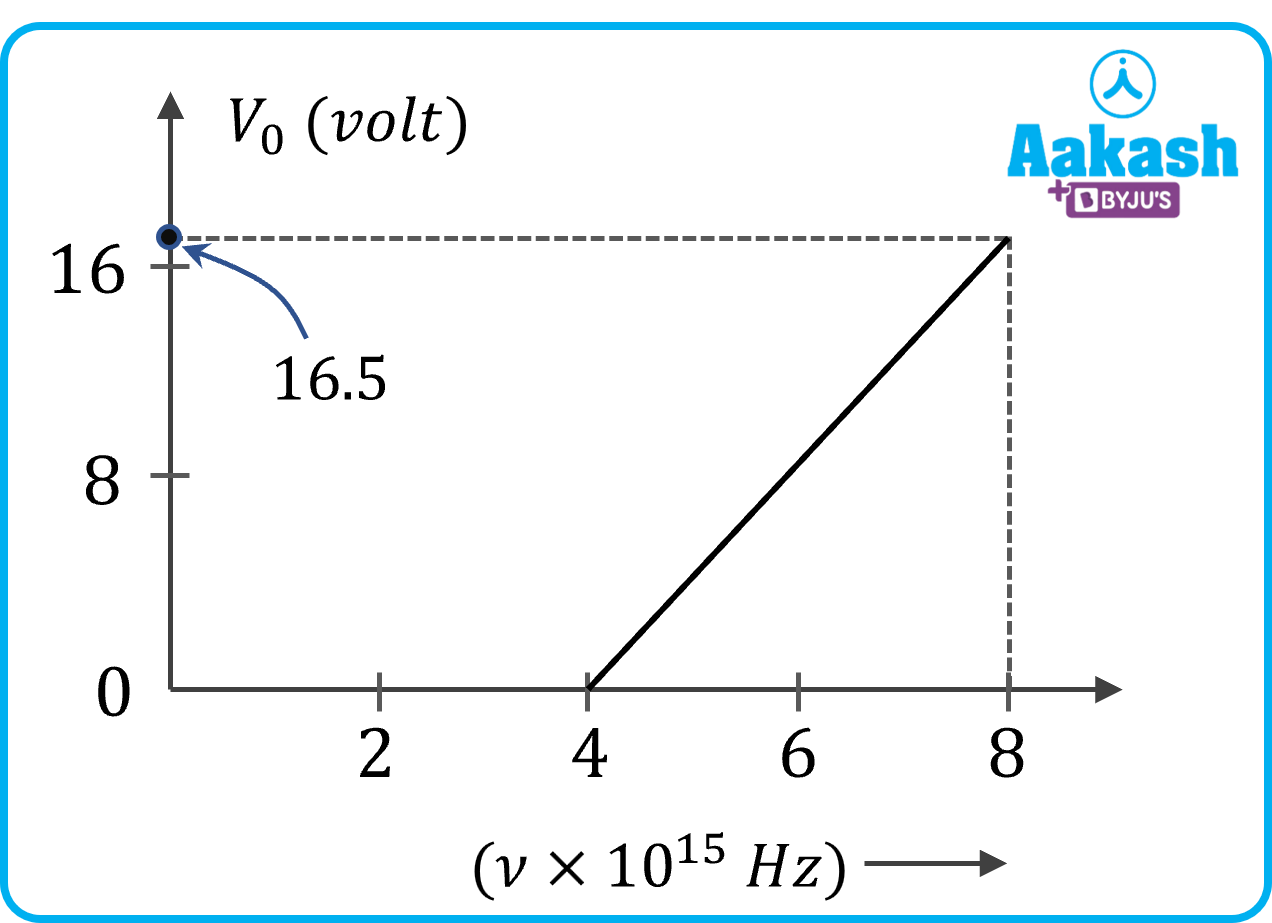
A.
From Einstein's photoelectric equation,
E=o+KEmax
E-o=eVo
Where o=Work function=ho
From, the figure we get, the threshold frequency, o=4 1015 Hz
o=ho=(6.6 10-34) (4 1015)
=26.410-19 J
=26.410-191.610-19=16.5 eV
FAQs
Q. Every metal has a definite work function. Why do all the photo-electrons not come out with the same energy, if incident radiation is monochromatic?
A. An electron is bound with the nucleus of the metal atom and has different internal energy which depends on the orbit in which they are orbiting.
When a photon is incident on the metal surface, some energy of the photon is used up to overcome the coulombic interaction, which is known as the work function of the metal, and the remaining energy of the photon becomes the kinetic energy of the emitted photoelectrons.
Maximum kinetic energy of the photo electrons, Kmax = h -o
Where, h is the energy of photons and o is the work function of metal.
K.Emax<h
Hence, photo-electrons are not emitted with the same energy as the incident photons, since they come out from different energy levels.
Q. Why can't the photoelectric effect explain the wave nature of the light?
A. Light has dual nature viz. particle nature and wave nature. Wave nature of light can not explain the particle nature of light. The photoelectric effect says photons (energy packages of light) incident on surfaces collide with electrons and hence provide them energy to jump out of the metal surface. Here, intensity of light which is associated with the amplitude of light waves i.e., the wave nature does not have any influence and the emission actually depends only on the value of frequency of incident light. The photoelectric effect itself is explained using particle nature. Wave nature cannot explain it.
Q. Give an example that is based on the application of photoelectric current.
A. Photocell is a device that converts light energy into electrical energy. It consists of a photosensitive metal plate emitting electrons when light is incident on it. They are made from a mixture of material that creates electricity for a wide range of wave-lengths.
Q. For the given experimental setup, to increase photocurrent, which of the following ways can be used?

- Source is brought near to window
- A 60 W bulb is replaced with 100 W bulb
- Frequency of radiation is increased
- Both (a) and (b)
A. (d)
If the source is brought closer to the window, the intensity of the incident rays increases. Thus, it increases the number of photons, which increases the emission of the number of free electrons from the cathode plate.
Thus, emitting more number of free electrons per second, increases the photo current.
Similarly, replacing the 60 W bulb with a 100 W bulb, increases the intensity of the source and thus increases the photocurrent.
Increasing the frequency of the source increases the energy of the incident photon and does not affect the photo current.
Hence, option (d) is the correct choice.


