-
Call Now
1800-102-2727
Moseley’s Law, Bragg’s Law, Practice Problems, FAQs
Since you were in school, you must have studied the periodic table of elements. How do you suppose we determine the atomic number of rare elements and rank them in ascending atomic number order? Have you ever wondered how scientists determine electromagnetic wave wavelengths? In this post, we'll look into Moseley's Law and Bragg's Law and see if we can figure out the solutions to these issues.
Table of Contents
- Moseley’s Law
- Application of Moseley’s Law
- Bragg’s Law
- Application of Bragg’s Law
- Practice Problems
- FAQs
Moseley’s Law
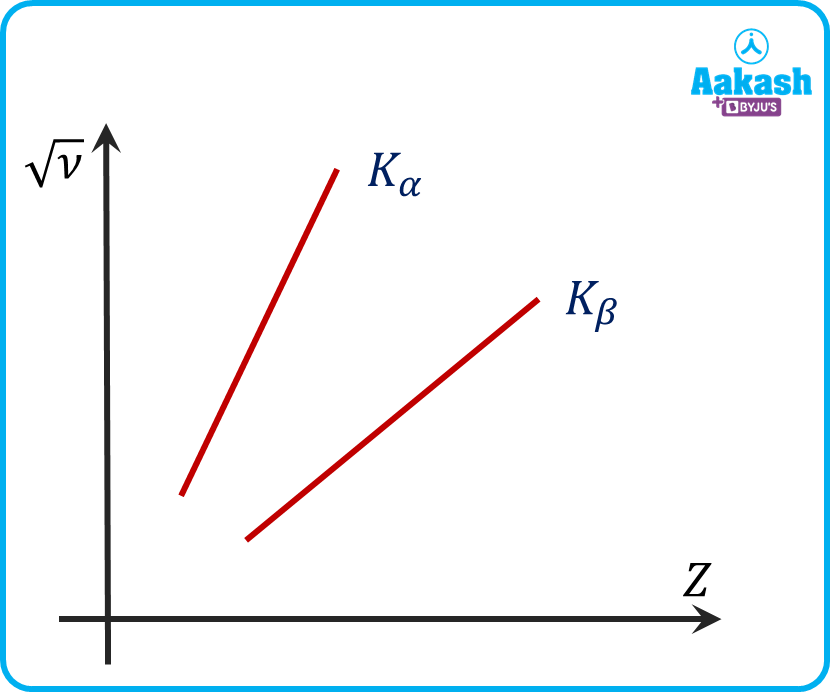
Moseley plotted the square root of frequency against the position number in the periodic table after measuring the frequencies of characteristic X-rays for a large number of elements. He observed that the plot resembles a straight line that does not pass through the origin. The relation of a straight line is expressed as
Where a and b are positive constants independent of Z. This relation is called Moseley's Law. It helps to determine the atomic number Z of an atom. Here, b is the screening constant. Following graph is drawn between √ν and Z for K and K. From the plot we can observe that a particular element curve is independent of wavelengths. The graph is a straight line and does not pass through the origin as ‘a’ and ‘b’ are non-zero constants. From the equation we can observe that the slope of the graph is equal to ‘a’.
Moseley’s Law can be derived on the basis of Bohr’s theory of atoms. Frequency of X-rays for multielectron system is given by
Here, b is screening constant and (Z-b) is the effective nuclear charge.
For K Line,
, therefore
By comparing, and for line b=1
Application of Moseley’s Law
- Moseley's law may be used to correct any inconsistencies in the periodic table's order by organising the elements according to their atomic numbers.
- New elements such as Hafnium (72), Technetium (43), Rhenium (75), and others have been discovered because of Moseley's rule.
- This equation has proved useful in calculating the atomic number of rare earths and, as a result, their periodic table location.
Bragg’s Law
Let's look at Bragg's Law to see what occurs when an X-ray collides with a crystal surface. As a result, when X-rays strike a certain atom, they cause an electronic cloud to move like an electromagnetic wave. Waves of identical frequency are generated by the movement of these charges. The re-emitted wavefields create a diffraction pattern on a film or detector, either destructively or constructively interfering with each other. The angle of scattering of an X-ray incident on a crystal surface is the same as the angle of incidence.
Bragg’s Equation
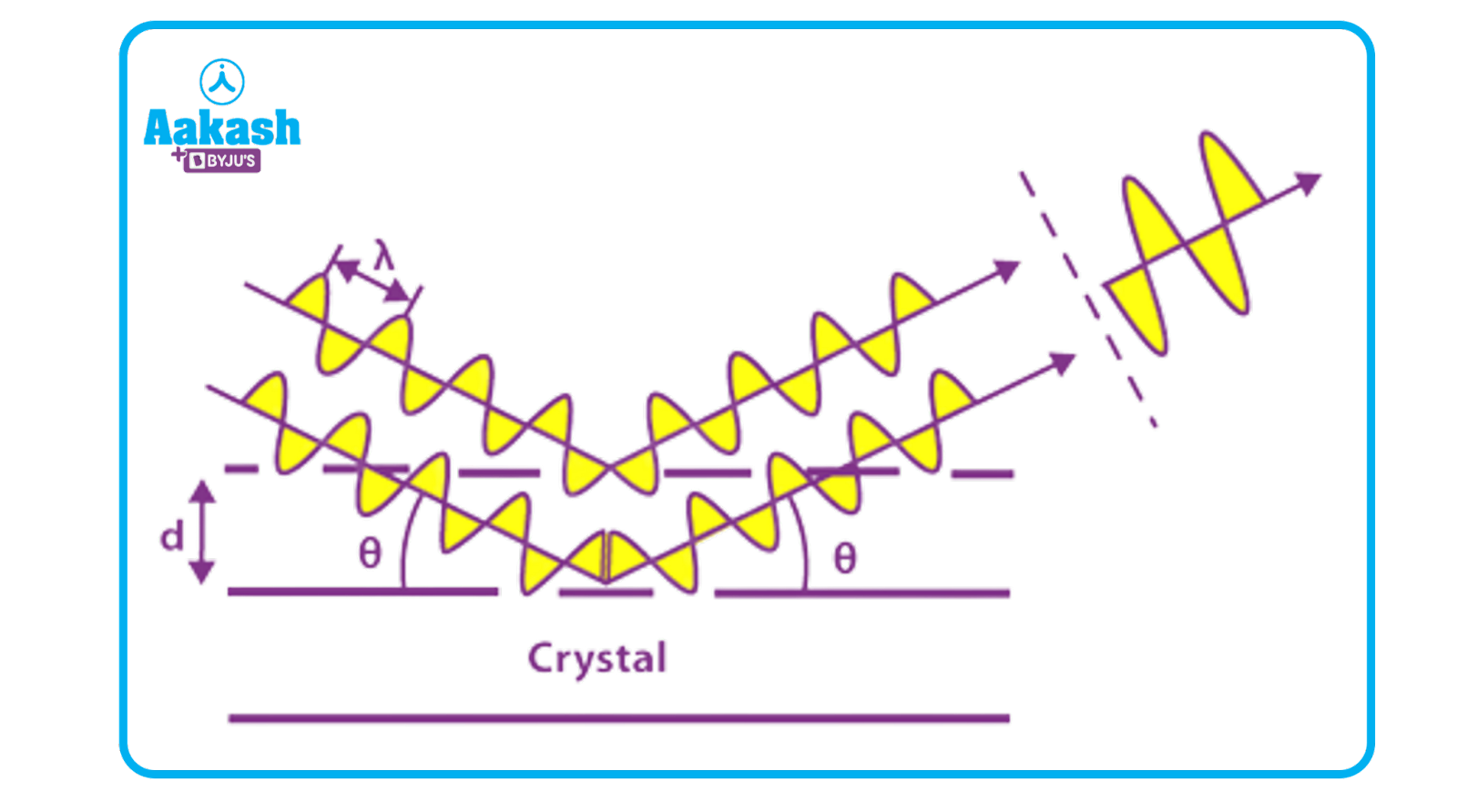
2d sin=nλ
Where, λ=wavelength of incident X-ray beam
d=distance between atomic layers
=angle of incidence
n=integer
Derivation
When the incidence angle and reflected angle are equal, the phases of the beams in the following beam diagram coincide. Up until point z, the incident beams are parallel to one another. They hit the surface and move upward when they reach point z. The second beam disperses at point B. The second beam's distance travelled is equal to AB plus BC. The additional distance is also referred to as the wavelength's integral multiple.
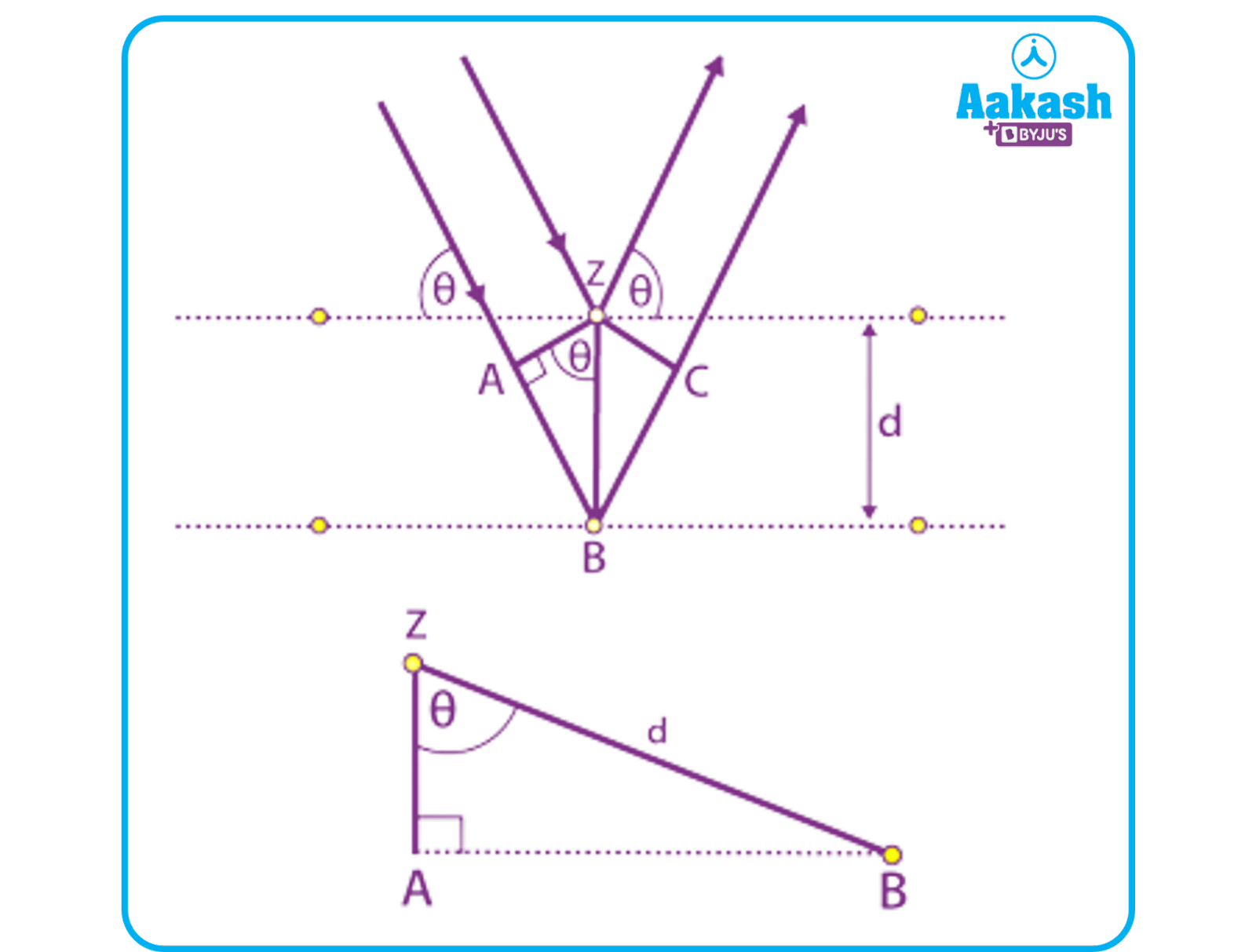
AB+AC=nλ
We also know that AB=BC
So, nλ=2AB………..(1)
From the figure we can see that, ‘d’ is the hypotenuse of right angled triangle ABZ,
So by using trigonometric ratios,
AB=dsin………….(2)
From equation (1) and (2) we can see that,
nλ=2dsin
Hence proved.
Application of Bragg’s Law
- Bragg's Law is important for determining X-ray wavelengths.
- It's also used to figure out how crystals' lattice spacings work.
Practice Problems
Q. If the K radiations of Mo (Z = 42) has wavelength of 0.71 Å, calculate the wavelength of the corresponding radiation of Cu, i.e. K for Cu (Z=29) assuming b=1?
A. According to Moseley’s law,
Q. Compare Z1 and Z2?
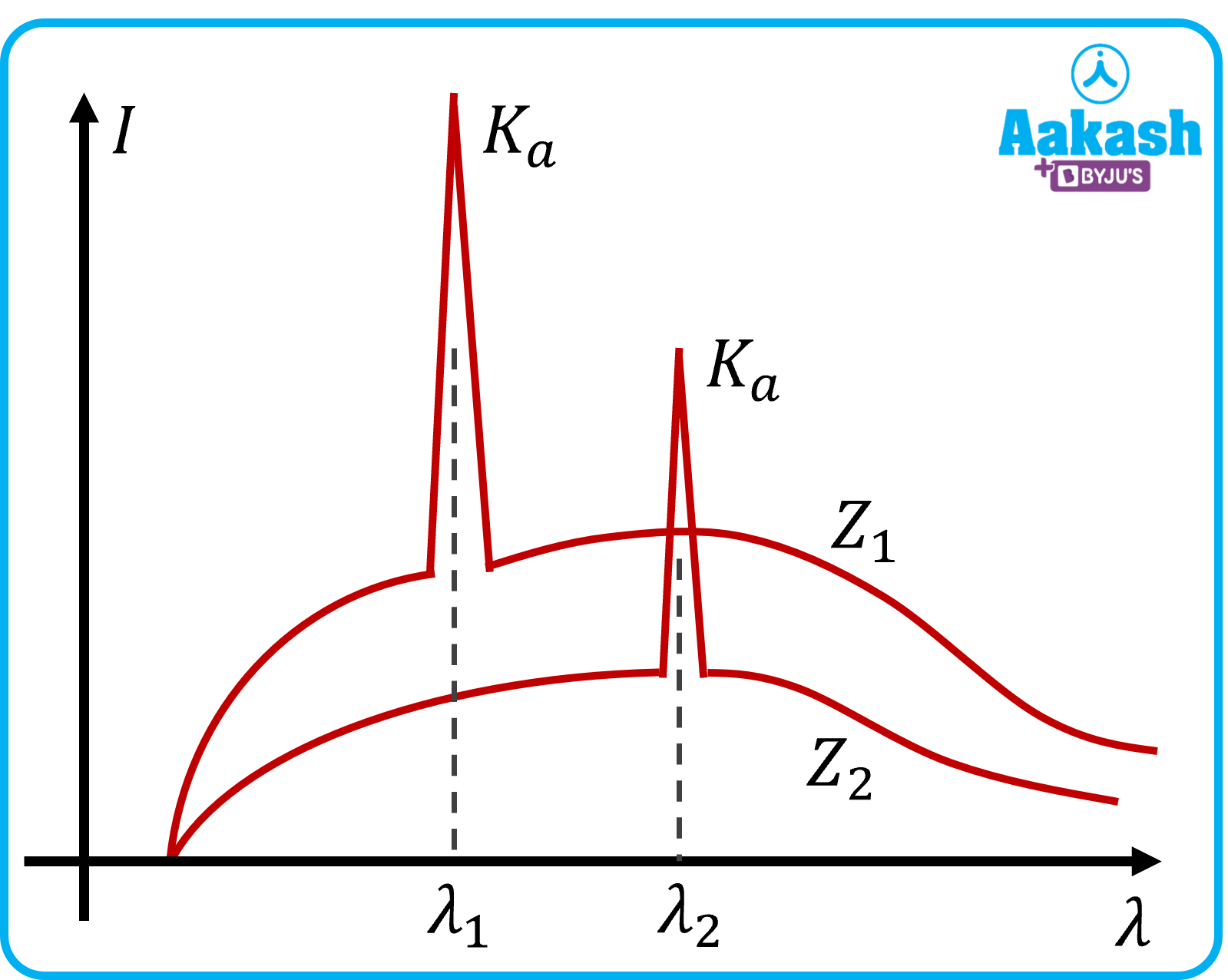
A. we know,
So if Z is greater, then ν will be greater and hence λ will be less.
Since
Q. Find the constants “a” and “b” in Moseley’s equation ν= a(Z-b) from the following data.
|
Element |
Z |
Wavelength of K X-ray |
|
Mo |
42 |
71 pm |
|
Co |
27 |
178.5 pm |
A. According to Moseley’s law,
For Mo, c….(1)
For Co, ……..(2)
Subtracting above equation (2) from (1)
By solving we get,
Dividing equation (1) by (2),
By solving we get,
b=1.37
Q. Find the wavelength of X-ray incident which show maxima at angle 300 on the surface having distance between atomic layers is 5 nm?
A. Given, d=5 nm=510-9 m and =300
From Bragg’s Law,
FAQs
Q. What is the use of Bragg’s Law?
A. The Bragg law is important for estimating crystal lattice spacings and measuring wavelengths.
Q. According to Bragg’s law, what parameters are interrelated?
A. Bragg’s law gives a relationship between the wavelength of X-rays and distance between atomic layers.
Q. How can we derive Moseley’s Law?
A. We can not derive Moseley’s Law mathematically. It is an empirical law deduced from laboratory experiments and observations.
Q. What is the role of Moseley’s law in the periodic table of elements?
A. This law is helpful in determining the atomic number of rare earths, thereby fixing their position in the periodic table in increasing order of atomic number.


