-
Call Now
1800-102-2727
Accelerating potential - Definition, Examples, Practice Problems, FAQs
When a charged particle at rest starts moving, it acquires kinetic energy in the process. For that to be possible, it needs to be supplied a certain amount of potential energy. In other words, a stationary charge can be accelerated when a definite potential difference is applied. In order to do so, a certain amount of voltage has to be applied between two electrodes. Such a voltage is called accelerating potential. In this article, we will explore accelerating potential in detail.
Table of contents
- What is accelerating potential?
- Uses of accelerating potential
- De-Broglie wavelength
- Practice problems
- FAQs
What is accelerating potential?
When a charged particle is at rest, it needs to be supplied with a certain potential difference between two electrodes. This potential difference would be responsible for accelerating the charge. Let us consider a charged particle of mass m and charge q. The KE of the particle when accelerated by an accelerating potential V can be written as,
Here, qV denotes the product of the charge and the accelerating voltage, which is also equal to the work done in accelerating the particle.
The SI unit of accelerating potential is volt. Its dimensional formula is
Uses of accelerating potential
X-rays are produced when energetic charged particles are stopped by a target metal of high atomic mass. They are produced in an evacuated tube called Coolidge Tube. The tube consists of a cathode, which supplies electrons by thermionic emission. (electrons are ejected when heated). A high potential difference V is maintained between the anode and the cathode.
This serves as the accelerating potential for the electron.
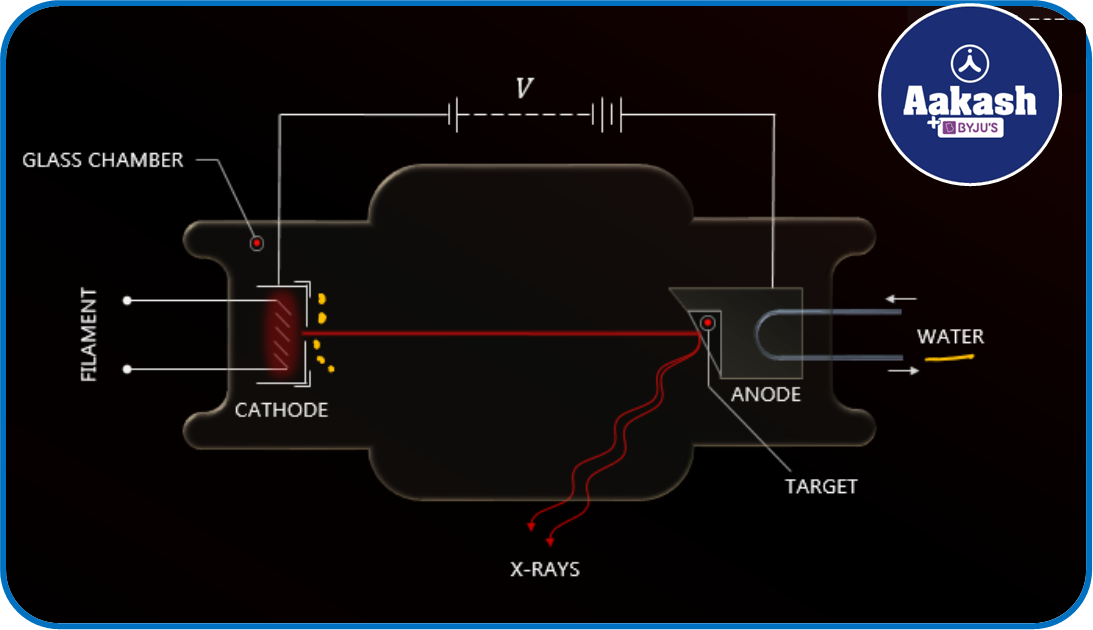
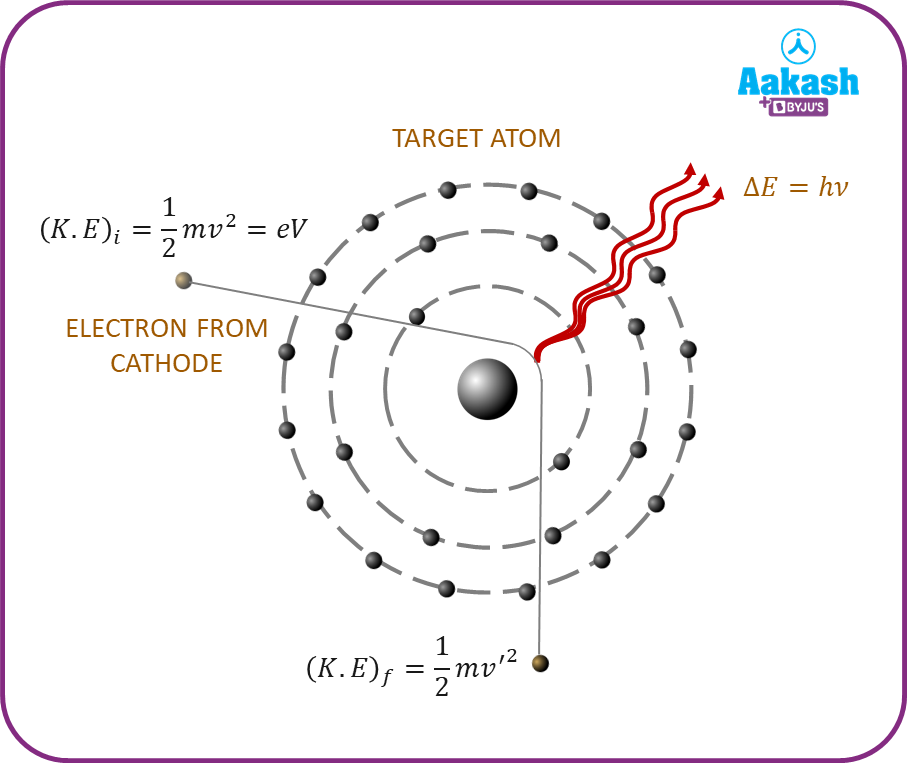
Continuous X-rays are said to be produced when accelerated electrons lose their energy upon entering the target material and are finally brought to rest. A part of this kinetic energy gets converted into the energy of photons, and the remaining energy is used to heat up the target element. If E represents the energy of a photon, then
E=h
- frequency of the photon.
Where h- Planck’s constant
- wavelength of the light used.
c-speed of light
But energy is also equal to eV, where V is the accelerating potential.
min-minimum or cutoff wavelength of the X-rays produced.
e-electronic charge
Substituting the known values of h,c and e
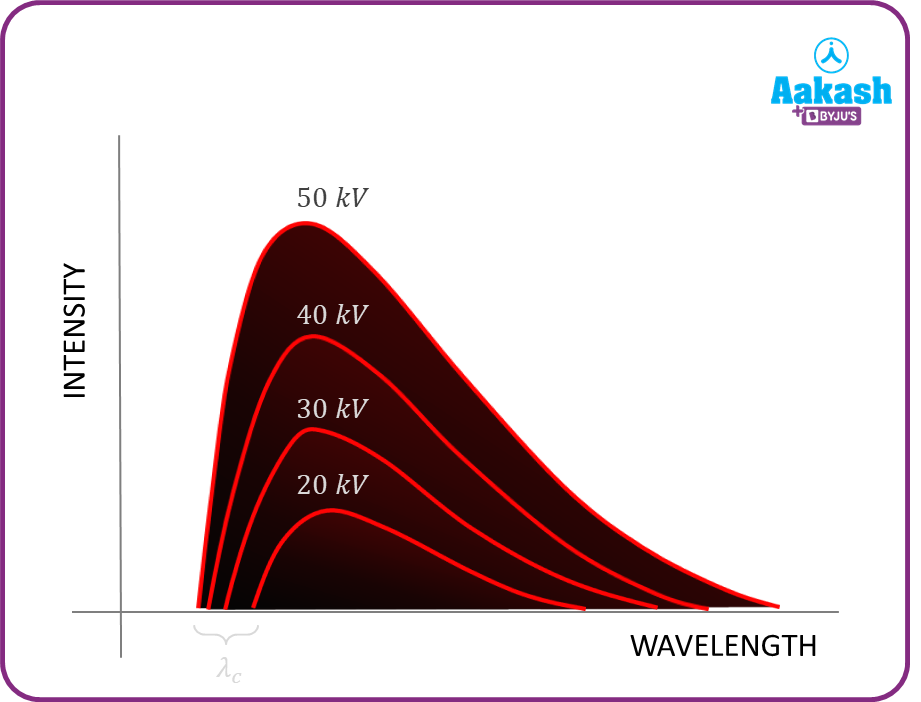
The above diagram shows the variation of intensity of the X-rays with the different wavelengths of radiation produced corresponding to the different accelerating voltages. The accelerating voltage is minimum for higher wavelengths and vice versa.
De-Broglie wavelength
De-Broglie stated that a moving object has a wave associated with it. These waves are called matter waves or de-Broglie waves. Let us consider a particle of mass m moving with a velocity v. Then the momentum p associated with the particle is mv.
If the particle in question travels with the speed of light, then the energy of the particle, E=mc2
Also, energy
Equating the above two equations, we get
Where is the de-Broglie wavelength. If the particle travels with velocity v
If a charged particle acquires a kinetic energy E when accelerated by a potential V, then p=2mE
Now energy E=qV where q denotes the charge of the particle.
If m=me denotes the mass of an electron, q=e denotes the charge, then the above equation becomes,
Similarly, for a proton,
For an alpha particle,
Practice problems
Q. Calculate the cut-off wavelength of the continuous X-rays coming out from an X-ray tube at 40 kV.
A. Given,
The minimum cut-off wavelength,
Q. If V denotes the accelerating voltage, then the maximum frequency of continuous X-rays is given by,
(a)ehV (b)hVe (c)eVh (d)heV
A. c
Let max indicate the maximum frequency. Then equating energies, we get
Q. Calculate the ratio of the momentum of an electron and an particle, which are accelerated from rest by applying a potential of 100 V.
A.
Since,
Ratio of momentum of electron and alpha particle,
The charge of an alpha particle is +2e
Q. Calculate the ratio de-Broglie wavelength of an alpha particle and an electron which have the same accelerating voltages.
A. For an alpha particle,
For an electron,
Required ratio,
FAQs
Q. What are particle accelerators?|
A. Particle accelerators like cyclotron, betatron, synchrotron are used to accelerate and increase the energies of subatomic particles like electrons or alpha particles. The cyclotron for instance, makes use of high accelerating voltage applied between two giant magnets.
Q. To what speeds can we accelerate electrons?
A. Electrons can be accelerated very close to the speed of light, to about 0.996 c where c is the speed of light. When accelerated to such high velocities, their mass would undergo relativistic variation, and they would emit gamma radiation.
Q. Give one application of accelerating potential.
A. Accelerating potential is used to produce fast moving charges. Accelerating potential applied between the cathode and the anode is responsible for imparting kinetic energy to the charged particle. The higher the accelerating potential applied, higher is the kinetic energy of the particle.
Q. How does voltage affect the wavelength?
A. The wavelength of the X-rays produced is inversely proportional to the accelerating potential. Higher the accelerating potential, lower is the wavelength of the X-rays produced and higher is the intensity of the X-rays produced.


