-
Call Now
1800-102-2727
Water of Crystallization - Introduction, Hydrated salts, Uses, Practice Problems & FAQs
Have you visited any fairs around you? You must find magic shows there.
Rahul, a 12-class student went to a magic show where a magician jack was performing magic and a huge crowd was there applauding Jack.
Rahul and his friends bought the tickets for a magic show and went inside the hall, where Jack was performing his unique magic. When the show started Jack and his team brought a table where there was one beaker, a stand and in a beaker, he poured a blue-coloured solution. Now Jack describes the brief of his magic show, he told the audience that without doing or touching the beaker the color of the solution starts fading and the sometimes blue-coloured solution gets disappeared into a colorless solution. The same thing happened, the audience started hooting and they started clapping.
Rahul and his friends were science students so they were finding the logic behind this. Then they decided to ask their chemistry teacher. They show the video which they recorded during the show. Their teacher explained that there must be a burner inside the covered stand and the solution in the beaker may be hydrated copper sulfate containing water of crystallization. When heating the solution, the water crystals disappear and the solution looks colorless.

Here we are studying the topic related to our magic show. Yes, we will cover water of crystallization in a bit more detail. We will learn its application and properties.
Let’s begin!
TABLE OF CONTENT
- Water of crystallization
- What are hydrated salts?
- Example of water of crystallization
- The action of heat on hydrated salts
- Practice Problems
- Frequently asked questions-FAQs
Water of crystallization:
The water molecules that make up a salt crystal's structure are referred to as the water of crystallization. The water of crystallization is water which is chemically joined with a crystal structure.
Another definition for this can be stated as: A few water molecules serve as a crucial structural element in the crystal structure of certain salts. Water molecules that build up a crystal's structure are considered as "water of crystallization." Salts that have been hydrated with the water that crystallizes are known as hydrated salts. The crystallization of water, and hydrated salts, as well as the impact of heat on hydrated salts, are all explained in depth here.
The water crystallizes when crystals are made entirely of water form. These crystals are free of contaminants. These crystals are significantly impacted by heat. The water that crystallizes, sometimes referred to as hydrated water, is composed of the water molecules that crystals contain.
Water is commonly present while making crystals from the source of aqueous solutions. The entire amount of water in any substance at any specific temperature is known as the water of crystallization, and it often exists in a specific ratio in diverse situations. "Water of crystallization" refers to water that is present in the crystalline structure of any metal complex or salt but is not attached to the metal cation immediately.
Many chemicals which also absorb some water molecules into their crystalline structures after crystallizing from water or we can say that from water-containing solvents. By heating a sample, the water of crystallization can usually be removed, but the crystalline qualities are usually lost. For instance, at room temperature, sodium chloride dihydrate is unstable. Proteins crystallize with a lot more water in the crystal lattice than inorganic salts do.
Examples of crystallization water include proteins that contain more than 50% of water and copper sulfate pentahydrate, which contains five molecules of crystallization water.
What are hydrated salts?
- A crystalline form of the salt molecule which is weakly bound to a few water molecules is known as a hydrated salt. Salt is produced when an anion of the acid and cation of that base combine to form an acid-base molecule. A salt molecule that is anhydrate is one that is not attached to any water molecules, whereas a hydrated salt is one that is attached to water molecules.
- A hydrated salt has water molecules integrated into its crystalline structure. When many water molecules which are bonded to the ions in a salt's crystalline structure, the salt is said to be hydrated.
- Such water molecules are commonly known as hydrated waters or crystallization fluids. Salts that have retained water while crystallizing are known as hydrated salts.
Each hydrated salt's single "formula unit" has a predetermined number of crystallization water molecules.
Example of water of crystallization:
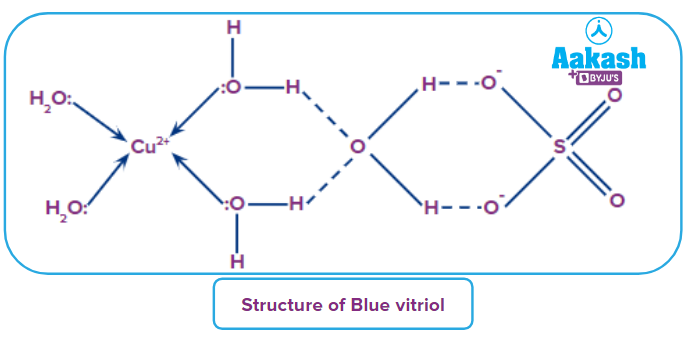
- Crystals of copper sulfate, CuSO4.5H2O, contain 5 molecules of the water used in crystallization. Its chemical name is pentahydrate of copper sulfate.
- Gypsum include two crystal molecules of the water in one single formula unit, calcium sulfate crystals, sometimes referred to as gypsum crystals, are written as CaSO4.2H2O. It is also known as calcium sulfate dihydrate.
- Because each unit of the formula contains ten water molecules during the crystallization process, sodium carbonate crystals, sometimes referred to as washing soda crystals, are written asNa2CO3.10H2O. You can call it sodium carbonate decahydrate.
- At the crystal lattice, water molecules can be found in interstitial spots or voids. For instance, the two water molecules in barium chloride dihydrate, BaCl2.2H2O, fill the spaces in the crystal lattice.
The action of heat on hydrated salts:
High temperatures cause hydrated salts to lose their crystallization water. When the water of crystallization is gone, the hydrated salts lose their characteristic shape and color and transform into colorless powdery particles. Since anhydrous salts lack the water necessary for crystallization, they hydrate when water is supplied, restoring their natural color.
This will be made clearer by the example that follows.
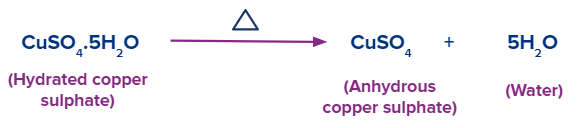
When heated aggressively, the blue-coloured form of copper sulfate crystals becomes white because water is lost during crystallization. Dehydration of copper sulfate crystals is a process that is reversible. As a result, hydrating anhydrous copper sulfate with water causes it to become blue and hydrate, producing hydrated copper sulfate.

Anhydrous copper sulfate becomes blue when water is added. The ability of anhydrous copper sulfate to identify the presence of water or moisture in a liquid is used. Several drops of the solution which need to be tested are then added to the white-coloured anhydrous form of copper sulfate powder. The emergence of blue color indicates the presence of available moisture in anhydrous copper sulfate.
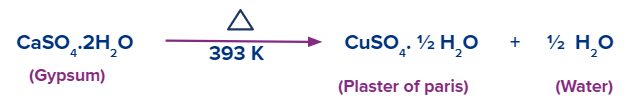
From gypsum (CaSO4.2H2O), plaster of Paris (CaSO4.12H2O) is created. Gypsum loses its water molecules when heated to 393 K, turning into calcium sulfate hemihydrate (Plaster of Paris).
Uses of Hydrated Salt:
- The most popular use of these hydrated salts in our daily life is Epsom salts (MgSO4.7H2O). The human body needs many of the substances that are present in salts. But some substances could be hard to digest or only available through food. In areas where hydrated salts are naturally found, people have long taken therapeutic baths in the belief that they have healing properties. As an illustration, consider Epsom salts.
- Epsom salt is economically more potential as a home cure even if its therapeutic effects haven't been established.
- The industry is a significant area in which hydrated salt is used. Salt, especially hydrated salts, is used in many different industrial processes. Hydrated salts are an important component of many businesses.
- More than half of these products in the industry’s principal component list are salt-based. industries including glass, rubber, paper, and textiles also use hydrated salt. In both commercial and residential water softening agents or salt is also used as a water softening salt. Furthermore, due to its potential to sustain a consistent temperature over a lengthy period of time, hydrated salt is widely used in the alternative energy industry.
- Salts that have been hydrated can exist in a number of environments, including freshwater.
- Due to the salt's adaptable crystalline structure, it may quickly hydrate by joining with water molecules. When sodium chloride, or salt, comes into contact with liquid water, it collects water vapor from the air.
- When the substances in a particular area's soil or rock dissolve and mix with the groundwater, finally getting hydrated with the water molecules, free-flowing chemicals, for instance, produce salt molecules.
Practice Problems:
Q1. Soda ash when hydrated with water of crystallisation____________ is formed.
a. Washing powder
b. Caustic soda
c. Baking soda
d. Lunar caustic
Answer: A
Solution: Soda ash, also known as anhydrous sodium carbonate (Na2CO3), is a frequent term. When washing soda dissolves in water, it re-crystallizes to produce crystals that contain 10 water molecules giving sodium carbonate decahydrate.

Q2. __________salt is known as white vitriol.
a. MgSO4.7H2O
b. ZnSO4.7H2O
c. FeSO4.7H2O
d. CuSO4.5H2O
Answer: B
Solution: The heptahydrate water of crystallization, having the formula ZnSO4.7H2O is known as white vitriol. CuSO4.5H2O is known as blue vitriol in chemistry.FeSO4.7H2O is considered as green vitriol and MgSO4.7H2O is Epsom salt.
Q3. Calculate the total number of atoms present in blue vitriol.
a. 20
b. 21
c. 22
d. 3
Answer: B
Solution: Blue vitriol contains a total of 21 atoms. The chemical formula is CuSO4.5H2O . It contains 5 molecules of water of crystallization. When heated this water of crystallization will lose and we get anhydrous copper sulfate.
Q4. Among the following, which term is used to represent the water of crystallization?
a. Buffer solution
b. Hydrate
c. Anhydrous
d. Aqueous
Answer: B
Solution: Hydrate is the correct term used for water of crystallization. We use hydrated salt which contains water crystals attached to them. The anhydrous term is related to salt containing no water. Aqueous is a physical state where the solvent present is water. The buffer solution is a substance that doesn't change its pH when acid or alkali are introduced. Hence, Hydrate is the correct answer.
Frequently asked questions-FAQs:
1. Salts that include water of crystallization seem to be completely dry. Why?
Answer: A component of water's crystal structure is the water that crystallizes. The salt is not moistened because the water used for crystallization is not free water. As a result, the salts that were formed during crystallization seem to be completely dry.
2. How can you tell that the water of crystallization is beneficial for salt crystals?
Answer: The form and, in certain situations, the color of salt crystals are imparted by the water of crystallization. For instance, iron sulfate crystals acquire a green hue due to the presence of crystallization water.
3. What color are crystals of FeSO4.7H2O ? When it is heated, what should happen to the color of the salt ?
Answer: Crystals of FeSO4.7H2O have a green color. When heated, it loses the water necessary for crystallization and becomes anhydrous ferrous sulfate. This salt is reddish-brown in color.
4. What influences the crystallization of water?
Answer: The water that crystallizes is sometimes referred to as hydration water or crystallization water. The water crystallizes when pure crystals from an aqueous solution form. These crystals are free of contaminants. These crystals are significantly impacted by heat.


