-
Call Now
1800-102-2727
Unit Cell: Lattice, crystal systems, definition of Unit Cell, Number of atoms in a unit cell, Unit cell constant, Effective number of atoms, Practice Problems, FAQs
Have you ever been involved in a group project? If you have done that activity then you know that in every group project there’s one student who does most of the work and definitely there must be a student who will go into hibernation during the work and suddenly appears when the submission happens.
Well, similar phenomenon happens in a crystal. Just like the way one project was assigned to many students and everyone has a different amount of contribution, similarly in a crystal lattice, particles are connected to one or more than one unit cell. Hence, it is important to find out how an atom contributes in a unit cell and what percentage they share.
You might heard about crystalline salt(NaCl) and crystalline sugar? But of the two adjectives one is correct and of another wrong. We have crystalline salt. Crystalline sugar is a misnomer.


Pure solids are of two types- amorphous and crystalline. Crystalline solids have a definite pattern, with a small unit repeating in all three directions. But amorphous solids do not have any such repeating units and is an assembly of small atoms or molecules in random order. Salt is a giant molecule meticulously arranged in a pattern while sugar is amorphous but looks like a crystal.
You will study here about the order and arrangement of atoms /ions in crystalline solids.
Table of Content
- Lattice and Lattice types
- Seven Crystal system
- Contribution of Atoms in a Unit Cell
- Effective number of atoms in a unit cell
- Practice Problems
- Frequently Asked Questions (FAQs)
Types of lattice
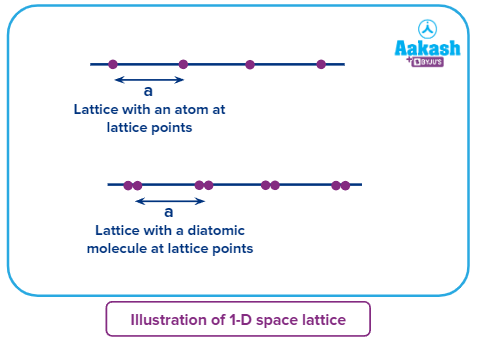
1-D (One-dimensional) space lattice
Atoms or ions are considered as spherical particles. In solid state atoms are in close contact with each other in perticular directions and separated in some other directions.
For representation, the atoms can be considered as point charges, located at their nucleus and separated by a distance.. This representation is called lattice points
● If we arrange atoms/ions touching each other in a line (one dimension), Lattice points are uniformly separated in a one-dimensional space lattice.
● Only one parameter (edge length - a) is required to define a 1-D space lattice and that is the distance between any two consecutive lattice points. Requirements to fix the arrangement is called as lattice parameter.In one dimensional lattice the lattice parameter is only one, namely length.
Repeating units can be observed at every lattice parameter. The unit is the basic or fundamental unit of the entire lattice like a polymer made of monomer in one direction.
● In a 1-D space lattice, lattice points can be either occupied by a single
atom/ion (case A) or by a polyatomic molecule (case B).
2-D (Two-dimensional) space lattice
● Regular arrangement of lattice points in a plane(on a surface) gives a two-dimensional space lattice.
● The lattice is defined by three parameters: two edge lengths a and b, and the angle between them .
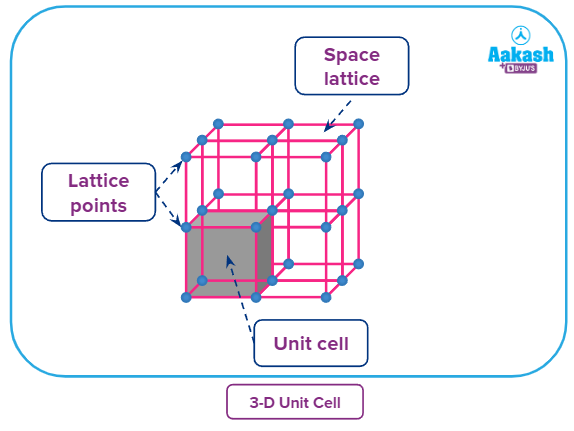
3-D (Three-dimensional) space lattice
● The regular arrangement of lattice points in space will give a three-dimensional space lattice.
● The lattice points in space can be fixed In terms of the six lattice parameters (3 lengths and 3 angles). The crystalline solid, repeats the small unit with the lattice parameters in three dimensions. The small repeating unit repeating to make the entire crystal is called unit cell.,
The lattice parameters in 3D space lattice can be arranged in seven unique unit cell shapes known as crystal systems. The crystal systems have varying elements of symmetry in a three-dimensional space.
Seven Crystal system and Bravis lattices
Almost all the crystals found in nature conform to one of these seven crystal systems. The basic repetitive units present in these system are of 14 types and called as Bravis lattice systems. As a whole the unit cell of all the solid shall fall under any of these 14 Bravis lattice types with differing lattice parameters.
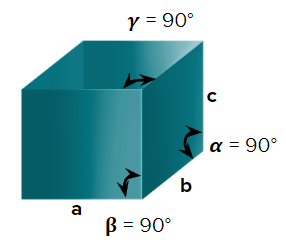
- Cubic
- Axial Length : a=b=c & Axial angle: ===900
- NaCl, ZnS, Cu, KCl, Alums, Diamond, Zinc blende
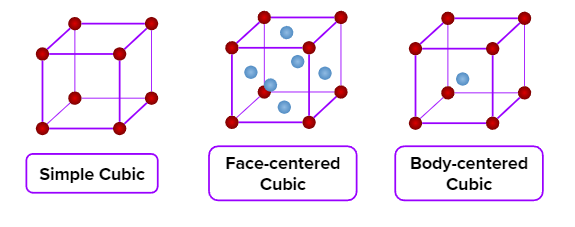
- Simple cubic (primitive), body-centred cubic, and face-centred cubic unit cells are possible in a cubic crystal system.
- Tetragonal
- Axial Length: a=bc & Axial angle: ===900
- White tin(Sn), SnO2,TiO2,CaSO2
- Simple (primitive) and body-centred unit cells are possible in a tetragonal crystal system.
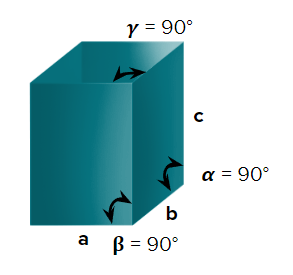
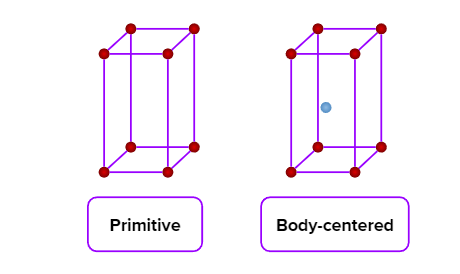
- Orthorhombic or Rhombic
- Axial Length: abc & Axial angle: ===900
- KNO3,K2SO4,BaSO4, PbCO3, CaCO3Rhombic sulfur
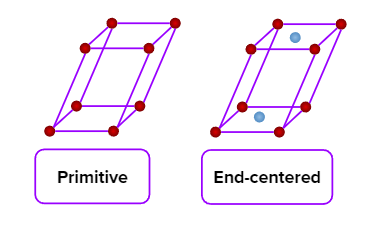
- Simple (primitive), end-centred, body-centred, and face-centred unit cells are possible in an orthorhombic crystal system.
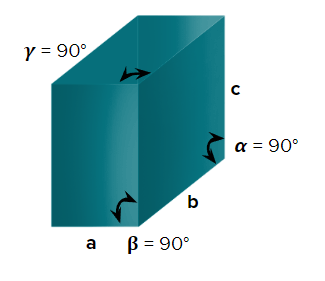
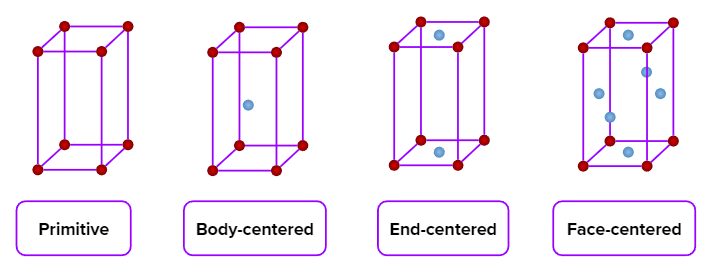
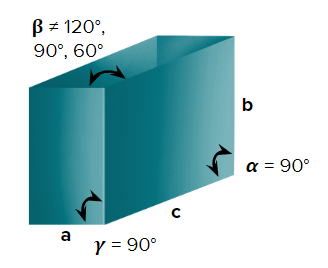
- Monoclinic
- Axial length: abc & Axial Angle: ==900
- Na2SO4. 10H2O,PbCrO4, Monoclinic Sulfur
- Simple (primitive) and end-centred unit cells are possible in a monoclinic crystal system.
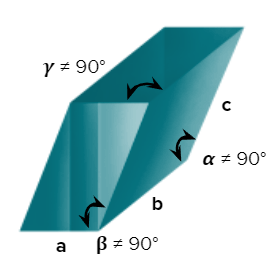
- triclinic
- Axial Length: abc & Axial angle: 900
- CuSO4, 5H2O, K2Cr2O7, H3BO3
- Only a simple (primitive) unit cell is possible in a triclinic crystal system.
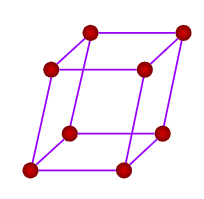
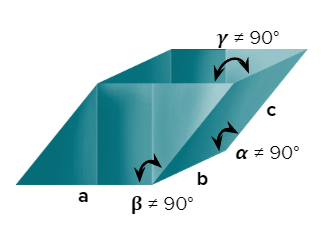
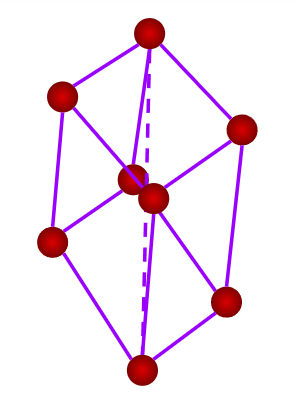
- Rhombohedral or Trigonal
- Axial Length: a=b=c & Axial angles: ==900
- NaNO3, ICl, As, Sb, Bi, Calcite(CaCO3)
- Only a simple (primitive) unit cell is possible in a rhombohedral crystal system.
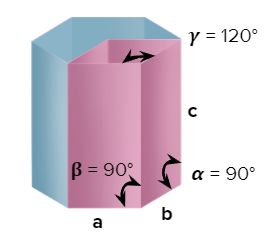
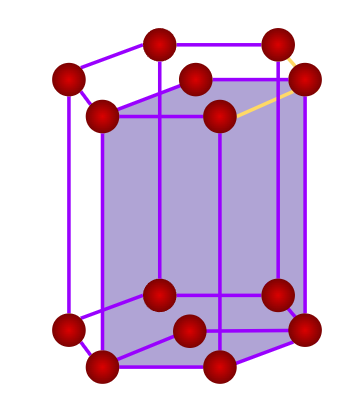
- Hexagonal
- Axial Length: a=bc & Axial Angle: ==900, =1200
- Cinnabar(HgS), Ice Graphite, Mg, Zn, Cd, ZnO, CdS, AgI, PbI2
- Only a simple (primitive) unit cell is possible in a hexagonal crystal system.
Contribution of Atoms in a Unit Cell
In a crystal lattice a particle present at a point may be shared by a number of unit cells, as each unit cell is touching a number of other unit cells.
Here we will calculate:
- Portion or fraction of the particle that belongs to a particular unit cell.
- Number of particles in the unit cell of different lattices of a cubic crystal system.
Calculation of the contribution of atoms present at different lattice sites
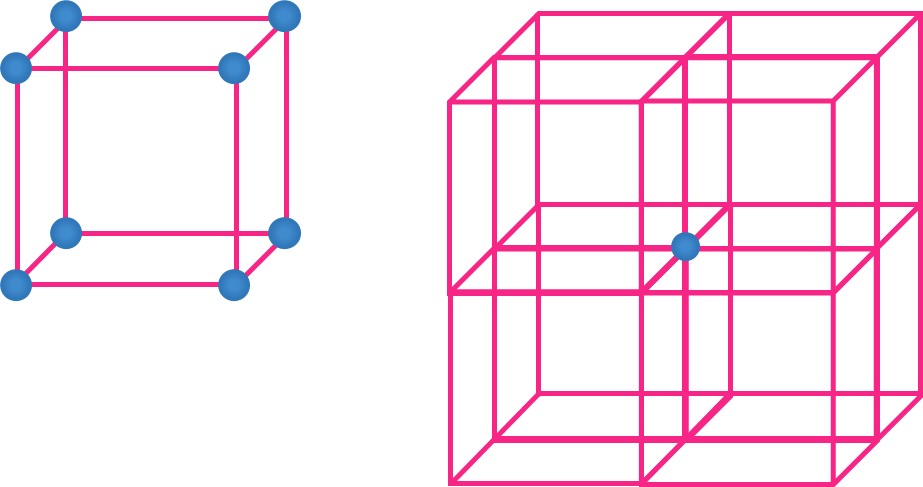
- Atom at the corner
- Eight unit cells share an atom in the corner..
- Hence, Contribution of each atom present at the corner = 18
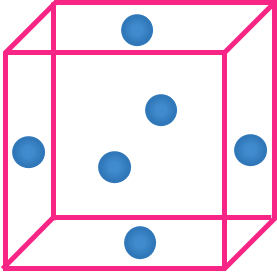
- Atom on the face
- A single atom on the face is shared by two unit cells.
- Hence, Contribution of each atom on the face = 12
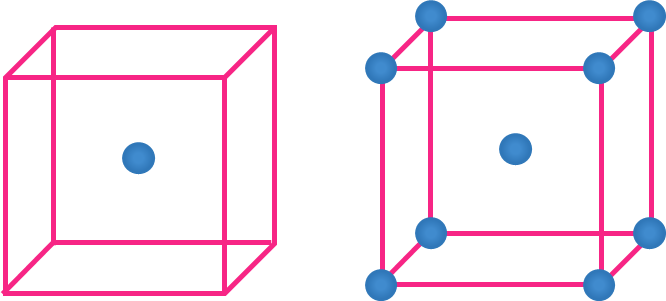
- Atom within the body
- No other unit cell shares an atom found within the body of the unit cell.
- Hence, Contribution of each atom within the body = 1
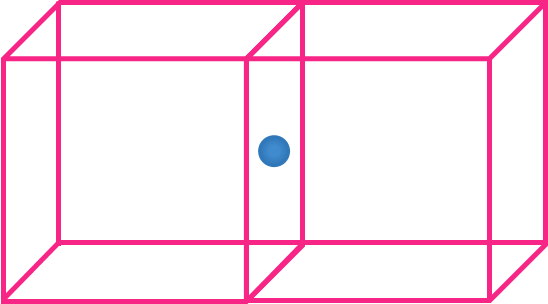
- Atom on the edge
- Four unit cells share an atom located on the edge.
- Hence, Contribution of each atom on the edge = 14
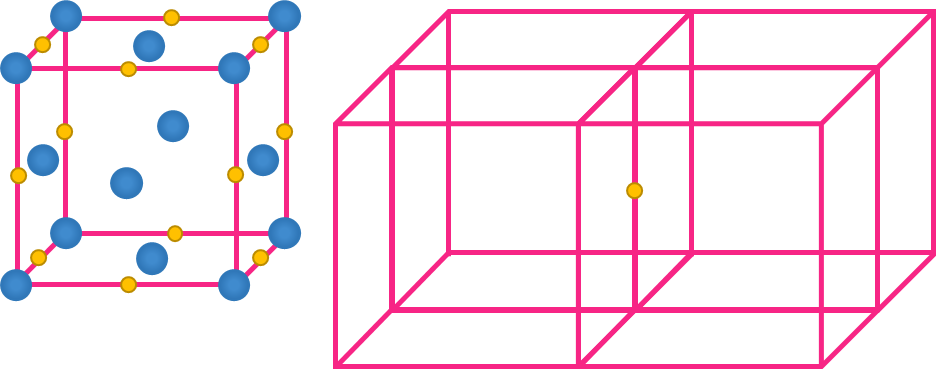
Effective number of atoms ( Z) in a unit cell
The effective number of particles ( Z) in a unit cell can be calculated by adding the contribution of each constituent particle to the unit cell.
Where,
Nc= Number of particles at corners of a unit cell
Nf= Number of particles at face-centre of a unit cell
Nb= Number of particles at body-centre of a unit cell
Ne= Number of particles at edge-centre of a unit cell
Practice Problems
Q1. A compound formed by the elements A and B has a cubic structure, with A atoms at the cube's corners and B atoms at the face centres. Determine the compound's formula.
- AB2 B) AB3 C) AB4 D) AB
Answer: (B)
Solution: As A atoms present at the 8 corners of the cube
Number of atoms of A in the unit cell = = 1
As B atoms are present at the face centres of the 6 faces of the cube
Number of atoms of B in the unit cell =
Ratio of atoms = A:B = 1:3
Hence, formula of the compound formed by element A & B is AB3
Q2. An ionic compound composed of atoms A and B has a face-centred cubic arrangement, with atoms A at the corners and atoms B at the face centres. What is the compound's simplest formula if one of the atoms is missing from the corner?
- A24B7 B) A21B7 C) A7B21 D) A7B24
Answer: (D)
Solution: Since one atom is missing at the corner therefore,
No. of atoms of A at the corners of the cube = 7
Contribution of atoms A towards unit cell =
No. of atoms of B at the face centres = 6
Contribution of atoms B towards unit cell =
Ratio of A:B =7 = 7:24
Hence, Formula of the compound is A7B24
Q3. Calculate the number of unit cells in 8.1 g of Aluminium if it crystallises in a FCC structure. (Atomic mass of Al=27 g mol-1)
- B) C) D)
Answer : (A)
Solution: 1 mole of Al = 27 g = 61023atoms
No. of atoms present in 8.1 g of
As 4 atoms are there at FCC unit cell, therefore,
Number of unit cells present
Q4. A cubic solid consists of two elements, X and Y. Atoms Y are present at the cube's corners, and atoms X are present at the centre of the body. What is the compound's formula? What are the coordination numbers for X and Y?
- 3 and 2 B) 4 and 4 C) 8 and 8 D) 1 and 1
Answer: (C)
Solution:
As atoms Y are present at the 8 corners of the cube, therefore, number of atoms of Y in the unit cell =
Because atoms X are present at the body center, the number of atoms X in the unit cell equals one.
Ratio of atoms X:Y = 1:1
Hence, the formula of the compound is XY
And coordination of each of X and Y =8.
Frequently Asked Questions (FAQs)
Q1. What is coordination number in a crystal structure?
Answer: The coordination number in a crystal is the number of constituent particles which are the immediate neighbours of that particle in a crystal. In case of ionic crystals, the coordination number of an ion in the crystal is the number of oppositely charged ions surrounding that particular ion.
Q2. Who introduced the term unit cell?
Answer: Only these 14 types of unit cells are compatible with the orderly groupings of atoms observed in crystals, which was demonstrated by the French physicist Auguste Bravais in 1850.
Q3. Is it possible for a unit cell to have more than one type of atom?
Answer: Yes, it is possible. For example: A BCC unit cell has two atoms: one-eighth of an atom at each of the eight corners (818=1 atom from the corners) and one atom in the middle. Every atom in this arrangement comes into contact with four atoms in the layer above it and four atoms in the layer below it.
Q4. Is there any difference between lattice point and unit cell?
Answer: A lattice is a regular recurring three-dimensional organisation of atoms, ions, or molecules in a metal or other crystalline solid, whereas a unit cell is a simple arrangement of spheres (atoms, molecules, or ions) that resembles a lattice's repeating pattern.