-
Call Now
1800-102-2727
Thin Layer Chromatography – Principle, Experiment, Procedure, Types, Applications, Disadvantages, Practice Problems and FAQ
At some point of time in our lives, we as students have had the privilege of using fountain pens, isn’t it? And quite obviously some of us might have stained our dresses with the same ink. When the ink doesn't adhere effectively to the cloth, as is occasionally the case, we may clean the shirt by washing it. But sometimes, when it strongly sticks to the fabric, no matter how many times the item is cleaned in bleach and detergent, the ink stains remain permanently on the cloth or change its colour slightly.

This is synonymous with the degree to which chemical compounds adhere to a solid often allowing them to be distinguished from one another. Chromatography is the process of separating the components of a mixture using this variation in "stickiness."
When we dip the ink-stained cloth in water slowly, the ink mixes with water and gradually moves up spreading. And if instead of water we use detergent solution, the ink stain while moving up changes its colour and might show up two different hints at a distance from each other, on the same fabric. This actually depends on the varying degree of stickiness of the constituting chemicals present in the ink. And this very logic is what forms the basis of “Thin Layer Chromatography”
Excited to know more? Let's get started!
TABLE OF CONTENTS
- Thin Layer Chromatography – Introduction
- Thin Layer Chromatography – Principle
- Thin Layer Chromatography – Retention factor
- Thin Layer Chromatography – Instrumentation
- Thin Layer Chromatography – Working
- Thin Layer Chromatography – Applications
- Thin Layer Chromatography – Disadvantages
- Practice Problems
- Frequently Asked Questions – FAQs
Thin Layer Chromatography – Introduction
Thin layer chromatography (TLC) is an affinity-based chromatographic separation technique that is majorly applicable to non-volatile mixtures. M. Tswett made the discovery of chromatography in 1906.
On a sheet of glass, plastic, or aluminium foil that has been covered with a thin layer of an adsorbent substance—typically silica gel, aluminium oxide, or cellulose (blotter paper)—thin layer chromatography is done.

The stationary phase refers to this adsorbent layer. A solvent or solvent combination (referred to as the mobile phase) is dragged up the plate by capillary action after the sample has been placed on the plate. Separation is accomplished because various analytes ascend the TLC plate at different speeds.
The sample is inserted vertically into a closed chamber with an organic solvent after being spotted onto one end of the TLC plate (mobile phase). Due to their differing affinities for the stationary and mobile phases, sample components migrate over a range of distances when the mobile phase is propelled up the plate by capillary forces. The plate is taken out of the developing chamber and dried after the solvent has reached the top of it. The separated components show up as spots on the plate, and each component's retention factor (RF) is evaluated.
Thin Layer Chromatography – Principle
The competition between the solute (analyte) and the mobile phase for binding sites on the stationary phase is the basis for compound separation.
In thin layer chromatography, the solid phase is a thin glass plate covered with either silica gel or aluminium oxide. A solvent is selected for the mobile phase based on the characteristics of the mixture's constituent parts. The distribution of a component between a liquid mobile phase (eluting solvent) that is moving across the solid phase and a solid fixed phase (the thin layer) placed on a glass or plastic plate is the basis of TLC. A starting point slightly above the bottom of the TLC plate is covered with a little quantity of a compound or combination.
In the developing chamber, which includes a small pool of solvent immediately below the level at which the sample was applied, the plate is next developed. Through capillary action, the solvent is pulled up through the plate's particles; as it passes over the mixture, each component either stays in the solid phase or dissolves in the solvent and travels up the plate. Whether a compound climbs the plate or lags behind depends on its physical characteristics and, consequently, on its molecular structure, particularly its functional groups. The "Like Dissolves Like" solubility rule is adhered to.
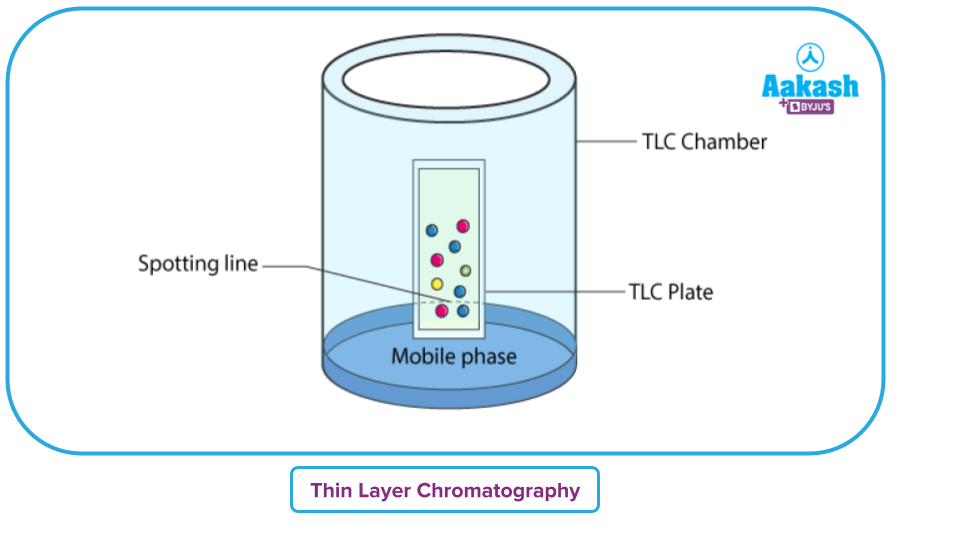
The more physically similar its characteristics are to those of the mobile phase, the longer an analyte will stay in the mobile phase. The most soluble substances will be carried up the TLC plate the farthest by the mobile phase. The substances that have a greater affinity for the stationary phase particles (adsorbent) on the TLC plate and are less soluble in the mobile phase will remain back.
After the separation procedure is complete, the mixture's constituent components show up as spots at the appropriate levels on the plates. Suitable detecting techniques are used to determine their type and character.
Thin Layer Chromatography – Retention Factor
In paper and thin layer chromatography, components' movement rate is stated as follows using the parameter, RF, which is commonly known as Retardation Factor or Retention factor.
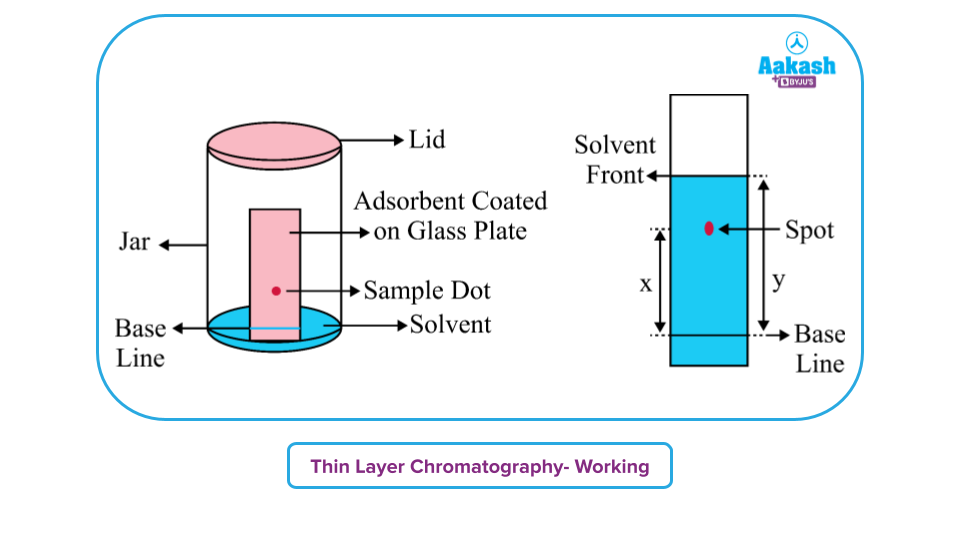
The mobility of compounds along the TLC plate is gauged using the retention factor (RF). Its value is always in the range of 0 and 1. For example, in the given image below, the solute has moved till the spot (represented by x). And solvent went up till y.
So,
For the same solvent, various adsorbents will provide different RF values. Only an adsorbent with a consistent particle size and binder may be reproduced. Before usage, plates should be kept over silica gel in desiccators, and samples should be applied rapidly to avoid the plate absorbing atmospheric water vapour. It is much preferable to use plates that have been held at room temperature rather than activating them due to the challenges involved in the activation operations.
Thin Layer Chromatography – Instrumentation
Before beginning the Thin Layer Chromatography experiment, let's grasp the many steps and components needed to carry out the method.
Thin Layer Chromatography Plates (Silica gel plates): The TLC plates are pre-made, chemically stable plates. A small coating of the stationary phase is placed on its surface. The stationary phase on the plate has uniform thickness and tiny particle size.
TLC Chamber: Develop plates in a chamber for thin layer chromatography. It is accountable for maintaining a stable atmosphere that will aid in the development of spots. Additionally, it stops the solvent from evaporating and keeps the process completely dust-free.
Mobile Phase: A solvent mixture or a solvent makes up the mobile phase, which is the part of the reaction that moves. This stage ought to be devoid of particles. The formation of spots is better as purity quality increases.
Stationary Phase: (Paper/porous alumina sheets) for thin layer chromatography must be put inside the chamber. In the mobile phase, it is wet.

Thin Layer Chromatography – Working
Stationary phase is created using glass, plastic, or aluminium, covered with a thin coating of an adsorbent substance, most frequently silica gel or aluminium oxide, It is inert and dry.
- With the use of a pencil, make tiny markings at the plate's base where sample spots will be applied.
- To the designated areas, apply the sample solutions.
- Place a moistened filter paper in the mobile phase to ensure an even humidity level when you pour the mobile phase into the TLC chamber.
- After putting the plate inside the TLC chamber, cover it with the lid. The sample is kept towards the mobile phase at all times.
- Submerge the plate to promote growth. Maintain in mind to keep the sample locations far above the mobile phase. Avoid submerging it in the solvent.
- Wait until spots start to appear. Take out the plates and dry them when the stains have emerged. A UV spectrophotometer may be used to view the sample spots and analyse them.


Thin Layer Chromatography – Applications
- TLC is applicable in qualitative testing of a variety of drugs, including sedatives, local anaesthetics, anticonvulsant tranquilisers, analgesics, antihistamines, steroids, and hypnotics.
- In biochemical analysis such as the separation or isolation of biochemical metabolites from blood plasma, urine, bodily fluids, serum, etc, TLC is incredibly helpful.
- It is possible to detect natural compounds using thin layer chromatography, such as volatile or essential oils, fixed or fixed oils, glycosides, waxes, alkaloids, etc.
- It is frequently employed to separate complex medicinal compositions.
- It is employed to clean samples, and a direct comparison between the sample and the original sample is made.
- To distinguish and identify colours, sweeteners, and preservatives in the food sector.
- It is employed in the cosmetics sector.
- It is also applied to study the extent of completion or kinetics of a reaction.
- It is used in forensic studies and crime-related sample detections.
Thin Layer Chromatography – Disadvantages
- The stationary phase is not long enough in thin layer chromatography plates so it is limited to a certain quantity of sample mixture alone.
- The separation time is short in comparison to other chromatographic methods.
- TLC produces outcomes that are challenging to duplicate.
- Since TLC is an open system, variables like humidity and temperature may have an impact on the chromatogram's ultimate result.
- Its inability to distinguish between certain isomers and enantiomers is one of its drawbacks.
- It exclusively uses qualitative analytical methods; it does not use quantitative methods.
Practice Problems
- How is the RF value of an analyte present in a mixture dependent on its polarity?
Solution: For instance, silica gel employed as the stationary phase in a normal phase reaction might be regarded as polar. When two compounds with different polarities are present, the more polar molecule interacts with silica more strongly and is, therefore, better able to remove the mobile phase from the binding sites. As a result, the less polar compound (analyte) ascends the plate (resulting in a higher RF value).
All compounds on the TLC plate will rise higher up the plate if the mobile phase is changed to a more polar solvent or combination of solvents because it is better able to dispel solutes from the silica binding sites. Practically speaking, this implies that increasing the amount of ethyl acetate in the mobile phase, which is a combination of heptane and ethyl acetate, raises the RF values for all the compounds on the TLC plate.
- Thin Layer Chromatography is a method that is used to separate:
- Non-volatile mixtures
- Volatile organic compounds
- Volatile mixtures
- Same composition mixture
Answer: A)
Solution: Thin layer chromatography (TLC) is an affinity-based chromatographic separation technique that is majorly applicable to non-volatile mixtures that do not sublimate or evaporate at room temperature.
So, option A) is the correct answer.
- Which of the following is used in setting up a TLC on working mode?
- Silica gel
- Alumina
- Cellulose
- All of the above
Answer: D)
Solution: On a sheet of glass, plastic, or aluminium foil that has been covered with a thin layer of an adsorbent substance—typically silica gel, aluminium oxide, or cellulose (blotter paper)—thin layer chromatography is done. They act as the stationary phase.
So, option D) is the correct answer.
- TLC is based on the principle of
- Absorption
- Adsorption
- Precipitation
- Sedimentation
Answer: B)
Solution: The principle of thin layer chromatography is adsorption-based separation. The separation depends on the relative affinities of chemicals/analytes to the stationary and mobile phases.
So, option B) is the correct answer.
Frequently Asked Questions – FAQ
1. What is the significance of TLC?
Answer: Thin-Layer Chromatography (TLC) offers a wide range of applications in pharmaceutical analysis since it is less time-consuming, cheaper, and easier to execute. TLC can separate 32 amino acids if done correctly. It has several uses in locating contaminants in a chemical. It can be used as an interim analytical technique before HPLC (High-Performance Liquid Chromatography). Simple TLC principles and minimum sample preprocessing are often required. TLC may be used to identify chemicals in a sample and track the development of a reaction. In a combination, TLC is also used to separate the same chemicals. Numerous industry-standard techniques in environmental toxicity, food chemistry, water, inorganic and pesticide analysis, dye purity, cosmetics, plant materials, and herbal analysis.
2. Can the RF value be equal to 1?
Answer: RF value is always between 0 and 1. RF values are never greater than or equal to one. The spot and solvent front move too closely together and consequently, the method of separation becomes vague and unreliable in that case when the RF value is 1 or too close to it.
3. Why is Al2O3 plates used in TLC
Answer: The aluminium oxide layer is chemically inert and has a large surface area for adsorption. It has a pore size of around 60 Å and a specific surface area of about 200 m2g-1. For the separation of weakly polar chemicals, alumina is preferred over silica gel because of its simple surfaces. Alumina is available in several forms, just as silica gel.
4. How are colourless spots (corresponding to various analytes) of a mixture detected in TLC?
Answer: To detect colourless spots, usually a UV lamp source or fluorescence lamp is used. The TLC plate's silica gel is infused with a fluorescent substance that lights when exposed to ultraviolet (UV) radiation. A spot that obstructs the fluorescence will be visible as a black spot against a brilliant backdrop. The spots can be marked with a pencil to indicate their positions when they are exposed to UV light.



