-
Call Now
1800-102-2727
Thermodynamic Processes - Reversible, Irreversible, Isothermal, Adiabatic, Isobaric, Isochoric, Cyclic Processes, Practice Problems, FAQs
Why am I feeling restless?

It was 101°F, and I knew something was wrong.
We know when we fall sick, the first thing the doctor checks are- body temperature, heart rate, blood pressure, etc, right?
And based on that he tells us about what’s wrong and gives us medicines accordingly.
So we can say- body temperature, heart rate, blood pressure, etc, are some of the crucial parameters that describe the condition or state of the human body.
Similarly, in a thermodynamic system, it’s the holy trinity of the variables - Pressure, Volume and Temperature – that majorly defines the state of a system - particularly for a gaseous one.
A system or simply a body changes its physical state defined by the variables P, V and T in different ways. Thermodynamic processes can be defined as the method or process by which a system undergoes a change of state with different thermodynamic properties.
Everything around us is dynamic and undergoes physical or chemical changes. All the exposed iron gets more and more rusted. Motor vehicles burn fuel to move. Water cooled in the fridge becomes solid ice crystals. Earth gets warmer with sun rays to cool back in the evening.
If we consider all these changes, iron metal becoming ferric oxide, liquid fuel being converted to carbon dioxide, liquid water changing to solid ice and earth undergoing a cyclic process of getting heat to come back to the initial temperature, though are different, have one thing in common. That they all involve the exchange of energy in their different forms. Again changes(or processes) like rusting and burning of fuel are one-way processes and cannot be reversed -an irreversible process. On the other hand, the formation of ice and hot earth can be brought back to their original state of liquid water and cold earth- a reversible process.
Come let us understand this phenomenon in more detail.
Some of the types of thermodynamic processes are as follows-.
Table of Contents
- Reversible Process
- Irreversible Process
- Isothermal Process
- Isobaric Process
- Isochoric Process
- Adiabatic Process
- Cyclic Process
- Practice Problems
- FAQs
Reversible Process
A thermodynamic process (state i → state f ) is said to be reversible if the process can be turned back such that both the system and the surroundings return to their original states by reversing the infinitely small changes that caused. These are usually closed system processes. They also operate on maximum efficiency in ideal working conditions and thus the target design can be set.
Examples of Reversible Process
Here, we have listed a few examples of reversible processes:
- Electrolysis
- Melting of ice at 0°C
- vaporation and condensation of a volatile liquid in a closed container.
- Acid hydrolysis of esters.
Irreversible Processes
An irreversible process is one that, once started, cannot be reversed by changing the factors that cause the change. This includes the system and its surroundings. Using an automobile engine as an example, a distance of 'x' miles has been covered by the combustion of fuel. The process involves the fuel burning to produce energy for the engine while simultaneously transforming it into smoke and heat energy. If we take the vehicle back to the starting position, we cannot recover the energy or gasoline that was expended.
Accordingly, based on their ability to change back to their initial condition from their final state, certain processes can be classified as reversible and irreversible in nature.
Examples of Irreversible Processes
A few examples of Irreversible Processes are:
- Burning of a fuel
- Reactions involving gasses.
- Open system processes
- Electricity flow through a resistance
|
Reversible process |
Irreversible process |
|
Reversible processes are infinitely slow changes such the system and surroundings are always at equilibrium with each other. |
Irreversible processes represent rapid changes occurring in the system so that the system will never be in equilibrium with the surrounding during the process. Very fast process. . |
|
The direction of the reversible process is determined by external conditions. Change of conditions at any stage during the process can reverse the direction of the process. |
This is a one-way process and will not be reversed by a change in conditions. |
|
The difference between the internal and external forces is very small. |
The driving force of the change is larger and gets wasted as heat is lost to the surroundings, making it irreversible. |
|
As there is no waste of energy, work done during a reversible process will always be the maximum possible. |
As there is a loss of energy, work done in an irreversible process is always less than that of a reversible process. |
|
A reversible process can be brought back to the initial state without making any change in the surroundings. |
An irreversible process cannot be brought back to its initial state without making a change in the surroundings. |
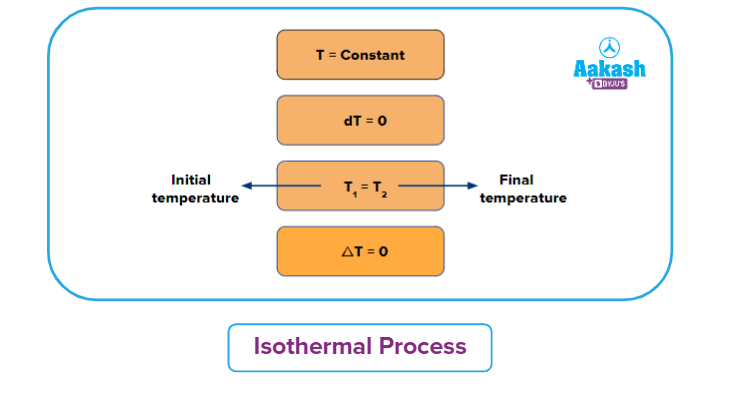
Isothermal Process
It is a thermodynamic process in which the temperature will remain constant throughout the process. Thus, in an isothermal process, dT (for infinitesimally small change) or ΔT (for a finite change) is equal to zero. Boyle's law states that the product of pressure and volume is constant for a given amount of gas as long as the temperature is constant. Therefore, isothermal processes obey Boyle's law.
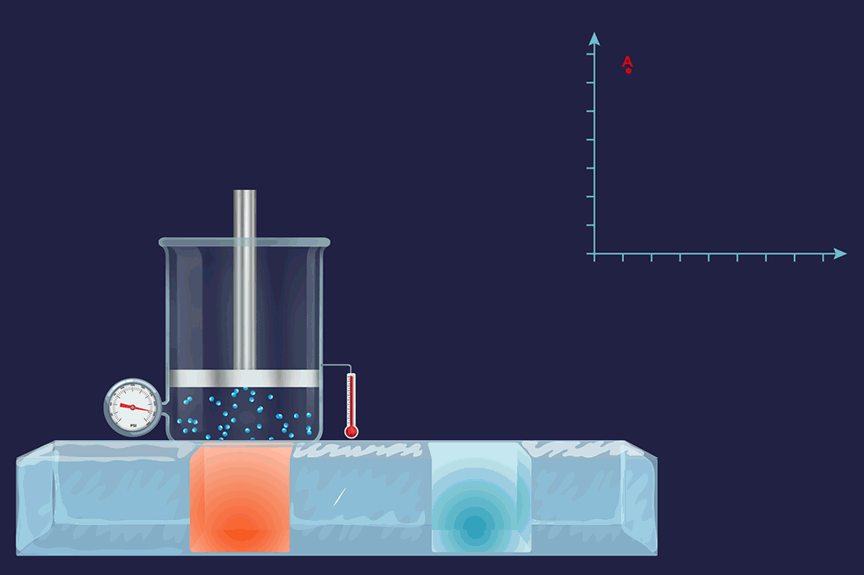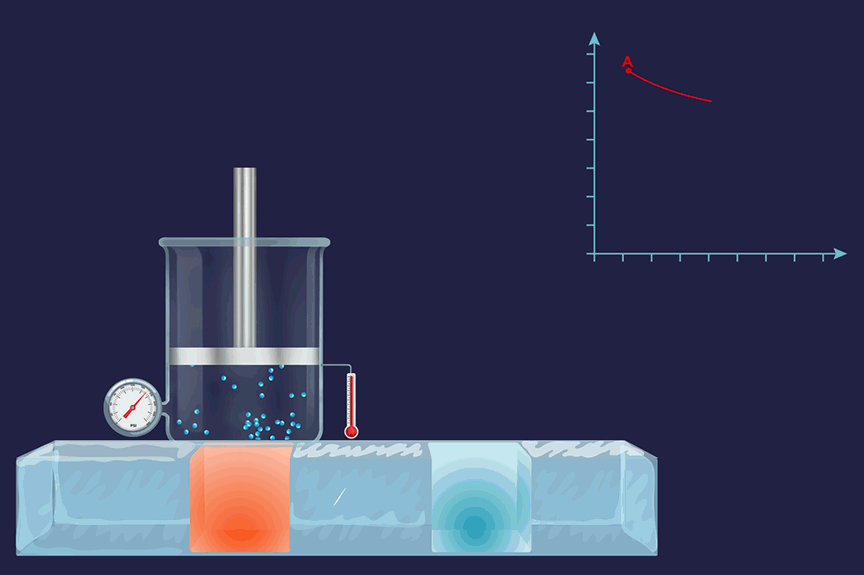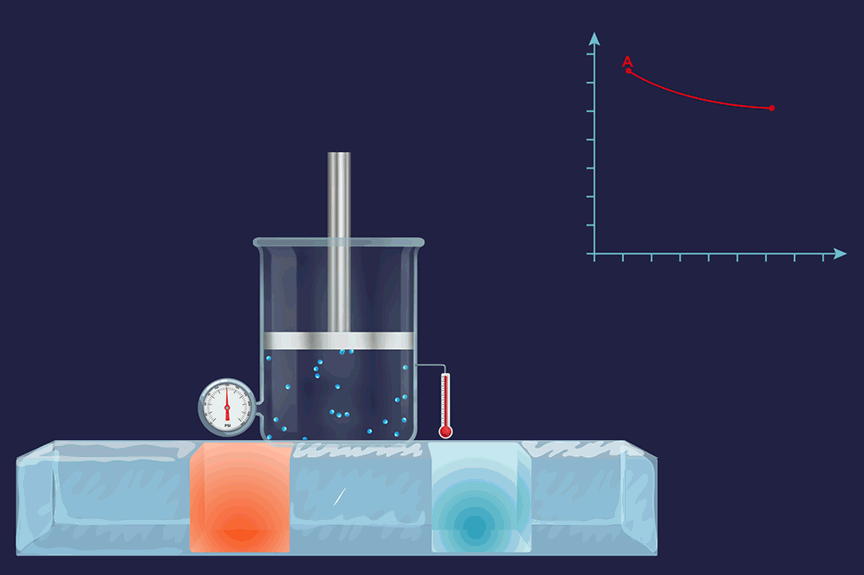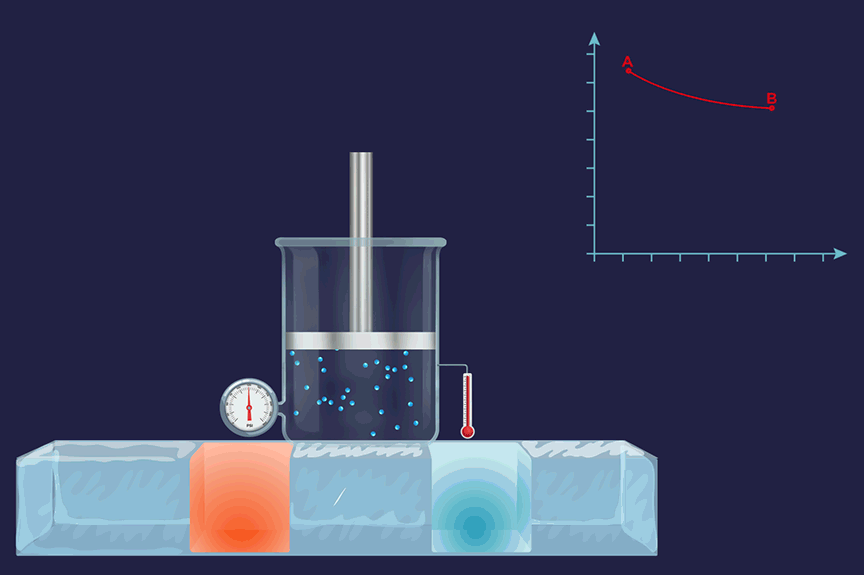
An illustration of an isothermal thermodynamic process is given. During the melting of ice, the thermometer reading will remain constant throughout the process.

Isobaric process
It is a thermodynamic process in which the pressure will remain constant throughout the process. Thus, in an isobaric process, dP (for infinitesimally small change) or ΔP (for a finite change)is equal to zero.
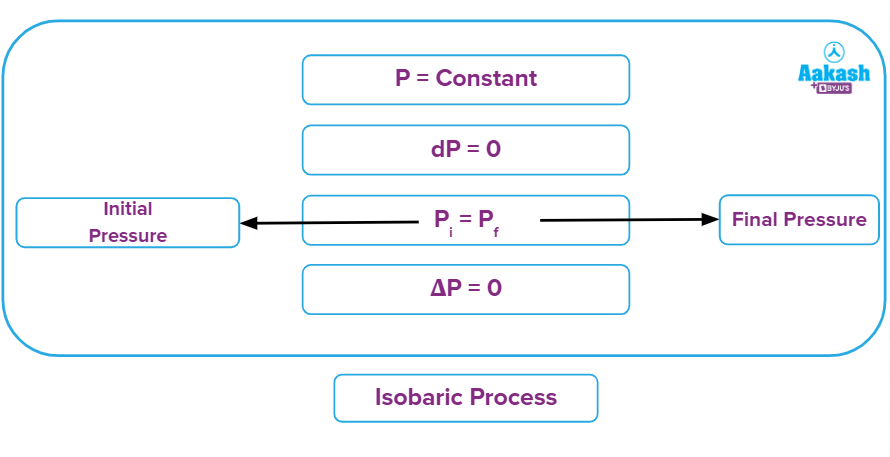
Every process happening in a system which is exposed to the atmosphere is an isobaric process as the atmospheric pressure will remain constant. Thus, cooking food inside an open utensil that is exposed to the atmosphere is an isobaric process.
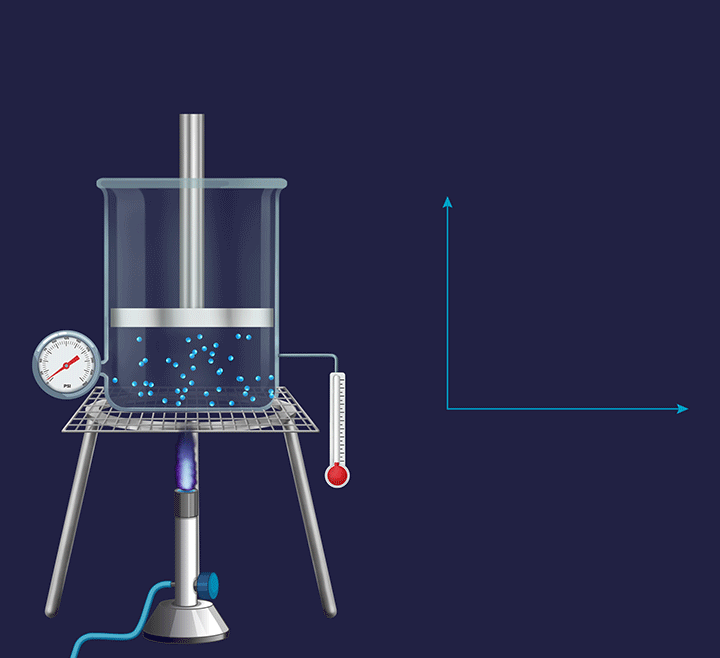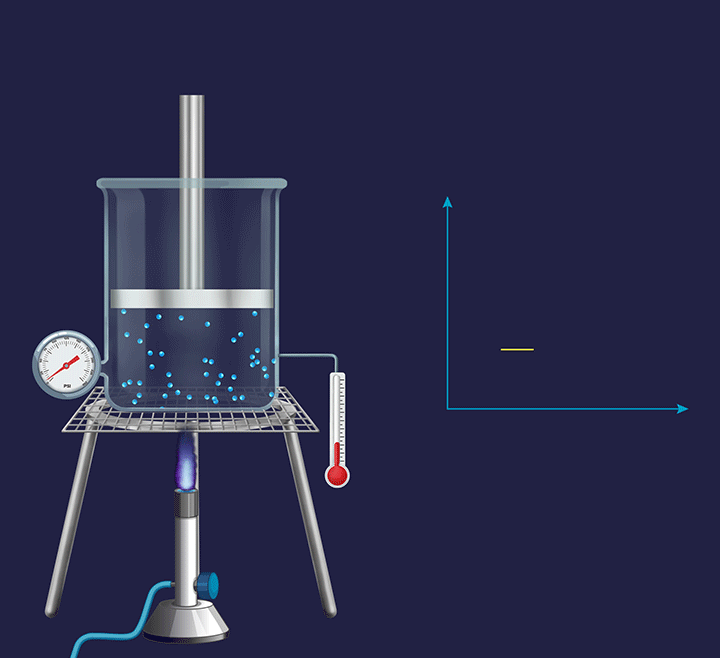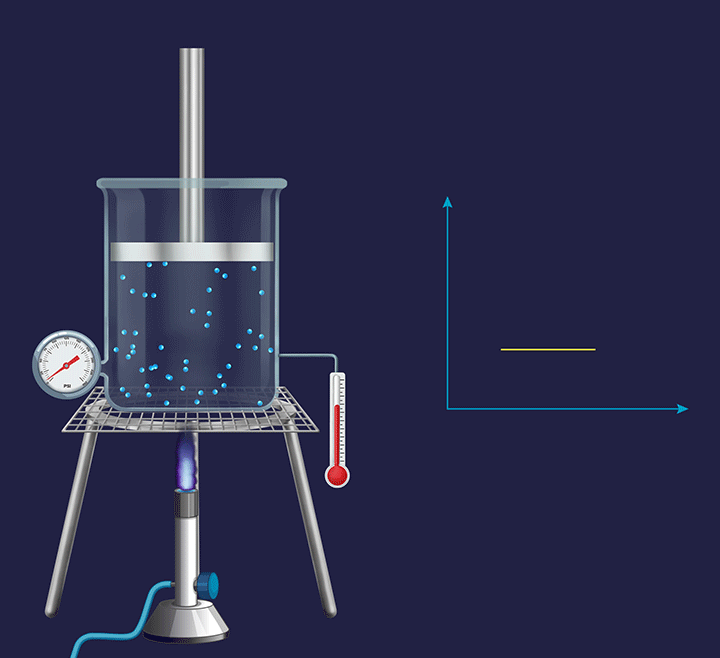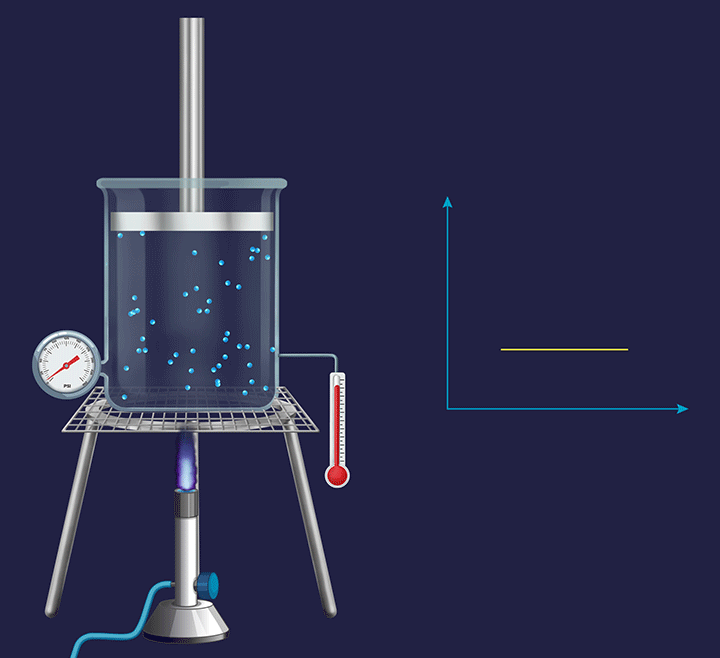

Isochoric process
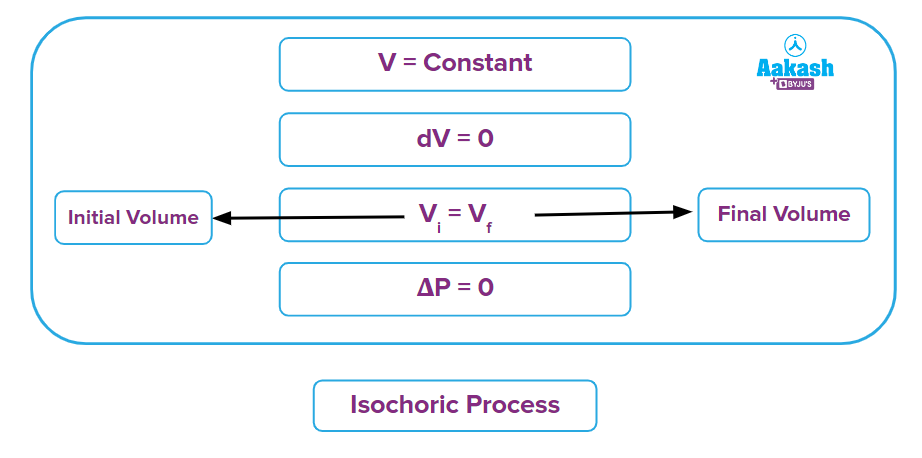
It is a thermodynamic process in which the volume will remain constant throughout the process. Thus, in an isochoric process, dV (for infinitesimally small change) or ΔV (for a finite change) is equal to zero. The thermodynamic process happening inside a closed system with rigid boundaries is an isochoric process.
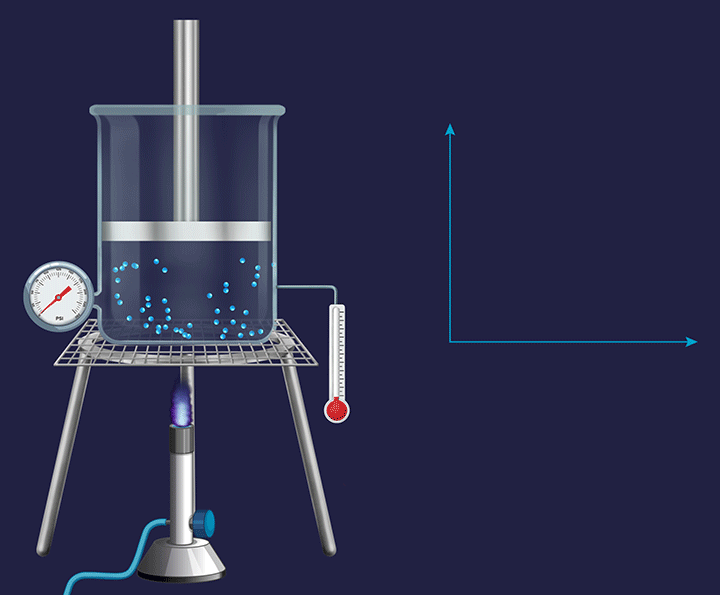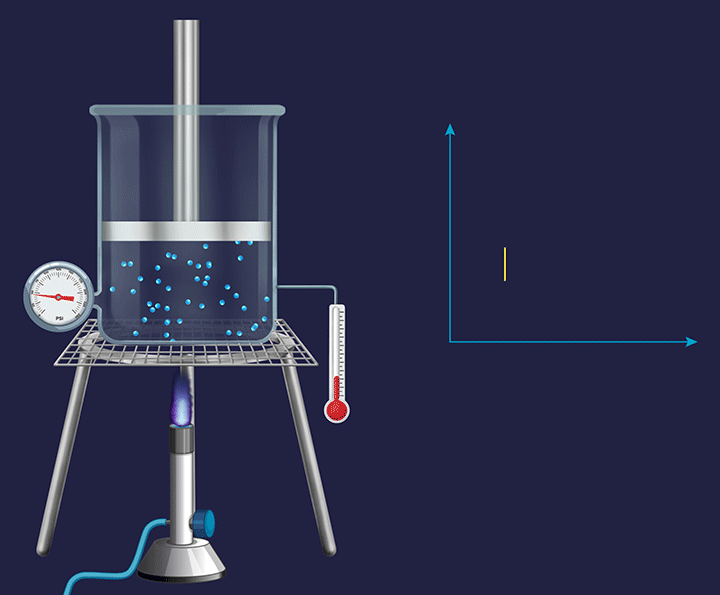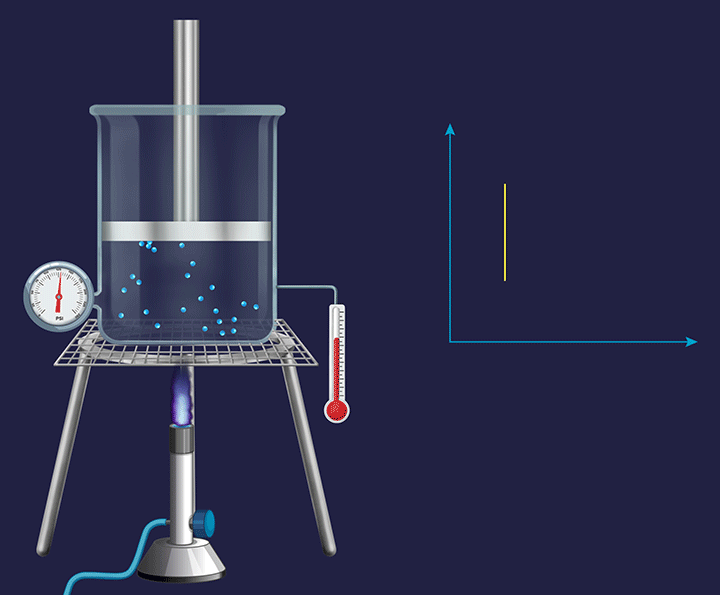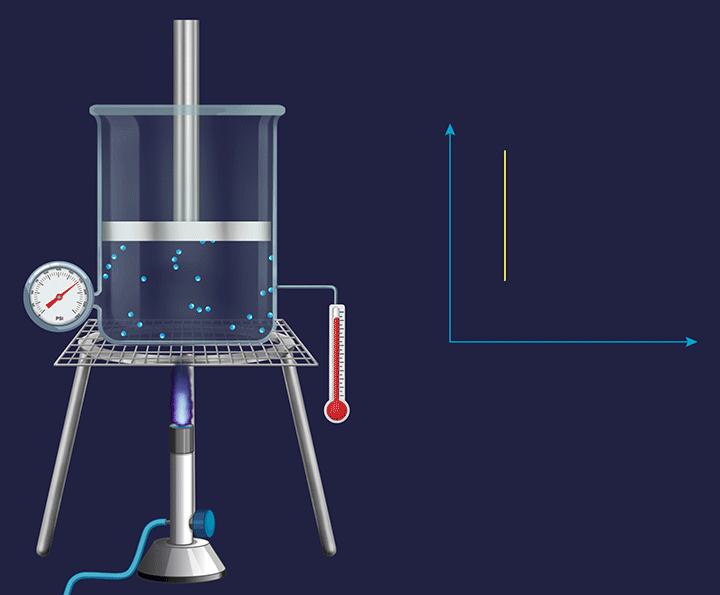

Adiabatic Process
It is a thermodynamic process in which the heat exchange between the system and the surroundings is not possible, i.e., the heat content of the system is constant throughout the process.

The adiabatic process happens in a system with walls similar to that of a thermos flask as there is no exchange of heat energy between the system and the surroundings .

Cyclic Process
A process where the initial state is returned after a set of a sequential thermodynamic processes is called a cyclic process. .
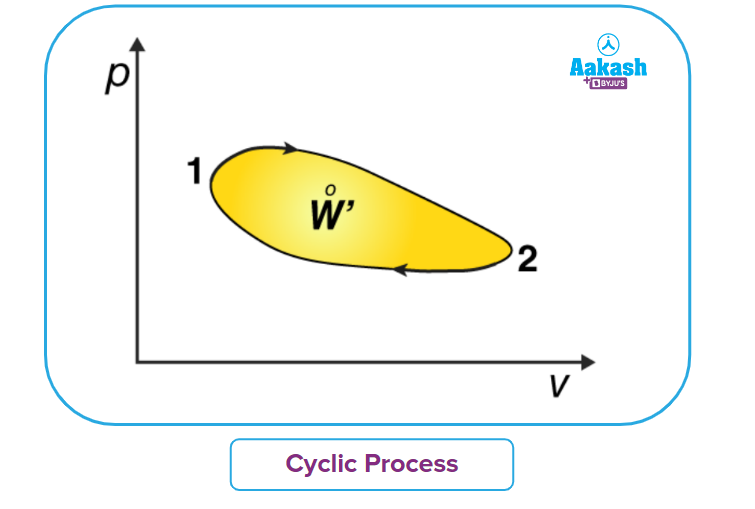
Since the initial and final states are the same in a cyclic process, the internal energy has to be the same indicating the net change in the internal energy of all the processes will be zero.
Practice Problems
Q1. The isothermal process can be represented by which law?
A) Charle’s law
B) Boyle’s law
C) Gay-Lussac’s law
D) 2nd law of thermodynamics
Answer: Option B
Explanation: Boyle’s law states, that PV = a constant. Charle’s law says = constant. As per Gay-Lussac’s law = constant. 2nd law of thermodynamics states that the total entropy of the universe is increasing.
In the isothermal process, the temperature is constant, but P and V can relatively vary as given by Boyle’s Law.
Q2. The thermodynamic process in which temperature remains constant is known as:
- Isothermal Process
- Isochoric Process
- Isobaric Process
- Adiabatic Process
Answer: Option A
Solution: In an isothermal process, the temperature is constant.
Q3. In the isochoric process the change in volume of the thermodynamic system is
- Zero
- Maximum
- Minimum
- None of the above
Answer: Option A
Solution: In thermodynamics, an isochoric process, is a constant volume process, so change in volume during the process will be zero.
Q4. In which thermodynamic process no heat is exchanged between the system and the surrounding:
- Isothermal Process
- Isochoric Process
- Isobaric Process
- Adiabatic Process
Answer: Option D
Solution: An adiabatic process is carried out in a completely insulated system from the surroundings. So any exchange through the walls is prevented. Any thermodynamic changes taking place in the system hence will be at its own internal energy resulting in the change in the temperature of the system at the end of the process..
Frequently Asked Questions – FAQ
Q 1. Is the adiabatic process slow or fast?
Answer: In thermodynamics, an adiabatic process is one in which there is no exchange of heat between the system and its surroundings. One way to accomplish this is to have perfect insulation between the surroundings and the system. In such cases, it doesn’t matter how rapidly the reaction takes place because no matter what the heat exchange will be zero. Another way to perform the thermodynamic change quite rapidly is such that there is no time for the system and surroundings to be in equilibrium to exchange energy as in the expansion of air from a tyre burst.
Question 2. Are all isolated systems adiabatic?
Answer: For the Isolated systems there is no exchange of mass and energy with surroundings. The adiabatic process is the one in which heat transferred to the surrounding is zero. Heat is a form of energy. So all the isolated systems are adiabatic.
Question 3. What is a quasi-static process and why quasi-static processes are slow processes?
Answer: A quasi-static process is a process which occurs infinitesimally slow such that at every stage of the process thermodynamic equilibrium is maintained. A reversible process is said to be a quasi-static process, where the change is likely to be occurring at a highly slow rate. The rate of change is so slow that it seems to be in equilibrium at all times.
Question 4. Is the isothermal process fast?
Answer: In the isothermal process, the system is supplied externally. The heat can enter the system only through its boundary and hence will be limited to a slow process.


