-
Call Now
1800-102-2727
Thermodynamic Terms - System, Surrounding, Universe, Boundary, Macroscopic Properties of the System, State of the System, Practice Problems and FAQ
The word thermodynamics was proposed by William Thomson in 1749. The term thermodynamics is made up of two words: thermo, which means heat, and dynamics, which means movement.

Thermodynamics gives us an understanding of heat flow.
The heat transfer from a cup of coffee to the atmosphere is an example of heat transfer happening in a physical process.

The heat transfer during the combustion of log woods is an example of heat transfer in chemical processes.
TABLE OF CONTENTS
- System
- Surroundings
- Universe
- Types of Systems
- Boundary
- Macroscopic properties of the system
- State of the system
- Practice Problems
- Frequently Asked Questions - FAQ
System
A system in thermodynamics refers to that part of the universe in which thermodynamic observations are made.
Surroundings:
Everything else in the universe other than the system will constitute surroundings.
Universe: The universe is made up of the system and its surroundings.

Types of Systems
- Open System: In an open system, there is an exchange of energy and matter between the system and the surroundings.

- Closed System: In a closed system, there is no exchange of matter. However, an exchange of energy is possible between the system and the surroundings.

- Isolated System: In an isolated system, there is no exchange of energy and matter
between the system and the surrounding. Thermos flask is an example of an isolated system where the exchange of matter and energy will not take place.

Systems can also be classified as follows:
- Ideal system: The term ideal system refers to a hypothetical mechanical system in which energy and power are not lost or dissipated through friction, deformation, wear, or other inefficiencies. They exist at high temperatures and low pressures. This does not exist in reality.
- Real system: Every system observed in real life is an example of a real system. They exist at high pressure and low temperature. Generally, all open systems where the transfer of energy and matter both takes place are considered real systems.
Boundary
A system is separated from the surroundings by the means of a real or imaginary wall. This wall, which separates the system from the surroundings, is known as a boundary.
- Real boundaries and Imaginary Boundaries
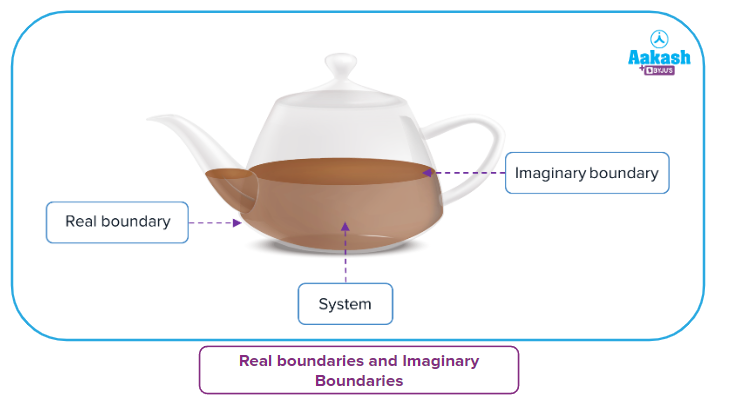
Water in the vessel is defined as a system and everything else as surroundings. Here, the wall of the container is separating the system and surroundings which exists in reality and hence is known as a real boundary.
When a small portion of water (within the entire volume of water inside the container marked in white colour) is defined as a system, the boundaries of this system are imaginary as it does not exist in reality.
- Flexible boundaries and Rigid boundaries
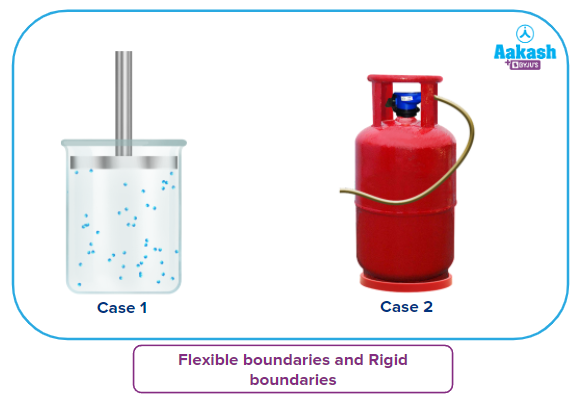
Flexible boundaries will allow pressure-volume work to be done on or by the system, whereas rigid boundaries are inflexible, hence they will prevent any pressure-volume work to be done on or by the system.
- Adiabatic boundaries and diathermal boundaries

A boundary across which the flow of heat will not happen is known as an adiabatic boundary. The wall of the thermos flask is an example of an adiabatic boundary. A boundary which will allow the exchange of heat between the system and the surroundings is known as a diathermal boundary..
Macroscopic Properties of the System
Macroscopic properties of matter are the properties in bulk matter. These properties arise according to how the constituents of matter are arranged in the matter and how the particles are held together. These are properties that can be visualized by the naked eye; thus, we can take measurements easily.
Some common examples of macroscopic properties include pressure, volume, temperature, etc.
- Intensive Properties: Properties which are independent of the amount of the substance (i.e., quantity or size of matter) present in the system are referred to as intensive properties.
- Temperature, pressure, density, melting and boiling point, etc., are intensive properties as they do not change with the change in the quantity of matter.
- Extensive properties: depend on the amount of the substance (i.e., quantity or size of matter) present in the system.
- Volume, mass, and energy are examples of extensive properties.
- If the total value of the property of a system is equal to the sum of the values for the parts of the system (i.e., additive in nature), then such property is known as extensive property.
- Relation between extensive and intensive properties : The ratio of two extensive properties is an intensive property. Density is given as mass per unit volume. Mass and volume are extensive properties, but the ratio of mass and volume (i.e., density) is an intensive property.
State of the System
When the macroscopic properties of a system have definite values, the system is said to be in a definite state. Whenever there is a change in any one of the macroscopic properties, the system is said to be changed into a different state. As a result, the system's state is determined by its macroscopic properties.
- State functions: These are the physical quantities whose values depend only upon the state of the system and do not depend upon the path by which this state has been attained. A few examples of state functions are pressure, volume, temperature, etc.
- Path functions: These are the physical quantities that are dependent on the path/way by which the system has achieved a particular state. Heat and work are examples of path functions.
An illustration of work as a path function is given in the figure below. Here, the work done by the athletes while running around the track will depend on the path followed by them, i.e., if the athlete chooses to run through another path (i.e., the trajectory of the athlete changes), then the work done by the athlete during running will also change.

An illustration of state function is given in the figure below. There is a change in volume as a consequence of the expansion and compression of the syringe. This change in volume, ΔV, will depend only on the initial and final volumes and not upon how we press the syringe.

FIG: Compression and Expansion of Syringe (Illustration for state function)
Practice Problems
Q1. A system wherein the exchange of energy and matter takes place with the environment is called:
A. Open System
B. Closed System
C. Isolated System
D. None of these
Answer: Option A
Solution: In thermodynamics, a closed system can exchange energy (as heat or work) but not matter, with its surroundings. An isolated system cannot exchange any heat, work, or matter with the surroundings, while an open system can exchange both i.e. energy and matter.
Q2. The system where only energy can be exchanged with the surrounding and not the matter is called:
A. Open System
B. Closed System
C. Isolated System
D. None of these
Answer: Option B
Solution: In thermodynamics, a closed system can exchange energy (as heat or work) but not matter, with its surroundings. An isolated system cannot exchange any heat, work, or matter with the surroundings, while an open system can exchange both i.e. energy and matter.
Q3. The volume of a system is a/an:
A. Intensive property
B. Extensive property
C. Neither Extensive nor Intensive
D. None of these
Answer: Option B
Solution: An extensive property is a property that depends on the amount of matter in a sample. The volume of an object is a measure of the space that is occupied by that object. Hence, Volume is an extensive property.
Q4. Which of the following is an intensive property?
A. Enthalpy
B. Entropy
C. Density
D. Mass
Answer: Option C
Solution: Mass and volume are extensive properties as they depend on the amount of matter. The ratio of two extensive properties of the same object or system is an intensive property.
For example, the ratio of an object's mass and volume, which are two extensive properties, is density, which is an intensive property.
Q5. Which of the following is an intensive property?
A. Temperature
B. Viscosity
C. Surface Tension
D. All of these
Answer: Option D
Solution: A property which depends only upon the nature of the substance and not on the amount(s) of the substance(s) present in the system is known as an intensive property.
Some common examples are temperature, pressure, concentration, refractive index, viscosity, density, surface tension, specific heat, etc.
The quotient obtained by dividing any extensive variable by another extensive variable gives an intensive variable. For example, concentration (moles/liter), density (mass/volume), mole fraction (n/N), specific heat(heat capacity per unit mass).
Frequently Asked Questions – FAQ
Question 1. What is the difference between intensive property and extensive property?
Answer: An extensive property is a property that depends on the amount of matter in a sample. Mass and volume are examples of extensive properties. An intensive property is a property of matter that depends only on the type of matter in a sample and not on the amount. Density and temperature are examples of intensive properties.
Question 2. Why is molar volume an intensive property?
Answer: The property which depends upon the quantity of matter present in the system is known as extensive property. The volume of a system is extensive. But the volume of one mole of gas or matter in the system is fixed, and independent of the amount of matter present in the system.
Thus, molar volume is an intensive property.
Question 3. What are Diathermal walls, diabatic Walls, and adiabatic Walls?
Answer: A diabatic wall (also referred to as a diathermal wall) is a wall that allows heat to pass through it. Therefore, it can be understood that diabatic walls are made up of thermal conductors and are essentially thermal walls. an Adiabatic wall is a wall that does not allow heat to pass through it. Therefore, it can be understood at adiabatic walls are made up of thermal insulators and that they are ideal for maintaining low or high temperatures while storing certain objects.
Question 4. What is an Intrinsic property? Is it the same as intensive property?
Answer: Intrinsic - It refers to the property of a material which is due to the matter contained within the material itself. In short, the natural state or configuration of the material.
Intensive - Properties which are independent of the mass of material or amount of substance are called intensive properties.
Example: Temperature, density, etc.
Related Topics:
| Isothermal Process | First Law of Thermodynamics |
| Thermodynamic Processes | Degrees of Freedom |
| Heat Capacity Cp Cv relation |
Zeroth Law of Thermodynamics
|
|
Second Law of Thermodynamics
|
Third Law of Thermodynamics
|



