-
Call Now
1800-102-2727
Sulphonation of Benzene and its Derivatives - Sulphonation of Substituted Benzene, Their Mechanisms, Practice Problems & FAQs
Let's say that Tom and Jerry are two brothers. Their father wants them to be engineers, but their mother wants them to be doctors.
They both had confusion, so they sought career counseling for the future to help them with their issues.
According to the counselor, Tom had a strong passion for cooking and needed to seek a career in hospitality, whereas Jerry had a strong passion for travel and needed to pursue a career in tour and travel.

Chemical reactions are similar in that they don't always go as expected. They react to obtaining the products based on their own interests and behaviors. Derived or substituted benzene based on their nature and behavior, reacts with fuming sulphuric acid.
Table of Contents
- Sulfonation of Benzene
- Electrophilic aromatic substitution reaction of Substituted benzene
- Practice Problems
- Frequently Asked Questions
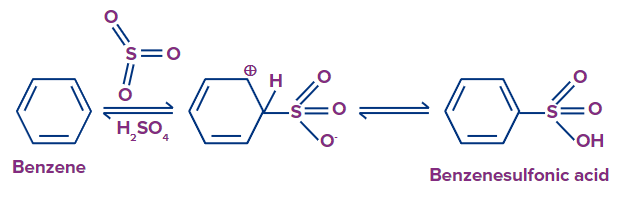
Sulfonation of Benzene:
SO3 act as an electrophile in this electrophilic aromatic substitution reaction. In this reaction, fuming sulfuric acid (H2SO4+SO3) is used to heat benzene to produce benzene sulfonic acids. Reactions may be reversed in this process.
Electrophilic aromatic substitution reaction of Substituted benzene:
(i) Substituted benzene with electron-releasing group:
When benzene is substituted with electron-releasing groups or ortho para-directing groups, the sulfonic group will get attached to the ortho- and para- position.
These electron-releasing groups are -OH, -NH2, -OCH3,-Cl, -Br,-I etc.
For example: When phenol is heated in the presence of fuming sulphuric acid (H2SO4+SO3), there is the formation of both 4-hydroxy benzene sulfonic acid (para-hydroxy benzene sulfonic acid) and 2-hydroxy benzene sulfonic acid (ortho-hydroxy benzene sulfonic acid) as the hydroxyl group is ortho-para directing group. But 4-hydroxy benzene sulfonic acid is formed as a major product because the SO3H group is bulky and creates less steric hindrance at the para position than at the ortho position.

Mechanism of Sulphonation of Substituted benzene with the electron-releasing group:
Step - 1: The production of electrophiles from fuming sulfuric acid is the first step in this reaction. Sulfur trioxide is created by the loss of a water molecule from sulfuric acid.
Step - 2: In the second step, the ortho and para positions of phenol are more electron-rich as compared to other positions as seen in resonating structures.
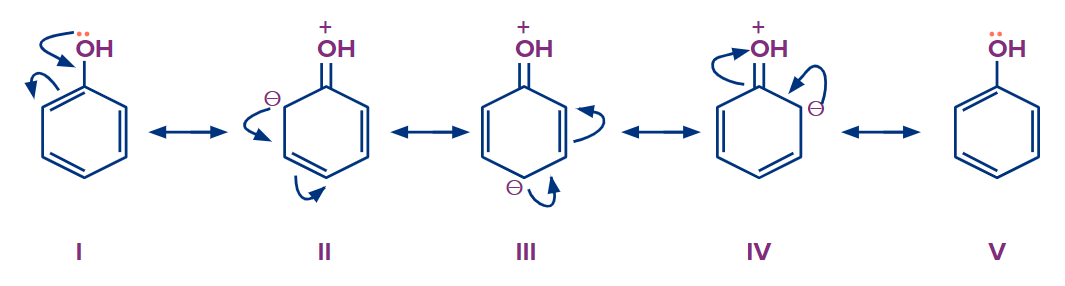
The electrophile created in the first step will attack on electrons present at the ortho-para position. A sigma complex -complex is formed when an electrophile abstracts the electron from the phenol. This step is the slow step and the rate-determining step of this reaction.
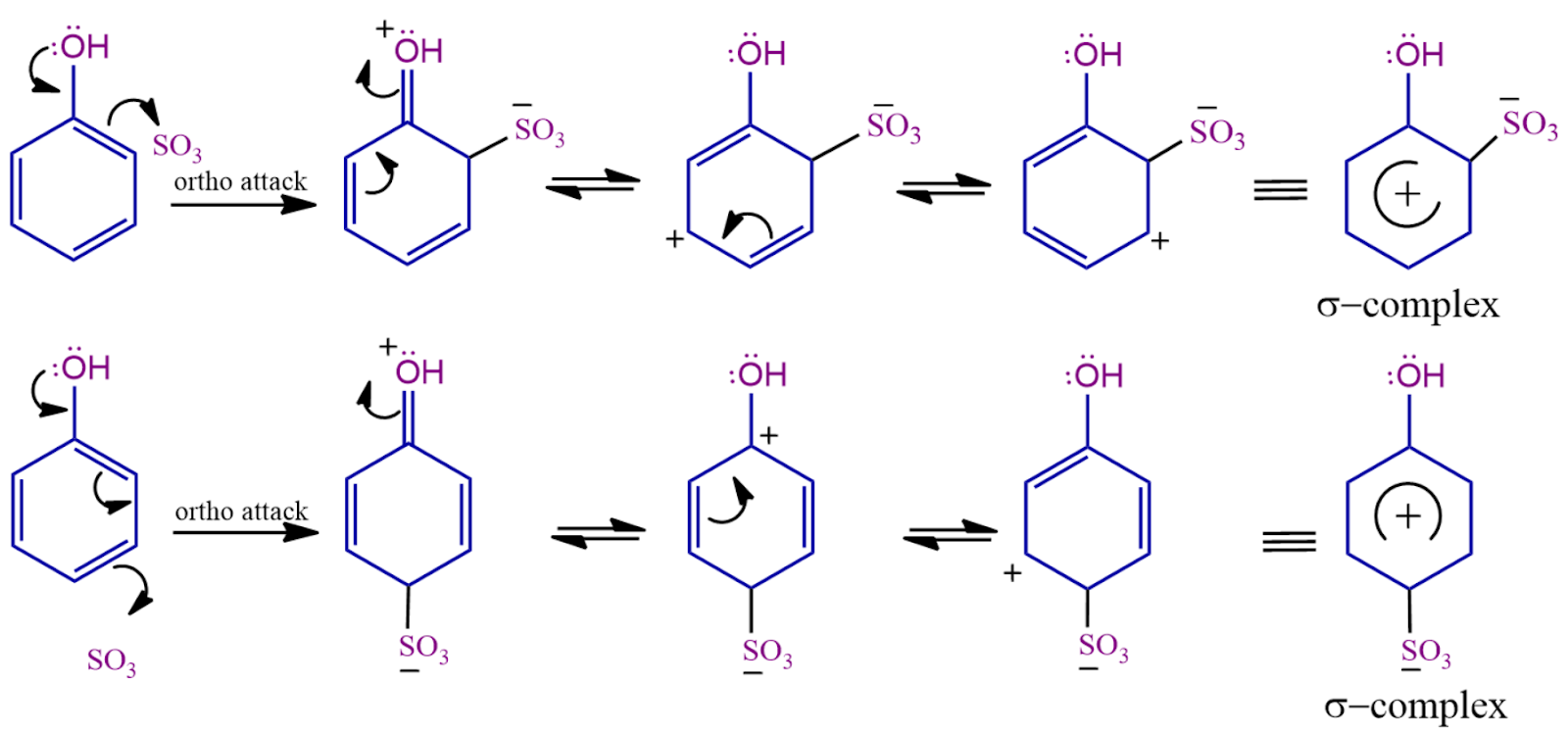
Step - 3: 2-hydroxybenzene sulfonate is produced when the HSO4- removes the hydrogen bonded to a carbon that has a sulfonic group.
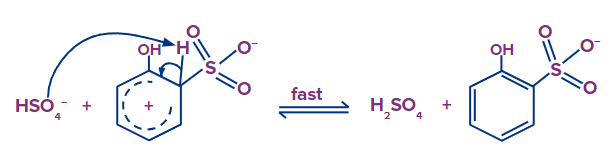
Step - 4: In the fourth step, 2-hydroxybenzenesulfonate abstracts hydrogen from the H2SO4, resulting in the formation of 2-hydroxybenzenesulfonic acid in the final step.
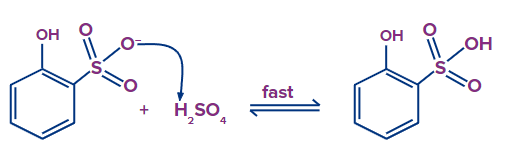
(ii) Substituted benzene with electron-withdrawing group:
When benzene is substituted with electron-withdrawing groups or meta-directing groups, the sulfonic group will get attached to the meta-position.
These electron-releasing groups are -CHO, -NO2, -COOH,-COCH3 etc.
For example: When nitrobenzene is heated in the presence of H2SO4/SO3, there is a formation of 3-chloro benzene sulfonic acid (m-chloro benzene sulfonic acid) as the nitro group is a meta directing group.
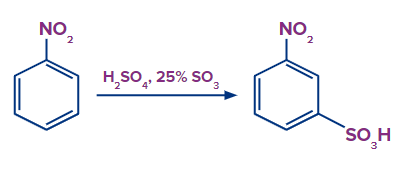
Mechanism of Sulphonation of Substituted benzene with the electron-withdrawing group:
Step - 1: The production of electrophiles from fuming sulfuric acid is the first step in this reaction. Sulfur trioxide is created by the loss of a water molecule from sulfuric acid.
2H2SO4H3+O+HSO4-+SO3
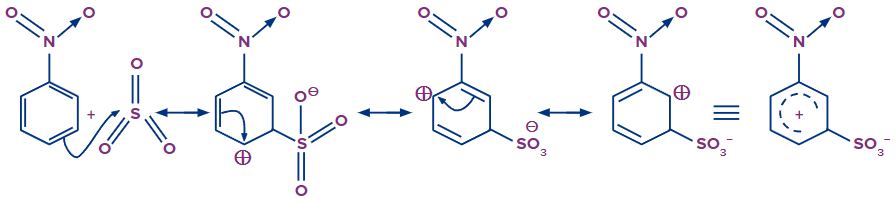
Step - 2: In the second step, the meta positions of nitro are more electron-rich as compared to other positions, as seen in resonating structures. The ortho and para positions have a positive charge, which shows the dis availability of electrons.
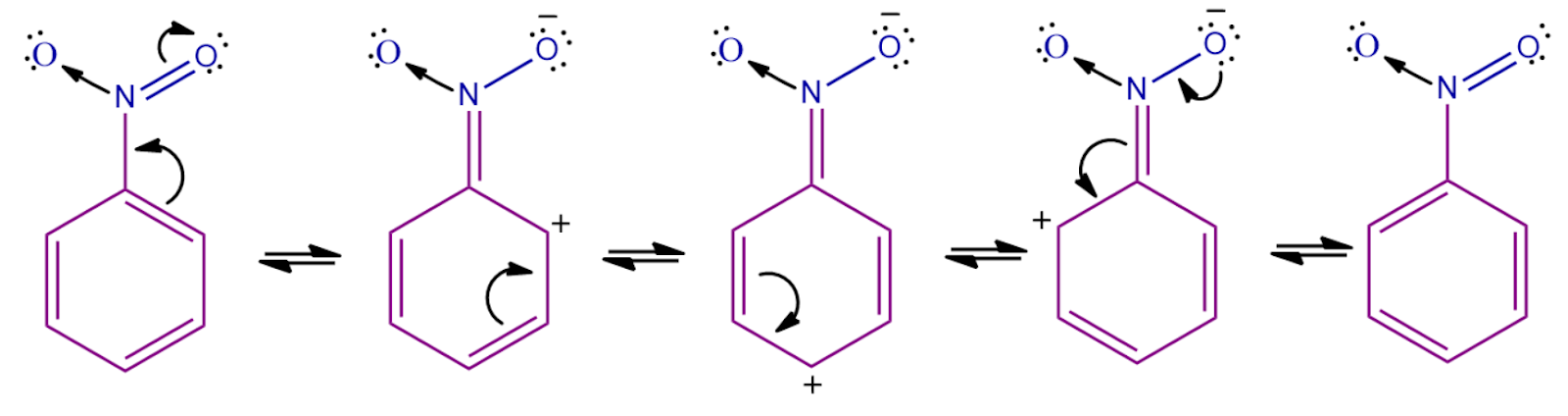
The electrophile created in the first step will attack on electrons present at the meta position. A sigma complex -complex is formed when an electrophile abstracts the electron from the nitrobenzene. This step is the slow step and the rate-determining step of this reaction.
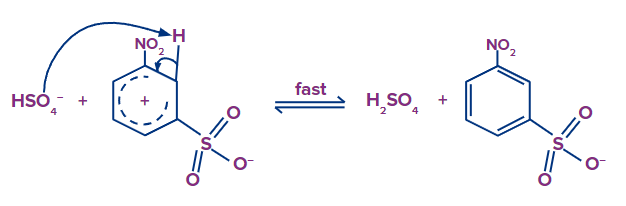
Step - 3: 3-nitrobenzenesulfonate is produced when the HSO4- removes the hydrogen bonded to a carbon that has a sulfonic group.
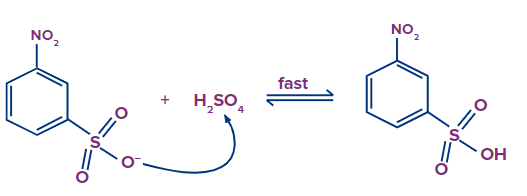
Step - 4: In the fourth step, 3-nitrobenzenesulfonate abstracts hydrogen from the H2SO4, resulting in the formation of 3-nitrobenzenesulfonic acid in the final step.
Practice Problems:
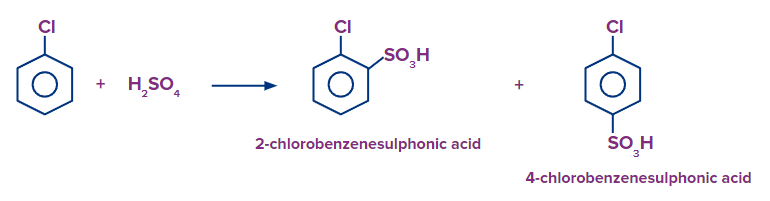
Q1. What products should be formed when chlorobenzene is reacted with fuming H2SO4/SO3?
- p-chloro benzene sulfonic acid
- o-chloro benzene sulfonic acid
- Both (A) and (B)
- m-chloro benzene sulfonic acid
Answer: (C)
Solution: When chlorobenzene is heated in the presence of fuming sulphuric acid (H2SO4+SO3), there is a formation of both 4-chloro benzene sulfonic acid (p-chloro benzene sulfonic acid) and 2-chloro benzene sulfonic acid (o-chloro benzene sulfonic acid) as the chloro group is ortho-para directing group. But 4-chloro benzene sulfonic acid is formed as a major product because the SO3H group is bulky and creates less steric hindrance at the para position than at the ortho position.
Hence, the correct answer is option (C).
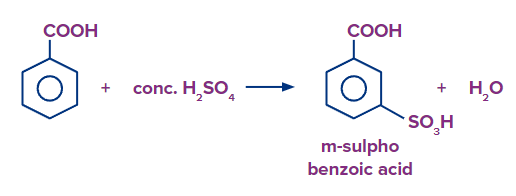
Q2. What products should be formed when benzoic acid is reacted with fuming H2SO4/SO3?
- 2-sulfobenzoic acid
- 3-sulfobenzoic acid
- 4-sulfobenzoic acid
- Both (A) and (C)
Answer: (B)
Solution: When benzoic acid is heated in the presence of fuming sulphuric acid (H2SO4+SO3), there is a formation of 3-sulfobenzoic acid as the carboxylic group is a meta directing group. Hence, the correct answer is option (B).
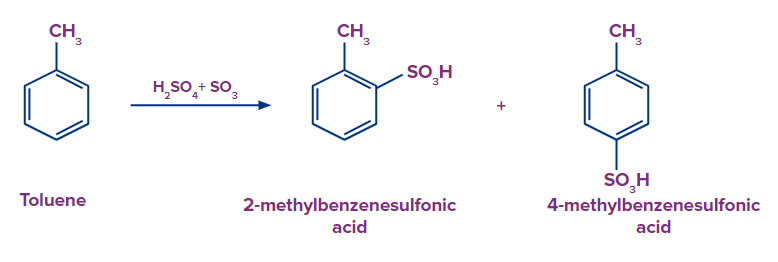
Q3. What should be the expected product formed in major when methylbenzene is treated with fuming sulphuric acid and benzenesulfonic acid is treated with CH3Cl/FeCl3 respectively ?
- 4-methylbenzenesulfonic acid and 4-methylbenzenesulfonic acid
- 2-methylbenzenesulfonic acid and 2-methylbenzenesulfonic acid
- 2-methylbenzenesulfonic acid and 3-methylbenzenesulfonic acid
- 4-methylbenzenesulfonic acid and 3-methylbenzenesulfonic acid
Answer: (D)
Solution: It is an electrophilic aromatic substitution reaction in which SO3 act as an electrophile. When methylbenzene is heated in the presence of fuming sulphuric acid (H2SO4+SO3), there is a formation of both 2-methyl benzene sulfonic acid and 4-methyl benzene sulfonic acid as the methyl group is an electron releasing group. But 4-methyl benzene sulfonic acid is formed as a major product because the SO3H group is bulky and creates less steric hindrance at the para position than at the ortho position.
When benzene sulfonic acid is heated in the presence of CH3Cl/FeCl3, there is the formation of 3-methylbenzenesulfonic acid as the sulfonic group is a meta directing group.
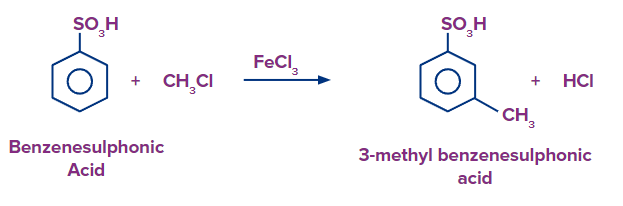
Hence, the products formed are 4-chloro benzene sulfonic acid and 3-chloro benzene sulfonic acid, respectively and the correct answer is option (D).
Q4. In the slowest step of electrophilic substitution of benzene with fuming sulphuric acid (H2SO4+SO3), there is a formation of:
- Electrophile
- -complex
- -complex
- All of these
Answer: (B)
Solution: The removal of the electron from the benzene by electrophile results in the formation of a sigma -complex. This is the slowest step in the reaction.
Hence, the correct answer is option (B).
Frequently Asked Questions-FAQs:
1. What exactly are nucleophiles?
Answer: A nucleophile is an attacking reagent that has a love or attraction for the nucleus or positive centre. It has at least one pair of readily donatable electrons and is hence either negatively charged or neutral.
2. What exactly are electrophiles?
Answer: An electrophile is an undesirable substance that loves or is drawn to electrons. It is hence devoid of electrons and either positively charged or neutral.
3. What is an electrophilic aromatic substitution reaction?
Answer: An atom that is connected to an aromatic ring is replaced with an electrophile in electrophilic aromatic substitution processes. Examples of these reactions include Friedel-Crafts reactions, aromatic nitrations, and aromatic sulfonation.
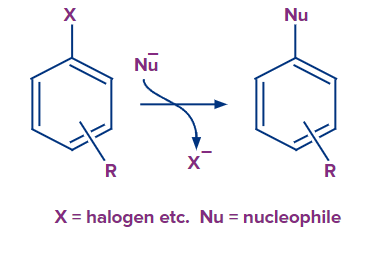
4. What is the reaction of nucleophilic aromatic substitution?
Answer: A good leaving group, like a halide, on an aromatic ring is displaced by the nucleophile in a nucleophilic aromatic substitution, a type of substitution process in organic chemistry. Although nucleophilic substitution does occur in some aromatic compounds, aromatic rings are typically nucleophilic.