-
Call Now
1800-102-2727
Sugars – Types, Tests, Sources, Importance, Side Effects, Practice Problems and FAQ
"Avoid consuming these beverages since they contain too much sugar. Don't consume too many sweets and chocolates!”
Our parents frequently provide such tough instructions, right? However, have you ever questioned why they say that?
There is a wise proverb that says "Too much of anything is not good". The same is true in this instance. Increasing our sugar intake is undoubtedly bad for us and can cause a variety of illnesses.

But does this imply that we need to drastically reduce our sugar intake? Consider that for a moment. Will fully reducing sugar be beneficial? Can we completely eliminate all use of sugar?
Well! No, we should not. We can remove sugar from our diet since, even if we don't consume it directly in raw form, it still exists in some form in the many foods we consume. Sugar has several health benefits for our body, thus completely eliminating it is also not a smart idea. However, we must approach it so that it does not become excessive or inadequate altogether.
Let's discuss sugars in more detail and examine their significance to us.
TABLE OF CONTENTS
- Carbohydrates
- Carbohydrates – Classification
- Sugars – Types
- Sugars – Glucose, Fructose and Sucrose
- Sugars – Tests
- Sugars – Importance
- Sugars – Importance
- Sugars – Side Effects
- Practice Problems
- Frequently Asked Questions – FAQ
Carbohydrates
Sugars are nothing but other names for simple carbohydrates. So, before actually getting into sugars, let’s understand carbohydrates.
Plants are the primary producers of carbohydrates. Carbohydrates comprise sugars, fibres, and starch. Grains, vegetables, and fruits, as well as milk and other dairy products, contain carbohydrates. They are the fundamental dietary groups that are required for a healthy lifestyle.

Carbon (C), hydrogen (H), and oxygen (O) atoms make up carbohydrate molecules. Carbohydrates gained their name from the fact that they typically have the formula Cx(H2O)y. They appeared to be the ‘hydrates of carbon.
When we look at the formula for glucose, which is C6H12O6, we can see that it clearly fits into the general formula for carbohydrates, which is Cx(H2O)y.
But all the compounds that fit into this formula may not be classified as carbohydrates. Carbohydrates are not necessarily all of the chemicals that fit into this formula. For example, though acetic acid (CH3COOH,) is not a carbohydrate, it does fit into the C2(H2O)2 formula. Similarly, rhamnose (C6H12O5) is a carbohydrate but it does not fit in the formula.
As a result, we can conclude that carbohydrates cannot be defined using this formula only.
Carbohydrates can be defined as polyhydroxy aldehydes and ketones, or substances that hydrolyse to produce polyhydroxy aldehydes and ketones. Sugars or saccharides are other names for simple carbohydrates.
Carbohydrates – Classification
Classification of carbohydrates is done based on:
- The product formed on hydrolysis of the carbohydrate
- Their reducing nature (Ability to reduce Tollens and Fehling’s Solution)
- The number of carbon atoms and functional groups
Simple carbohydrates
Simple carbohydrates are those that include only one or two sugar molecules. Because molecules are easily ingested and converted, they cause blood sugar levels to rise. A number of them are found in milk products, alcohol, fruits, refined sweets etc. Monosaccharides and disaccharides are the major components of sugars. Monosaccharides are the most basic carbohydrates that can not be hydrolysed into other sugars. Examples: Glucose, Fructose, etc.
On the other hand, disaccharides are formed when two monosaccharides are linked together. When disaccharides are hydrolysed, they produce two monosaccharide molecules.
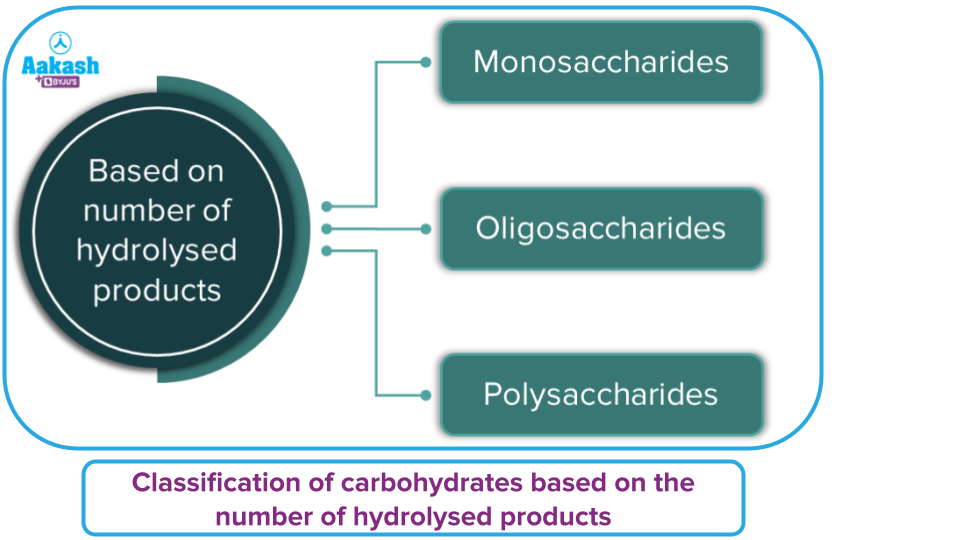
Carbohydrates are classified broadly into three categories on the basis of their behaviour on hydrolysis. Based on the number of hydrolysed products, they are classified as:
- Monosaccharides
- Oligosaccharides
- Polysaccharides
Monosaccharides
Monosaccharides are the most basic carbohydrates that cannot be digested into even more basic carbs.
Examples: Glucose, ribose, fructose etc.

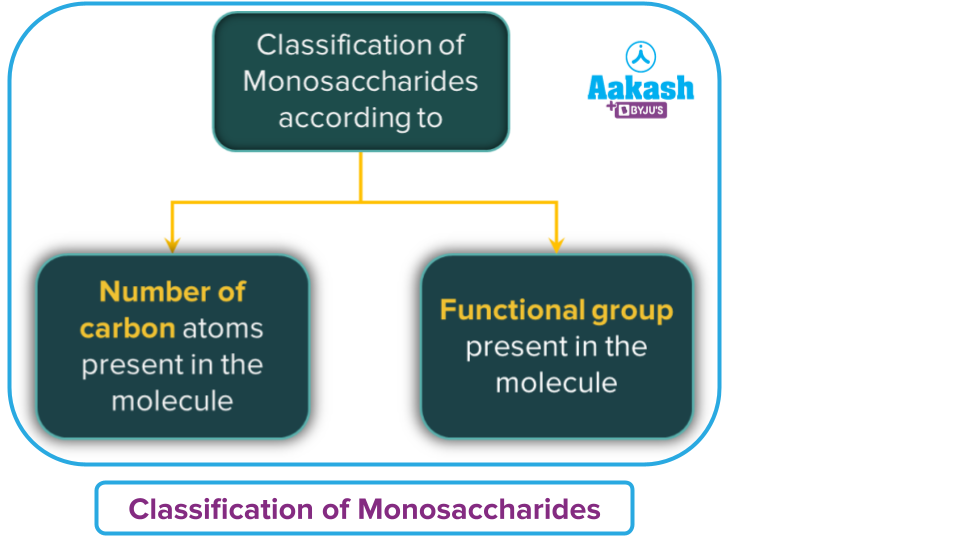
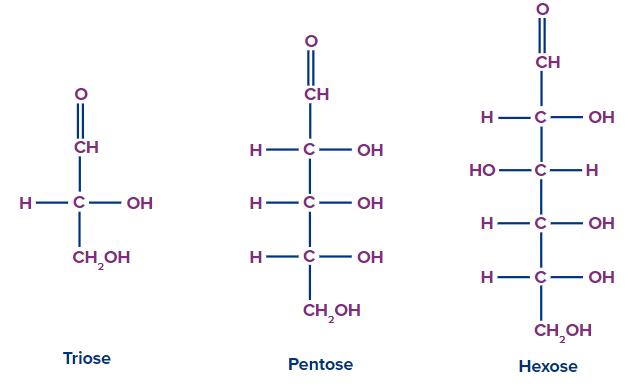
On the basis of the number of carbon atoms and the functional groups present, monosaccharides are further divided into two classes.
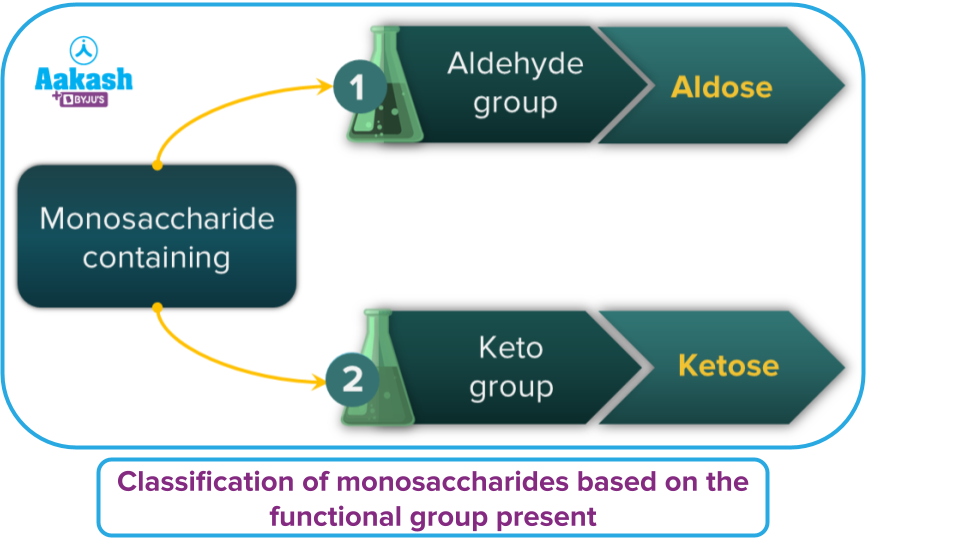
|
Number of carbon atoms |
General term for monosaccharides |
Aldehyde |
Ketone |
|
3 |
Triose |
Aldotrose |
Ketotriose |
|
4 |
Tetrose |
Aldotetrose |
Ketotetrose |
|
5 |
Pentose |
Aldopentose |
Ketopentose |
|
6 |
Hexose |
Aldohexose |
Ketohexose |
|
7 |
Heptose |
Aldoheptose |
Ketoheptose |
Oligosaccharides
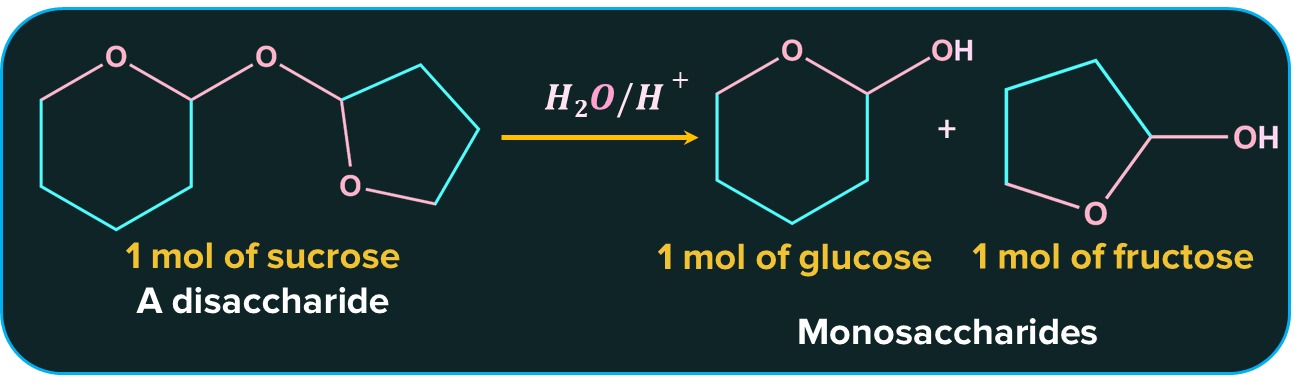
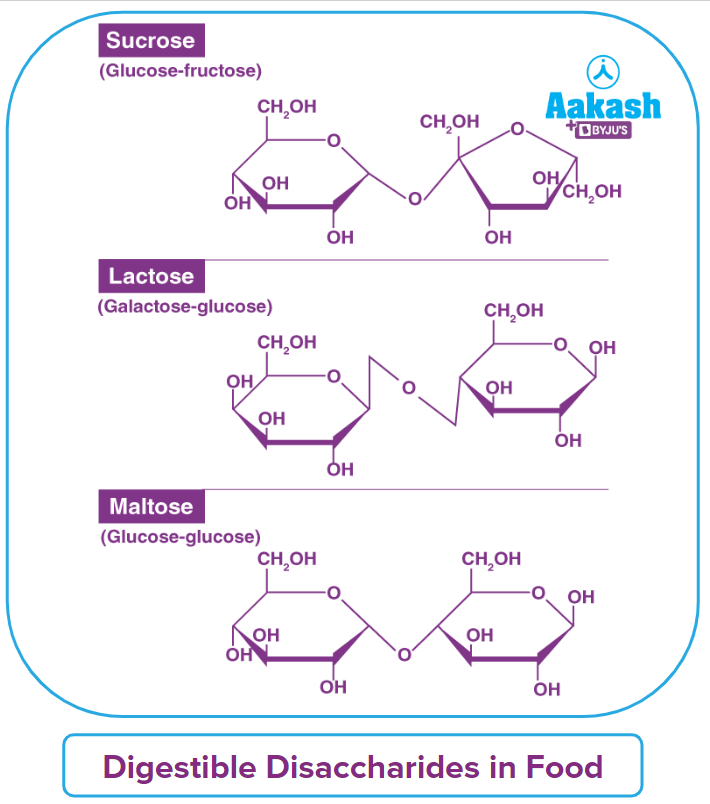
Oligosaccharides, which are molecules of carbohydrates, are produced by the condensation of 2–9 monomers. Oligosaccharides are also known as disaccharides, trisaccharides, and so on, depending on how many monosaccharide units are joined. When they include more than 10 sugar residues, polysaccharides are frequently categorised independently from oligosaccharides. Sucrose, maltose, and lactose are among examples.
One molecule of sucrose, for instance, hydrolyzes into two molecules: one of glucose and one of fructose.
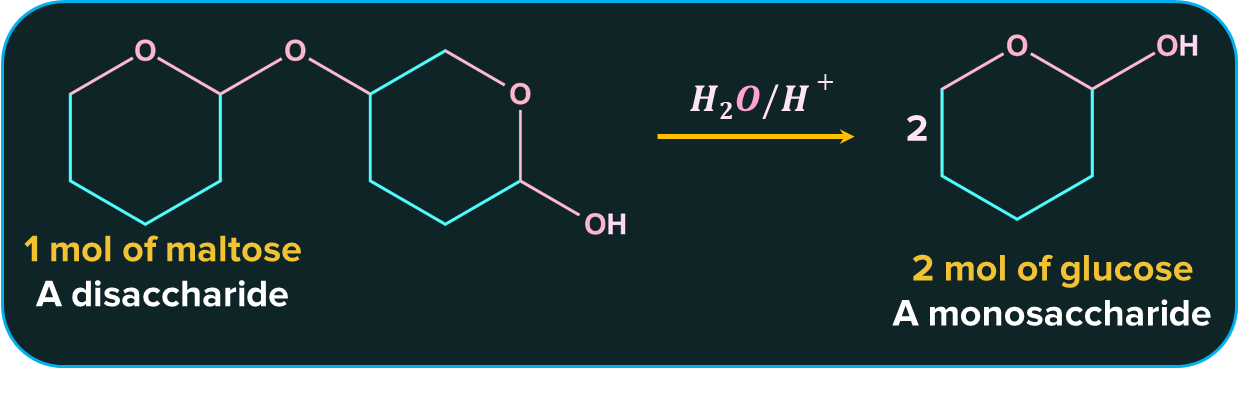
Also, maltose on hydrolysis gives two molecules of only glucose as shown below.
Polysaccharides
They are carbohydrates that are generated from more than ten monosaccharide molecules. They can also be explained as complex carbohydrates composed of a number of monomers that have been polymerized. Starch, glycogen, cellulose, and other highly branched and homopolymeric carbohydrates are examples of polysaccharides (made up of just glucose units).
Two or more sugar units make up complex carbohydrates. Complex carbohydrate molecules undergo slower digestion and transformation than simple carbohydrates' do. They are widely distributed in whole-grain bread, cereals, peanuts, corn, peas, potatoes, beans, lentils, and other foods.
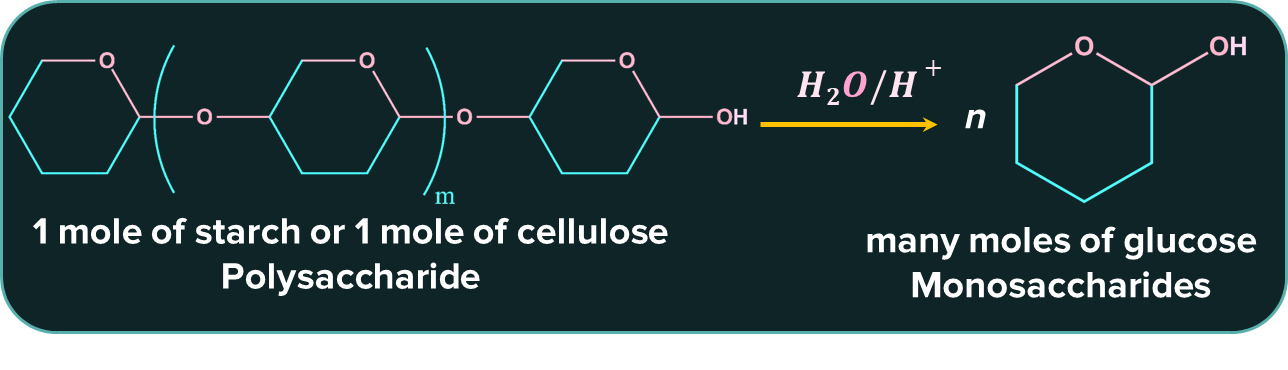
The following are some examples of polysaccharides.
- Starch - The two components that makeup starch are amylose and amylopectin. Amylose creates a straight chain, whereas amylopectin is a severely branched chain.
- Glycogen - Glycogen is also referred to as animal starch. Structured like starch, but with more pronounced branching.
- Cellulose - The primary structural element of the plant cell wall is cellulose, a structural carbohydrate. It has a high tensile strength and is a fibrous polysaccharide. Unlike starch and glycogen, cellulose polymerizes linearly to form a linear structure.
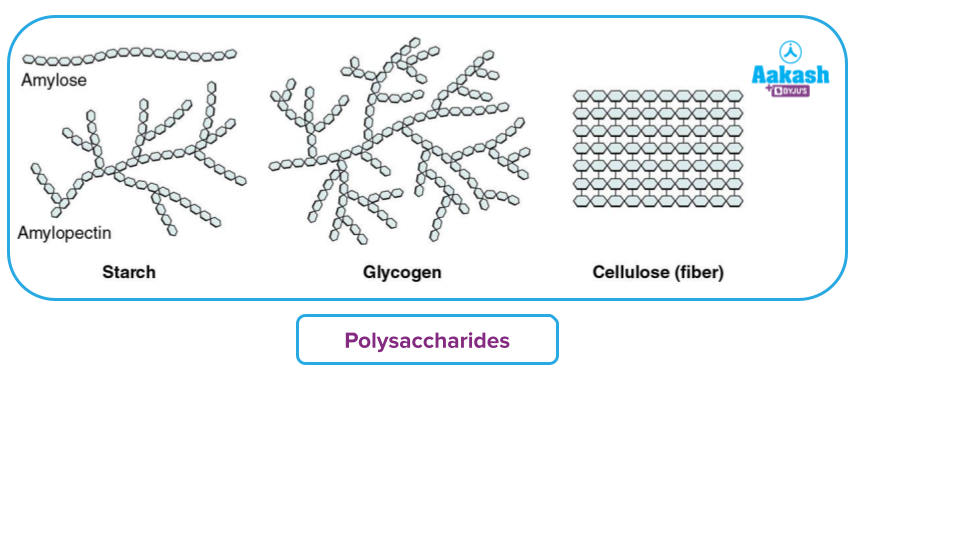
Sugars – Types
Based on their ability to reduce Tollens' & Fehling's reagent, carbohydrates can also be categorised as either reducing sugar or on-reducing sugar based on their reducing nature.
Sugars must have at least one hemiacetal or hemiketal functional group in order to reduce Tollens’ and Fehling's reagents. Non-reducing sugars should have acetal linkage since they do not reduce Tollens’ or Fehling's solution.
All polysaccharides are non-reducing sugars, but all monosaccharides and oligosaccharides, with the exception of sucrose, are reducing sugars.
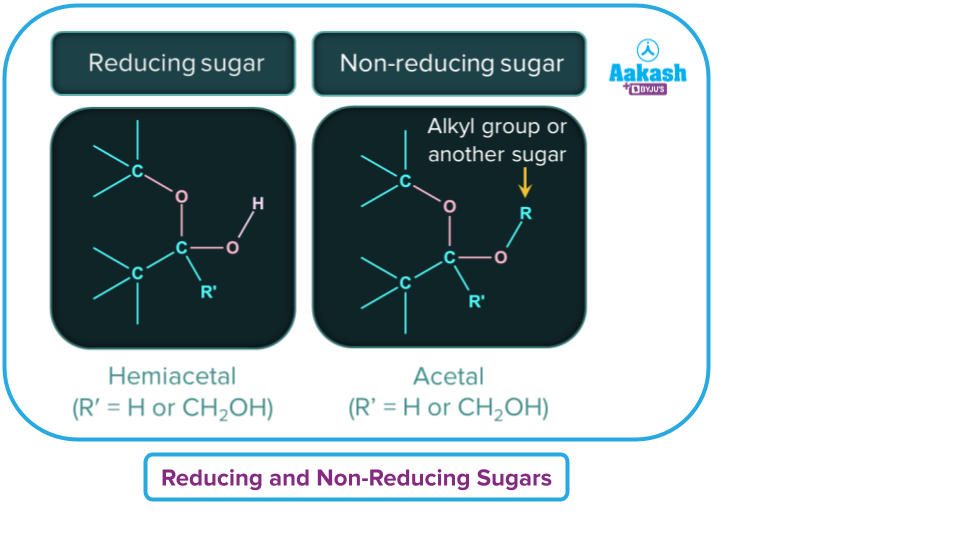
Based on the ability to react with Tollens’ and Fehling’s reagents, we have two different categories of sugars.
- Reducing sugar
- Non-reducing sugars
Reducing Sugars
- Reducing sugars reduces Tollens’ reagent and Fehling’s reagent.
- Reducing sugars should have at least one hemiacetal or hemiketal functional group. The Hemiacetal group gives positive Tollens’ or Fehling’s test.
- All monosaccharides and oligosaccharides except sucrose are reducing sugars.
- Examples of reducing sugars include glucose, fructose, glyceraldehyde, lactose, arabinose, maltose etc.
Non-Reducing Sugars
- Non-reducing sugars do not reduce Tollens’ and Fehling’s reagents.
- Non-reducing sugars have acetal linkages and the acetal group does not give positive Tollens’ or Fehling’s test.
- All polysaccharides and sucrose are non-reducing sugars.
- Examples of non-reducing sugars include sucrose, trehalose, raffinose, stachyose, verbascose etc.
Sugars – Glucose, Fructose and Sucrose
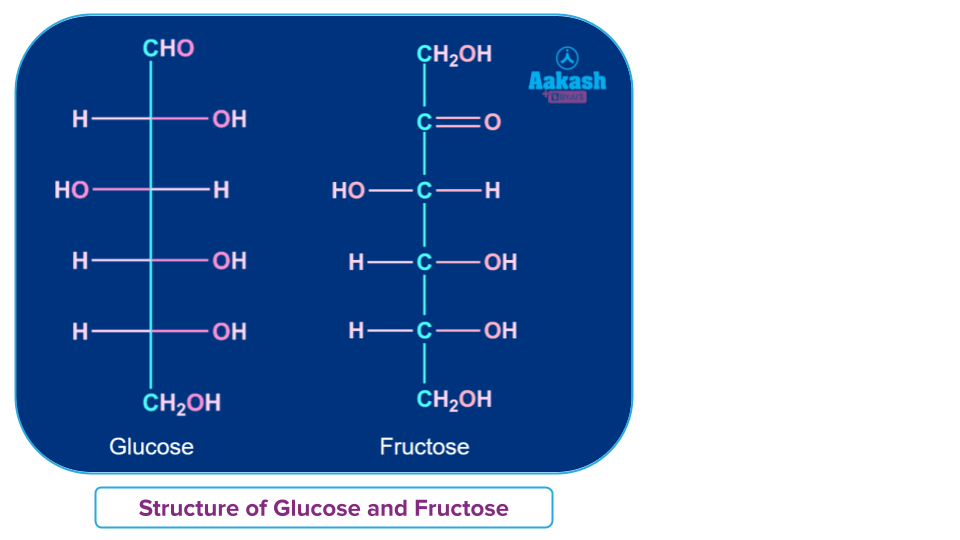
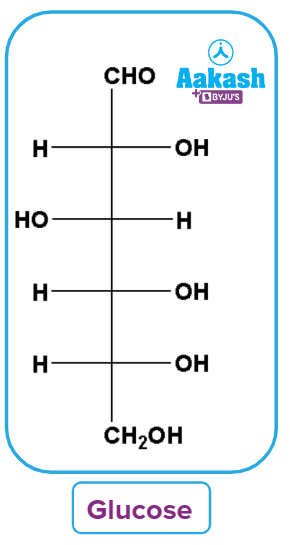
Glucose
Glucose is a carbohydrate that plays an important role in the body's metabolism. It is a basic sugar with a chemical formula C6H12O6. In simple terms, it is made up of six carbon atoms, twelve hydrogen atoms, and six oxygen atoms. It acts as a reducing agent (reduces both Fehling’s solution and Tollens’ reagent).
Glucose, often known as dextrose or blood sugar, is a simple monosaccharide that is readily available. Plants and the vast majority of algae create glucose during the photosynthesis process.
Glucose is a form of sugar, to put it another way. When we mention sugar, we do not just mean the white crystals we add to edible sweets. Carbohydrates are energy-giving substances present in our food, and sugars are a subclass of carbohydrates.
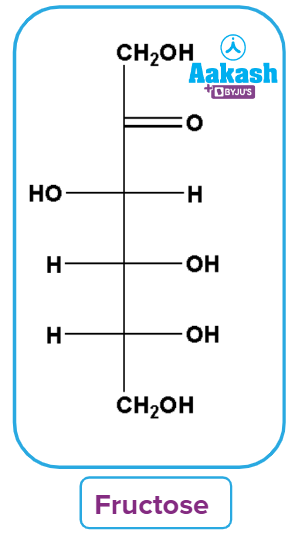
Fructose
Fructose is an important ketohexose. The hydrolysis of the disaccharide sucrose yields it along with glucose. It is a basic sugar with a chemical formula C6H12O6. It is a monosaccharide that can be found in fruits, honey, and vegetables. It is used as a sweetener in its purest form. It is a levorotatory compound that belongs to the D-series.
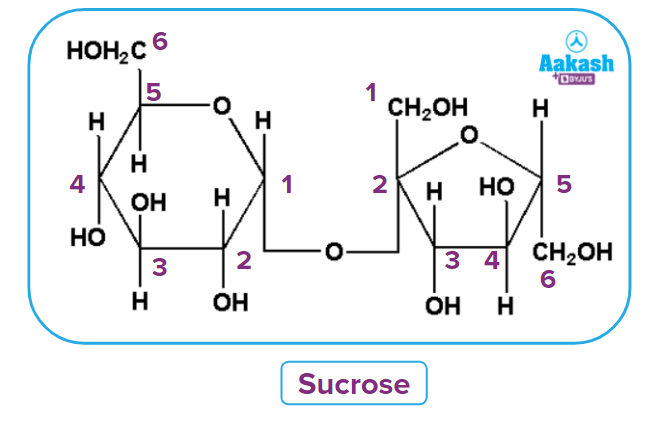
Sucrose
Sucrose is a white crystalline solid, which is soluble in water. Its molecular formula is C12H22O11. Sucrose is dextrorotatory, although it converts to dextrorotatory glucose and laevorotatory fructose following hydrolysis. Since the laevorotation of fructose (–92.4°) is more than the dextrorotation of glucose (+52.5°), hence, the mixture is laevorotatory. This is known as the inversion of cane sugar or inversion of sucrose and the mixture of glucose and fructose (1:1) is known as invert sugar.
As the hemiacetal group is absent from sucrose, we can say that sucrose is not a reducing sugar, i.e., it does not reduce Fehling’s or Tollens’ reagent.
Now that we have a clear idea of sugars and what are their types and sources, let’s see some tests to identify sugars.
Sugars – Tests
There are numerous tests that may be used to determine the presence of reducing sugars on a qualitative or quantitative level.
- Benedict’s Test
- Fehling’s Test
- Tollens Test
Benedict’s Test
Benedict's test is a chemical procedure for identifying the presence of reducing sugars in a sample. This test can therefore identify simple sugars that have a free ketone or aldehyde functional group. Sodium citrate, sodium carbonate, and copper (II) sulphate pentahydrate make up Benedict's reagent, also known as Benedict's solution.
Benedict's reagent reacts with reducing sugars and forms a brick-red precipitate as a result, signalling that the test is successful. As shown in the diagram below, Benedict's reagent turns from clear blue to brick-red when exposed to reducing sugars.
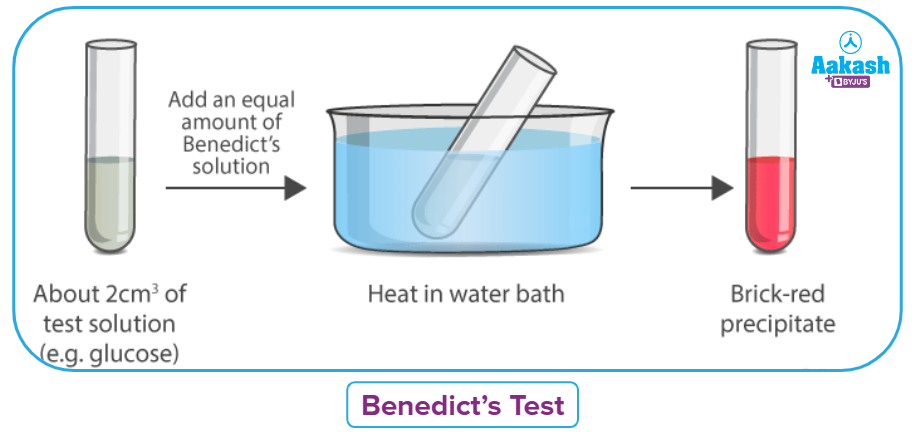
Procedure
Anhydrous sodium carbonate, sodium citrate, and copper (II) sulphate pentahydrate make up Benedict's reagent. It is an alkaline solution containing a cupric citrate complex. Reducing sugars convert the blue-coloured complex solution in Benedict's solution into a brick-red copper(I) oxide, which is seen as the precipitate and is responsible for the colour shift, after being added to the test solution. Non-reducing sugars are unable to accomplish this. Only a qualitative assessment of the presence of reducing sugars is provided by this test.
Benedict’s solution (Blue) Brick-red Precipitate
Fehling’s Test
Fehling’s test is an important method to tell the difference between aldehydes and ketones. Fructose is a ketohexose, while glucose is an aldohexose. So, we can clearly distinguish between them by using this test.
The solution is diluted in water and warmed until fully dissolved in order to perform Fehling's test. Then, while stirring, Fehling's solution is added. If there are reducing sugars in the solution, a rust or red-coloured precipitate should appear. The solution will remain blue or green if reducing sugars are not present.
Procedure
There are two solutions in the Fehling reagent: Fehling solution A and Fehling solution B. Aqueous copper sulphate is Fehling solution A, and alkaline sodium potassium tartrate is Fehling solution B. (Rochelle salt). Before the test, these two solutions are combined in equal parts. A reddish-brown precipitate is formed by heating an aldehyde with Fehling's reagent which indicates the presence of reducing sugars. The carboxylate anion is formed when aldehydes are oxidised. This test has no effect on aromatic aldehydes.
Red-brown precipitate
Tollen’s Test
Tollen’s reagent is silver nitrate (AgNO3) and ammonia solution (NH3) (ammoniacal silver nitrate). In the Tollens test, compounds with an aldehydic group (alpha-hydroxy ketones, aldehydes, and formic acid-its -COOH behaves as an aldehydic group) are usually utilised. The silver salt is transformed to silver metal, and the aldehyde is oxidised to carboxylic acid silver salt, yielding a white silver precipitate.
Procedure
The creation of silver metal produces a bright silver mirror when an aldehyde is heated with a newly prepared ammoniacal silver nitrate solution (Tollens' reagent). The aldehydes are oxidised to the carboxylate anion that corresponds to them. This process happens in an alkaline environment.
Sugars – Sources
Glucose: Honey, agave, molasses, dried fruit, fruits, fruit juices, and sweet maize are all naturally high in pure glucose. When consumed in moderation as part of a balanced diet, many foods, particularly fresh fruits, are nutritious. Sauces, salad dressings, pies, and sugary drinks are examples of foods with a lot of added glucose.
Fructose: Fructose, also known as "fruit sugar," is a monosaccharide that is similar to glucose. It is naturally found in fruit, honey, agave, and most root vegetables. It's also known as high-fructose corn syrup, and it's commonly found in processed foods. Fructose is found in sugar cane, sugar beets, and corn.
Sucrose: Sucrose is a glucose and fructose disaccharide. It's commonly referred to as "table sugar," but it can also be found in fruits, vegetables, and nuts in their natural state. It is, however, commercially made from sugar cane and sugar beets through a refinement process.
Sugars – Importance
- Carbohydrates are energy-giving fuels for the organism.
- The body breaks down carbohydrate-rich foods into glucose, which is then taken into the bloodstream.
- The brain, central nervous system, and red blood cells all require some glucose to function properly.
- Sugars and starches from carbohydrates are used by the body to provide glucose to the brain and energy to cells throughout the body.
Sugars – Side Effects
Sugar is good for our body but having excess sugar can lead to various health problems.
- Higher blood pressure
- Inflammation
- Weight gain
- Diabetes
- Fatty liver disease
- Damage to the retina
- Muscle and nerve damage
All the above illnesses are connected to an elevated risk of a heart attack when an excess amount of sugar is consumed. A healthy way to fulfil sugar requirements is to eat a variety of fruits, vegetables, and dairy products.
Practice Problems
1. What is the general formula of carbohydrates?
a. Cx(H2O)2y
b. Cx(H3O)y
c. Cx(H2O)y
d. Cy(H2O)y
Answer: C
Solution: Carbohydrates gained their name from the fact that they typically have the formula Cx(H2O)y., which was discovered early on. They appeared to be the ‘hydrates of carbon’.
So, option C is the correct answer.
2. Which of the given statements is true for reducing sugars?
a. Reduces Tollens’ reagent & Fehling’s solution.
b. It should have at least one hemiacetal or hemiketal functional group.
c. All monosaccharides and oligosaccharides except sucrose are reducing sugars.
d. All of the above
Answer: D
Solution: Reducing sugars reduce Tollens’ and Fehling’s solution. They contain at least one hemiacetal or hemiketal functional group. All monosaccharides and oligosaccharides except sucrose are reducing sugars. Hemiacetal (R’ = H or CH2OH) gives positive Tollens’ or Fehling’s test.
All the given statements are true regarding reducing sugars.
So, option D is the correct answer.
3. Fructose is a:
a. Ketohexose
b. Hemiketal
c. Aldohexose
d. None of the above
Answer: C
Solution: Fructose is an important ketohexose. The hydrolysis of the disaccharide sucrose yields it along with glucose. It is a basic sugar with the chemical formula C6H12O6 .
So, option C is the correct answer.
4. Which of the given statements is true about sucrose?
a. Its molecular formula is C12H22O11.
b. Sucrose is not a reducing sugar.
c. It does not reduce Fehling's or Tollens' reagent.
d. All of the above.
Answer: D
Solution: Sucrose is a crystalline white substance that dissolves in water. C12H22O11 is its molecular formula. Sucrose is dextrorotatory, although it converts to dextrorotatory glucose and laevorotatory fructose following hydrolysis. Sucrose is not a reducing sugar since it lacks the hemiacetal group, which means it does not reduce Fehling's or Tollens' reagent.
So, option D is the correct answer.
5. Tollens’ reagent is a solution of
a. Silver nitrate and ammonia
b. Formic acid and an aldehyde
c. Silver nitrate and formic acid
d. Ammonia and carboxylic acid
Answer: A
Solution: Tollen’s reagent is silver nitrate (AgNO3) and ammonia solution (NH3) (ammoniacal silver nitrate). In the Tollens test, compounds with an aldehydic group (alpha-hydroxy ketones, aldehydes, and formic acid-its -COOH behaves as an aldehydic group) are usually utilised. The silver salt is transformed to silver metal, and the aldehyde is oxidised to carboxylic acid silver salt, yielding a white silver precipitate.
So, option A is the correct answer.
6. Which of the following is correct for Benedict's reagent?
a. There is no glucose present if the colour of the sample turns to blue
b. It is a mixture of sodium citrate, sodium carbonate, and copper(II) sulphate pentahydrate
c. It forms a brick-red precipitate with non-reducing sugars.
d. Only A and B
Answer: D
Solution: Benedict's test is a chemical method for determining whether or not a substance contains reducing sugars. Benedict's reagent (also called Benedict's solution) is a complex mixture of sodium citrate, sodium carbonate, and copper(II) sulphate pentahydrate used in the test. When Benedict's reagent is exposed to reducing sugars, the reactions it undergoes produce a brick-red precipitate, indicating that Benedict's test is positive. The changes in the colour of Benedict's reagent (from clear blue to brick-red) are caused by exposure to reducing sugars.
So, option D is the correct answer.
Frequently Asked Questions – FAQ
1. What is blood sugar and why does it matter?
Answer: When we talk about blood sugar levels, we are talking about glucose, a molecule that powers our cells and has a big impact on our health. Glucose is the basic fuel that propels our bodies at the cellular level. Glucose is obtained from the foods we consume.
2. What is the chemical composition of the white sugar that we intake?
Answer: The most typical type of sugar is sucrose. Often referred to as "table sugar," it is a naturally occurring carbohydrate found in many fruits and plants. Usually, sugar is made from sugar cane or sugar beets. It is made up of bonded together amounts of 50% fructose and 50% glucose.
3. What are some healthy alternatives to sugar?
Answer: As wise substitutes for refined sugar, monk fruit, stevia, beetroot sugar, saccharin, certain sugar alcohols, and allulose have much fewer calories than table sugar and have minimal effects on blood sugar levels.
4. What is the type of sugar present in milk?
Answer: Lactose is present in milk. Because it can be found in milk and dairy products, lactose is also known as milk sugar. The primary disaccharide in milk is lactose, which the enzyme lactase catalyses into glucose and galactose. Because it is found in dairy products in high concentrations, lactose—also known as milk sugar—is a significant energy source.


