-
Call Now
1800-102-2727
Ionic Structure Type: Ionic Crystals, Rock salt (NaCl) Type Structure, Caesium Chloride
Do you like sketching/drawing?
If yes, then try to draw a structure of your study room in a three dimensional format?
Did I ask you for a tedious work?
Well, if this is difficult then imagine preparing a proper schematic drawing of your locality indicating all the details with precise measurement?

I know, even the greatest artist won’t be able to do that easily. Similarly, to determine the exact structure of a compound is quite difficult.
But the crystals contain such a elaborate arrangement that can be easily depicted in three dimensional form.
Simple ionic compounds are the compounds of the type AB or AB2 (or A2B) where A and B represent the positively and negatively charged ions respectively. It is more difficult to describe the structures of even these simple ionic compounds as compared to those of the elements. This is because an element consists of only one type of atoms, all of which occupy the lattice points, e.g., in case of copper which is face-centred cubic (FCC), all lattice points are occupied by copper atoms. On the other hand, in case of even the simple ionic compounds, the arrangement of both A and B ions has to be described.
Let’s find out how we can study them easily for a better understanding.
Table of content
- Ionic crystals
- Rock salt (NaCl) type structure
- Caesium chloride (CsCl) type structure
- Sphalerite or Zinc Blende (ZnS) type structure
- Structure of compound AB2type
- Structure of ionic compounds of the type A2B
- Effect of Pressure and Temperature on the Crystallization of NaCl into CsCl
- Calculation of number of unit cells in a given mass of an ionic compound
- Ions occupying voids
- Practice Problems
- Frequently asked questions(FAQs)
Ionic crystals
Most of the solids(92%) are crystalline and have a regular arrangement of small units. Though there are seven types of crystalline arrangement (crystal systems), solids prefer cubic or hexagonal close packing arrangement to maximize the available space.
Crystal formed by oppositely charged ions are called ionic crystal.
In ionic crystals-
- The ions are held together by strong electrostatic force of attraction
- Electrostatic attraction necessitates 1) contact between of cations and anions and 2) each ion being surrounded by oppositely charged ions.
- Total positive charges should be equal to the total negative charges to maintain neutrality of the solid. So, depending upon the relative charge carried by the ions we have a formula unit as AB, AB2, A2B etc.
- Ions occupy a close packed structure to minimize volume and there by maximize the electrostatic binding. Hence they crystallize in CCP or HCP structure.
- Generally anions are bigger in size and form a CCP structure.
- Cations occupy either of the tetrahedral or octahedral voids.
- The relative charge ratio of the ions and the relative radius ratio determine the structure adopted by the particular compound
- The ions are tightly bound and hence the ionic solids are insulators.
Structures of the ionic compounds of the type AB
Ionic compounds formed by ions having same charges have the formula unit as AB. These compounds have any one of the following three types of structures :
1. Rock salt (NaCl) type structure.
2. Caesium chloride (CSCI) type structure.
3. Zinc blende (ZnS) type structure.
The main features of each of these structures are discussed below:
Rock salt (NaCl) type structure
The following are the primary characteristics of this structure:
- It has face-centred cubic (fcc) arrangement (also called cubic close packing, i.e., ccp) in which Cl -ions occupy the corners and face centres of a cube while Na+ ions are present at the body centre and edge centres. (In fact, Na+ ions occupy all the octahedral voids).
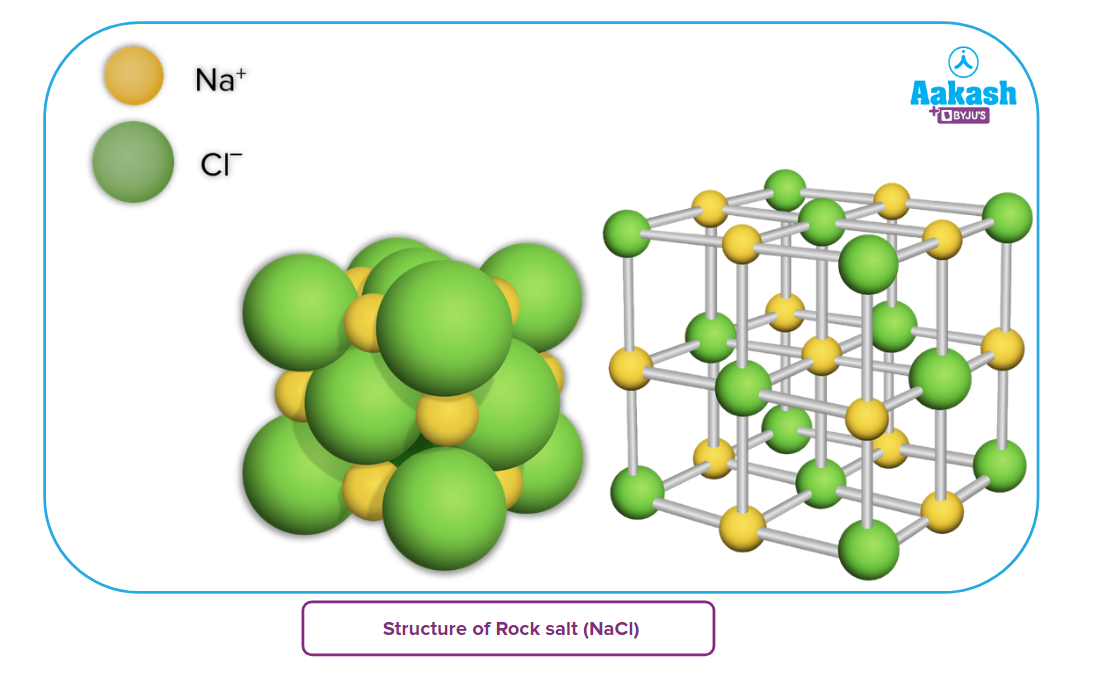
- Each Na+ ion is surrounded by six Cl -ions and each Cl -ion is surrounded by six Na+ ions. In other words, the Na+ ions as well as Cl - ions have coordination number of six, i.e., this structure has 6:6 coordination.
- A unit cell of NaCl consists of four NaCl units, i.e.,4 Na+ions and 4 Cl -ions as calculated below: 8 Cl -ions are present on the corners and 6 Cl -ions on the face centres so that Cl -ions per unit cell
Similarly, 12 Na+ ions are present on the edges and one within the body so that Na+ions per unit cell
A few examples of the compounds having a structure similar to that of NaCl are as follows:
(1) Halides of alkali metals (except those of caesium) and that of ammonium.
(ii) Oxides and sulphides of alkaline earth metals (except BeS).
(iii) Halides of silver (except silver iodide).
Caesium chloride (CsCl) type structure
The following are the primary characteristics of this structure:
- It has body-centred cubic (bcc) arrangement.
- Each Cs+ ion is surrounded by 4 Cl- ions and each Cl- ion is surrounded by 8 Cs+ions, i.e., this structure has 8: 8 coordination.
- A unit cell of CsCl consists of only one unit of CsCl, i.e., one Cs+ ion and one Cl- ion.
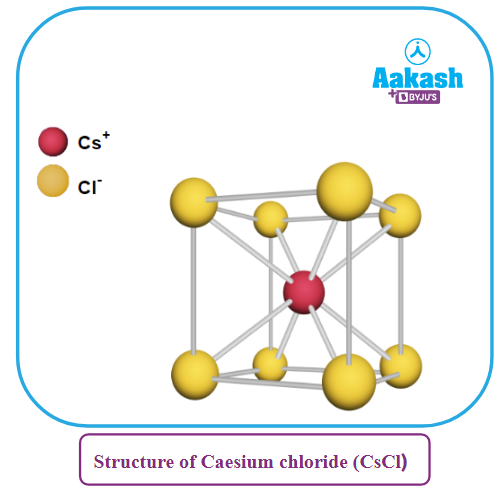
This explained as follows: may be
No. of Cs+ ions per unit cell = 1 (present within the body)
No. of Cl- ions per unit cell = (from corners) = 1
A few examples of compounds having CsCl structure are as follows:
CsBr, CsI, CSCN, TICI, TIBr, TII and TICN.
Sphalerite or Zinc Blende (ZnS) type structure
The following are the primary characteristics of this structure:
- The arrangement as possessed by ZnS is called cubic close packing (ccp) in which S2-ions form a face centred cubic, i.e.,S2- ions are present at the corners as well as at the centre of each face of the cube. Zn2+ ions are present at 1/4th of the distance along each body diagonal, one Zn2+ ion on each body diagonal (In fact, Zn2+ ions are present in the alternate tetrahedral voids).
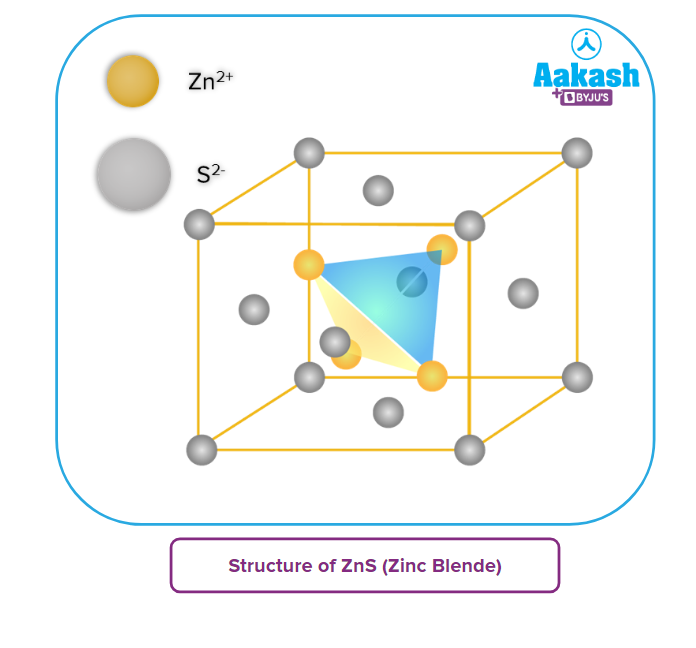
- Each Zn2+ ion is surrounded tetrahedrally by four S2- ions and each S2- is surrounded tetrahedrally by four Zn2+ ions. Thus, this structure has 4 : 4 coordination.
- One unit cell of ZnS consists of 4 Zn2+ ions and 4 S2- ions, i.e., it has 4 ZnS formula units per unit cell.
This may be explained as follows:
No. of S2- ions per unit cell = (from corners) + (from face centres) = 1 + 3 = 4
No. of Zn2+ ions per unit cell = 4 (present with the body of the unit cell)
- The structure of ZnS is similar to that of diamond in which in place of Zn2+ and S2-ions, there are carbon atoms at each site.
It is important to note that the electronegativity difference between Zn and S is very small (~ 0.9). therefore, the bond between Zn and S has a large amount of covalent character.
A few examples of ionic compounds having ZnS structures include CuCl, CuBr, Cul, Agl and BeS.
It may be noted that zinc sulphide, in fact, exists in two forms, called zinc blende and wurtzite. Similarities
- Both of them have a close-packed arrangement of S2- ions.
- In both of them, Zn2+ ions occupy the alternate tetrahedral voids (because number of tetrahedral voids is double the number of S2- ions)
- Both have 4 : 4 structure.
Differences
They differ only in the fact that zinc blende has cubic close packed (ccp or fcc) arrangement of S2- ions whereas wurtzite has hexagonal close-packed (HCP) arrangement of S2- ions. As a result, unit cell of wurtzite has 6 formula units of ZnS (whereas zinc blende has 4 formula units).
Structure of compound AB2type
These are the ionic compounds with doubly charged cation and singly charged such that the ratio of cation anionis 1:2. and most of these compounds have calcium fluorite (CaF2) type structure.
The following are the primary characteristics of this structure:
- It has cubic close packing (ccp) arrangement in which Ca2+ ions are present at the corners and the centre of each face of the cube and F ions occupy all the tetrahedral voids.
- Each Ca2+ ion is surrounded by 8 F - ions, i.e., has a coordination number of 8 whereas each F -ion is surrounded by 4 Ca2+ ions, i.e., has a coordination number of 4. Thus, this structure has 8:4 co-ordination.
- Each unit cell contains Ca2+ ions and 8 F - ions.
This may be explained as follows:
Ca2+ ions per unit cell
F -ions per unit cell =8 (present within the body) A few examples of compounds having CaF2 structure include

Structure of ionic compounds of the type A2B
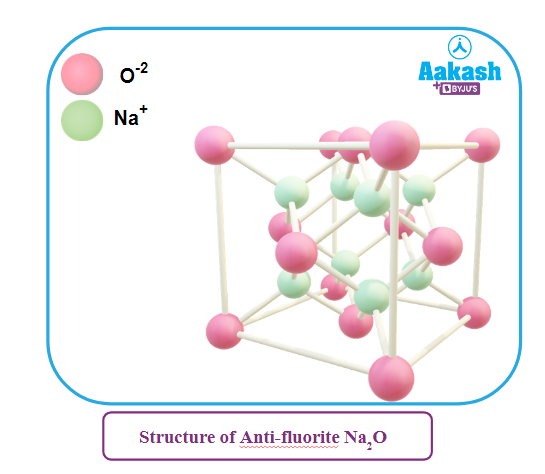
Some ionic compounds have the structure of the type A2B, e.g., Na2O. Their structure is similar to fluorite structure except that positions of the cations and the anions are reversed, the smaller cations occupying the position of F -ions whereas the larger anions occupy the position of Ca2+ ions. Hence, the compounds of the type A2B are said to possess antifluorite structure. For example, in Na2O, O2- ions have a cubic close packed arrangement and Na+ ions occupy all the tetrahedral voids. Each O2- ion is surrounded by 8 Na+ ions and each Na+ ion is surrounded by 4 O2- ions. Hence, it has 4: 8 coordination.
Many oxides and sulphides of alkali metals have antifluorite structure, e.g.,
Remember that CsCl lattice is slightly (about 1%) more stable than NaCl lattice because in CsCl, each ion is surrounded by more oppositely charged ions. The question arises then why halides of Li, Na, K and Rb do not have CsCl type lattice. This is answered by their radius ratio values as small cations cannot accommodate halide ions.
Effect of Pressure and Temperature on the Crystallization of NaCl into CsCl
On applying high pressure, NaCl structure having 6: 6 coordination changes to CsCl structure having
8: 8 coordination. Similarly, CsCl having 8: 8 coordination on heating to 760 K changes to NaCl structure having 6:6 coordination. Thus, increase of pressure increases the coordination
number whereas increase of temperature decreases the coordination number.
Calculation of number of unit cells in a given mass of an ionic compound
Remembering that one mole of an ionic compound contains Avogadro's number of formula units and also knowing the number of formula units per unit cell, the number of unit cells in a given mass can be calculated.
Ions occupying voids
Most of the ionic compounds have close-packed arrangement in which one type of ions (usually larger enes) form the close packed arrangement and the other type of ions (smaller ones) occupy the voids. A few examples are given below Compound forming the close-packed arrangement
|
Compound |
Ions forming the close packed arrangement |
Ions occupying the voids |
|
NaCl |
Cl- ions (fcc) |
Na+ions in all octahedral voids |
|
ZnS |
S2- ions (fcc) |
Zn2+ions in all alternate tetrahedral voids |
|
CaF2 |
Ca2+ ions (fcc) |
F- ions in all tetrahedral voids |
It is further interesting to mention here that
- In NaCl, there are 4 Cl-ions in the unit cell. Therefore, there are 4 octahedral voids, all of which are occupied by Na+ ions. Hence, there are 4 NaCl units per unit cell.
- In ZnS, there are 4 S ions in the unit cell. Therefore, there are 8 tetrahedral voids, half of which are occupied by Zn2+ions. Hence, again there 4 ZnS units per unit cell.
- In CaF2, there are 4 Ca2+ ions in the unit cell. Therefore, there are 8 tetrahedral voids all of which are occupied by F-ions. Hence, there are 4 CaF2 units per unit cell.
Practice Problems
Q1. Which of the following is not an ionic compound?
- NaCl
- ZnS
- CaF2
- None of the above.
Answer: (D)
Solution: Ionic compounds are those that are made up entirely of ions. These ions are electron-gaining or electron-losing atoms with a net positive or negative charge. Metals have a tendency to lose electrons, resulting in cations with a net positive charge. Nonmetals have a proclivity for gaining electrons, resulting in anions with a net negative charge. Examples: NaCl, ZnS, and CaF2.
Q2. Agl crystallizes in cubic close packed ZnS structure. What fraction of tetrahedral sites is occupied by Ag+ ions?
Solution: In the face-centred unit cell of Agl, there are 4 Ag+ ions and 4 I-ions. As there are 4 I- ions in the packing, therefore, there are eight tetrahedral voids. Of these, half are occupied by silver cations.
Q3. Write the coordination numbers of cations and anions in the following ionic compounds : the (a) Zinc blende (b) Fluorite?
Solution: Zinc blende = ZnS, Zn2+ = 4, S2- = 4
Fluorite = CaF2, Ce2+ = 8, F-= 4
Q4. In each of the compounds: NaCl, ZnS and CaF2, write (i) ions occupying the voids (ii) types of voids occupied (iii) fraction of voids occupied.
Solution: (a) NaCI = Na+ ions in all the octahedral voids
(b) ZnS = Zn2+ ions in alternate tetrahedral voids
(c) CaF2 = F-ions in all the tetrahedral voids.
Frequently asked questions(FAQs)
Q1. Why do the shape of ionic crystal varies?
Answer: Ionic crystals are made up of ions bound together by electrostatic attraction. Depending on the size of the ions or the radius ratio, they are arranged differently (the ratio of the radii of the positive to the negative ion).
Q2. Why solid ionic crystals are unable to conduct electricity?
Answer: Because the ions are firmly held in place, solid ionic compounds do not conduct electricity. The ions are unable to move in order to conduct an electric current. The charged ions in an ionic compound, on the other hand, are free to move when it melts. As a result, molten ionic compounds are capable of conducting electricity.
Q3. Can we dissolve ionic crystals in water?
Answer: The majority of ionic compounds are water soluble. Ionic compounds are soluble in water because polar water molecules have a strong attraction for charged ions, which causes the charged ions to become solvated as they dissociate into the water. But the solubility shall vary depending on the lattice energy and charge on the ions of the crystal
Q.4. Do lattice points and unit cells differ in any way?
Answer: A lattice is a regular recurring three-dimensional organization of atoms, ions, or molecules in a metal or other crystalline solid, whereas a unit cell is a simple arrangement of spheres (atoms, molecules, or ions) that resembles a lattice's repeating pattern.


