-
Call Now
1800-102-2727
Steady-State Approximation - Rate of Chemical Reaction, Reaction Mechanism Steady State Approximation, Application, Practice Problems, FAQs
You know that maize on heating gives the puffy popcorn. But, it is not immediate and, maize goes through stages of maize→ half cooked→ popcorn. Have you seen popcorn making? But, in the popcorn machine, the maize gets fed through the hooper continuously on the heater and the fluffy popcorn is regularly collected and served. The time interval is good enough for the maize to be completely converted and you do not get any half-cooked maize. Nevertheless, half-cooked maize is an intermediate though it is not seen. The heating rate is so adjusted that there is a minimum amount of half-cooked maize in the vessel.

Most of the chemical reactions are similar to this. They are mostly a multistep process, giving one or more intermediates. These intermediates can either go back to the reactant are react further to give other intermediates or product(s). At the start, the amount of these intermediates may be higher, but their concentrations remain constant over time, decreasing at the completion of the reaction. The rate of reaction of these intermediates decides the overall reaction rate.
Let us see what steady-state approximation is and how it is useful in determining the rate law.
TABLE OF CONTENTS
- Rate of Chemical Reaction
- Reaction Mechanism
- Steady State Approximation
- Application of Steady State Approximation
- Practice Problems
- Frequently Asked Questions - FAQs
Rate of Chemical Reaction
The occurrence of any noticeable chemical reaction is governed by two factors-
i) Feasibility of the reaction ie whether the reaction is spontaneous or needs some other inputs
ii) Speed(rate) at which the reactants will be transferred to the products.
The spontaneity of a reaction is governed by thermodynamic principles and can be predicted from Gibb’s free energy values.
The conversion rate is a kinetically controlled process that must be experimentally determined.
The rate of a reaction is measured as either rate of disappearance of the reactant of the appearance of the product.
For a reaction, A → B
Rate of the reaction, : the negative sign indicates the decreasing concentration of reactants, as rates can only be positive.
The Law of mass action relates the rate of reaction to the concentration of the reactants.
According to the Law of mass action, r = k[A]a, where k is the rate constant, [A] is the molar concentration and ‘a’ is a factor called the order of the reaction, to be determined experimentally, and not from the balanced stoichiometric chemical reaction. The overall order of reaction in multiple reactants can be of any value from zero and fractions too.
Reaction Mechanism
Chemical reactions are seldom direct single-step reactions. Either they, involve in one or more steps of intermediate products or intermediates (unstable reaction complexes) which further react to form the ultimate product. It means that most reactions are multistep processes and each reaction step(referred to as the elementary step) has its rate of reaction. All the reactions involved are called mechanisms of reaction.
Let us consider the reaction between hydrogen and iodine.
The stoichiometric reaction is written as - H2 + I2 → 2HI
This reaction is never a single-step reaction but involves three steps.
- H2 → 2H
- I2 → 2I
- H + I → HI
All the three individual reaction steps together makes the mechanism. Each one has different reaction rates. The rate of the third reaction is the slowest of all three.
The slowest of the multistep reaction decides the ultimate reaction rate of the conversion and is hence referred to as the rate-determining step of the entire reaction.
Rate of the reaction, r = k [H][I] and not r = k [H2][I2]
But, experimental characterisation of the step is impossible as we do not have the concentration of [H] and [I]. For determining the rate of such multistep reactions, some assumptions are needed to relate the intermediate concentrations to the known reactant concentrations, and the one used is a steady-state approximation.
Steady State Approximation
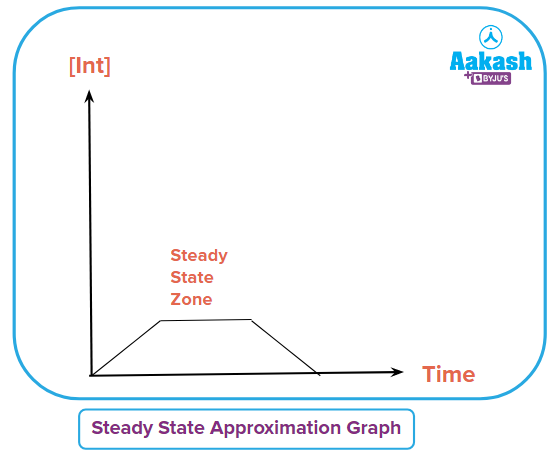
Steady state here means constant concentration. In steady-state approximation, concentrations of all the intermediates or complexes are considered to remain unchanged. At the start of the reaction, their amount could reach a peak value that remains constant there without any further change and decreases to zero at the end of the reaction. In other words, the concentration of the intermediate can be considered so small that it can be considered negligible or not changing. So during the reaction, the rate of change of the intermediate is considered zero.
Application of Steady State Approximation
Consider the decomposition of dinitrogen pentoxide to yield nitrogen dioxide and oxygen as per the stochiometric reaction-
2 N2O5 → 4 NO2 + O2
Rate of decomposition cannot be calculated from this stochiometric equation. The decomposition involves four elementary steps as follows
N2O5 → NO2 + NO3 ……..(i) r1 = k1 [N2O5 ]
NO2 + NO3 → N2O5 ……..(ii) r3 = k2[NO2 ][NO3 ]
NO2 + NO3 → NO + NO2 + O2 ……..(iii) r3 = k3[NO2 ][NO3 ]
NO + NO3 → 2 NO2 ……..(iv) r4 = k4[ NO][ NO3 ]
Multiplying equation (i) by 3 and adding of all these reactions gives, the stochiometric equation, 2 N2O5 → 4 NO2 + O2
Rate of the reactions are r1, r2, r3 and r4 and rate constants for the reactions are k1,k2, k3 and k4 respectively for the reactions (i), (ii), (iii) and (iv).
The rate of the reaction r3 is the slowest and rate-determining step.
Rate of the whole reaction r = r3 = k3[NO2 ][NO3 ]
But the concentrations of [NO2 ] and [NO3 ] are unknown and has to be expressed in terms of reactant N2O5
Steady state approximation is applied here for the relationship.
In this above reaction, NO and NO3 are intermediary products which don’t remain as end products when the reaction concludes. Products of the first reaction either return back to give the reactants or proceed further to give part of the products.
As per the steady state approximation, the concentration of the intermediates is assumed to be constant or not changing ie rate of formation = Rate of disappearance
Considering the intermediate NO, it is formed in reaction (iii) and consumed in reaction (iv).
So, Rate of reaction (iii) = Rate of reaction (iv)
k3[ NO2 ][ NO3 ] = k4[ NO][ NO3 ]
So, [NO] = k3k4[ NO2 ]
Considering the intermediate NO3, it is formed in reaction (i) and consumed in all other reactions.
So, Rate of reaction (i) =Rate of reaction (ii) +Rate of reaction (iii) + Rate of reaction (iv)
k1 [N2O5 ] = k2[NO2 ][NO3 ] + k3[ NO2 ][ NO3 ] + k4[ NO][ NO3 ]
Substituting the concentration of intermediate NO in the equation.
k1 [N2O5 ] = k2[NO2 ][NO3 ] + k3[ NO2 ][ NO3 ] + k3[ NO2][ NO3 ]
= k2[NO2 ][NO3 ] + 2k3[ NO2 ][ NO3 ] = [ NO3 ][NO2 ][ k2+2k3 ]
[ NO3 ] = k1 [N2O5 ][NO2 ][ k2+2k3 ]
Let ius find the rate of of decomposition of dinitrogen pentoxide,
Rate of the whole reaction r = r3 = k3[NO2 ][NO3 ]
Substituting for [NO3 ],
Rate of the whole reaction r = k3[NO2 ]k1 [N2O5 ][NO2 ][ k2+2k3 ] = k3k1 [N2O5 ] [ k2+2k3 ]
Since the ratio of rate constants also will only be constant and replacing, k3k1 [ k2+2k3 ] = k,
Rate of decomposition r = k[N2O5 ]
Note that the rate of decomposition through directly proportional to the concentration of the reactant, the proportionality constant is different.
Practice Problems:
Q1. Steady State Approximation is based on____
A) All intermediates are formed and decomposed at the same rates.
B) Intermediates are in large concentration and hence can be considered as not changing
C) Intermediates are produced and consumed at the same rates
D) Equilibrium state is also applicable to the multiple-step reactions
Answer: Option (C)
Solution: The steady-state approximation is a method assumes that the rate of formation and consumption of intermediates are same so as the change with time is zero.
Q2. The rate-determining step of a series of reactions is the one ____
A) That is fastest
B) That is slowest
C) That does not contribute to the reaction
D) That does not occur
Answer: Option (B)
Solution: Rate determining step is the slowest of all the elementary reaction steps. The rate-determining step is important in deriving the rate equation of a chemical reaction.
Q3. For the parallel reaction A → B and A → C, of rate constants k1 and k2 respectively, both reactions of order 1, the rate expression is given as ____
A) -rA = k1[A] +k2 [A]
B) - rA = k1[A] - k2 [A]
C) - rA = k2 [A]
D) - rA = k1[A]
Answer: Option (A)
Solution: For each of the first order reaction, −d[A]dt = k[A]
A forms two products: B and C. The concentration of A decreases as both reactions progress, parallel.
Q4. The reaction between hydrogen and iodine to form hydrogen iodide is by ____
A) direct reaction
B) Two-step mechanism
C) Three-step mechanism
D) complex mechanism
Answer: Option (C)
Solution: This reaction is never a single-step reaction but involves three steps.
- H2 → 2H
- I2 → 2I
- H + I → HI
Frequently Asked Questions
Q1. What does the steady state assumption assume?
Answer: Steady state occurs when the rate of formation and breakdown of the intermediate are equal. The steady-state assumption relies on the fact that both the formation of the intermediate from reactants and the formation of products from the intermediate have rates much higher than their corresponding reverse reactions.
Q2. What is the difference between the equilibrium reactions and steady state approximation.
Answer: In equilibrium reaction, all the concentration, of reactants, intermediates and products all remain constant, after some time and remains so.
In steady-state, only the intermediate's concentration is assumed to remain constant. Also, the intermediate concentration remains constant only in the middle of the reaction. At the start, it reaches a maximum and at the end, it also becomes zero.
Q3. Is steady-state approximation applicable only to chemical reactions?
Answer: A steady-state reaction is applicable when the intermediate concentration is very low to be considered negligible. It is widely applied to enzyme-catalysed biochemical reactions.
Q4. When the steady-state approximation is not applicable?
Answer: The steady-state approximation is applicable only in relatively stoichiometric concentration ranges. If one of the concentrations of the rate equation is very high, this approximation will not work.


