-
Call Now
1800-102-2727
Sigma Bonds - s-s Overlapping, s-p Overlapping, p-p Overlapping, Practice Problem & FAQs
Have a look at this kid and the travel guide crossing the bridge and try to answer these questions.
Will he be able to cross the river if the bridge isn't there?
Will he be able to cross this bridge if the bridge is damaged?
The answer to both the questions is No. The bridge helps him to cross the river and reach his destination. So, everything looks stable and sorted with this bridge.

Now if you’re thinking that why are we talking about this bridge here and how it is related to chemistry?
In chemistry, atoms and molecules also make bonds with each other and help each other in some way or another to attain stability just like this bridge is doing. This bridge is the bond between two river shores and connects these shores.
Let’s try to understand bonds in more detail.
Table of Contents
- What is a Sigma Bond?
- Types of Sigma Bonds
- Strength of Sigma Bonds
- Practice Problems
- Frequently Asked Questions – FAQs
What is a Sigma bond?
The sigma bonds are formed when along the internuclear axis orbitals are overlapped head-on and that too positively (both the lobes overlapping should have the same phase). As the overlapping is direct of the orbitals, sigma bonds are the strongest covalent bonds. The bonding electrons are the electrons which participate in the overlapping.
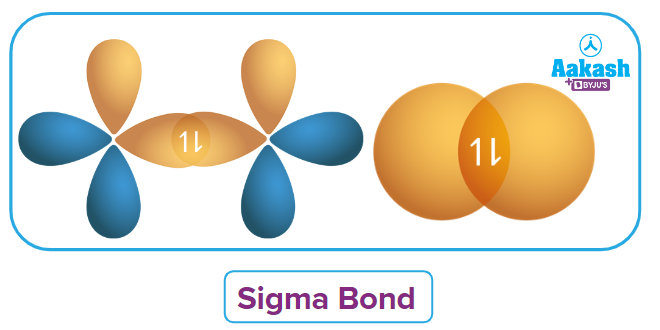
There are different types of overlapping of atomic orbitals. This differentiates sigma (𝜎) and pi (𝜋) bonds from each other. The overlap happens in two ways, resulting in two different types of covalent bonds: sigma and pi bonds. The head-to-head overlapping of atomic orbitals forms sigma bonds, whereas the pi bonds are formed by the lateral overlapping of the atomic orbitals. Atomic orbitals overlap affects a number of bond parameters, including bond length, bond angle, and bond enthalpy.
Types of Sigma Bonds
All single bonds are, in general, sigma bonds. Following are the basic combinations of the atomic orbitals overlapping:
s-s overlapping:
Orbitals undergo head-on overlapping along the internuclear axis in this type of overlapping. Both the s orbitals have to be half-filled before overlapping and the spin of both the electrons should be opposite.
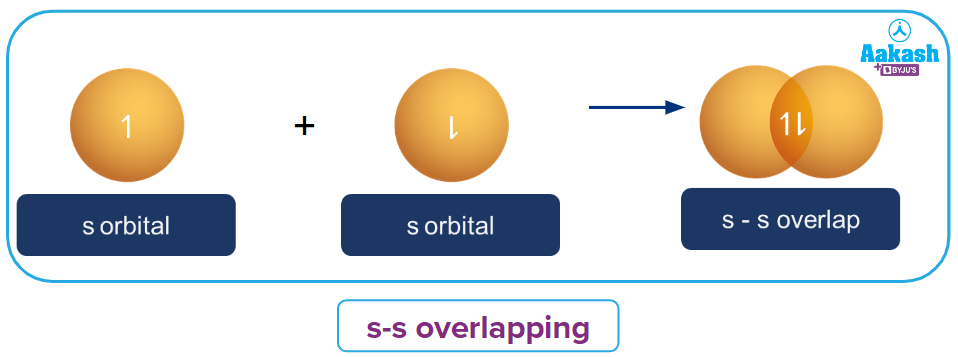
Above is an illustration of two s-orbitals overlapping to form a sigma bond.
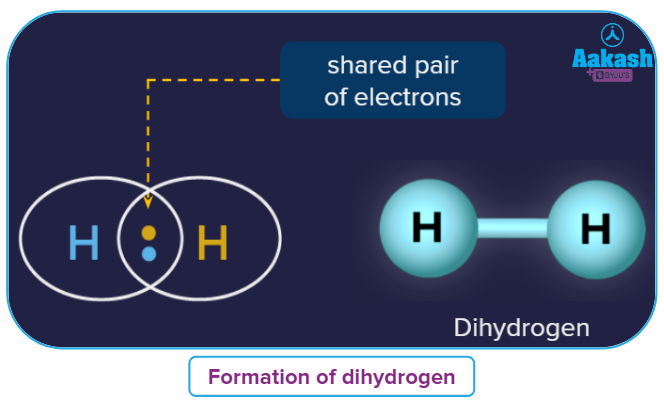
In H2 molecule, each hydrogen atom has a half-filled s-orbital and they overlap with each other to form a sigma bond.
s-p overlapping:
One s-orbital which is half-filled overlaps with a p-orbital which is also half-filled, forming a covalent sigma bond. This overlapping happens along the axis. This is depicted in the diagram below:
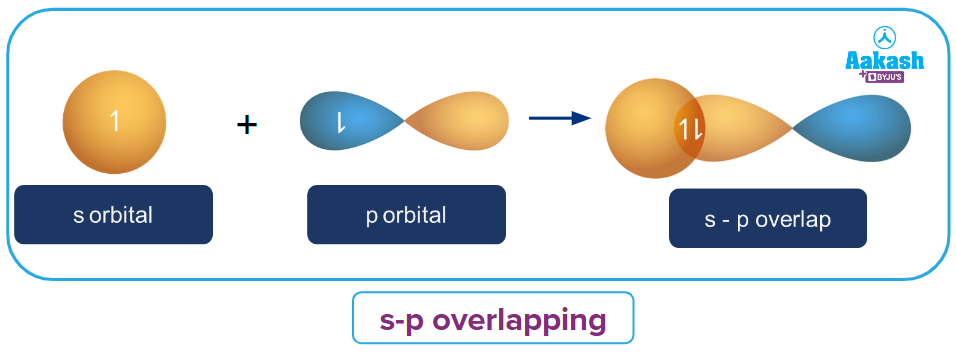 In hydrogen chloride (HCl), this form of overlapping can be seen. The overlap of the 1s orbital of the hydrogen atom with the 3p orbital of the chlorine atom forms the sigma bond.
In hydrogen chloride (HCl), this form of overlapping can be seen. The overlap of the 1s orbital of the hydrogen atom with the 3p orbital of the chlorine atom forms the sigma bond.
p-p overlapping:
Two half-filled p-orbitals overlap along the axis in this type of overlapping. This type of overlapping is depicted in the diagram below:
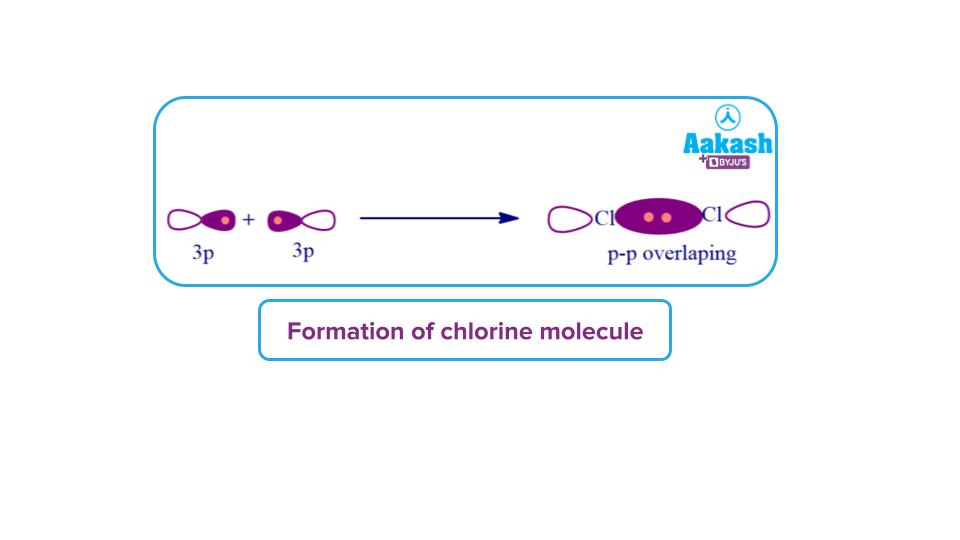
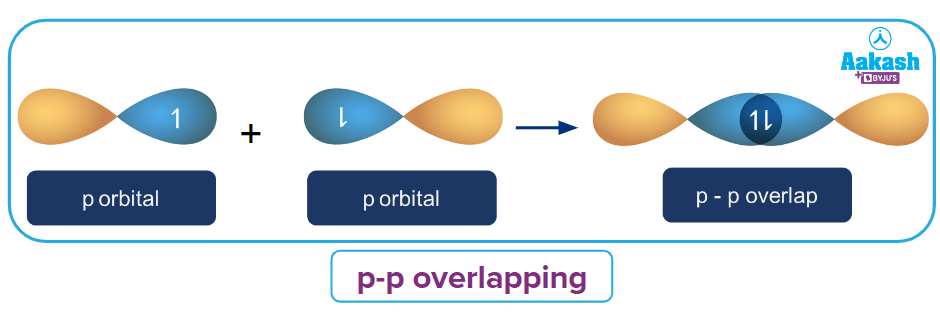 The 3pz orbitals of two chlorine atoms overlaps head on in a Cl2 molecule to form a sigma bond.
The 3pz orbitals of two chlorine atoms overlaps head on in a Cl2 molecule to form a sigma bond.
Strength of Sigma Bonds
The strength of a bond depends upon the extent of overlapping. The more the overlapping, the stronger the bond.
Order of strength of sigma bonds is:
p-p overlap > s-p overlap > p-p overlap
The s-orbital is non-directional as the charge distribution in s - orbital is spherical. p - orbital has directional charge distribution. Thus, the sigma bond formed from the overlapping of two p - orbitals will have greater overlapping compared to the bond formed by the overlapping of s-p orbitals followed by the bond formed by the overlapping of s-s orbitals.
Practice Problems
Q1. Which of the following compounds contain only the sigma bonds?
A. CH4
B. O2
B. C2H2
C. C3H4
Answer: (A)
Solution: We can clearly see from the structure of methane that the carbon atom is attached to the four hydrogen atoms with a single bond which shows that sigma bond formation is taking place. O2 has one sigma bond and one pi bond. C2H2 has three sigma bonds and two pi bonds. C3H4 has 6 sigma bonds and 2 pi bonds.
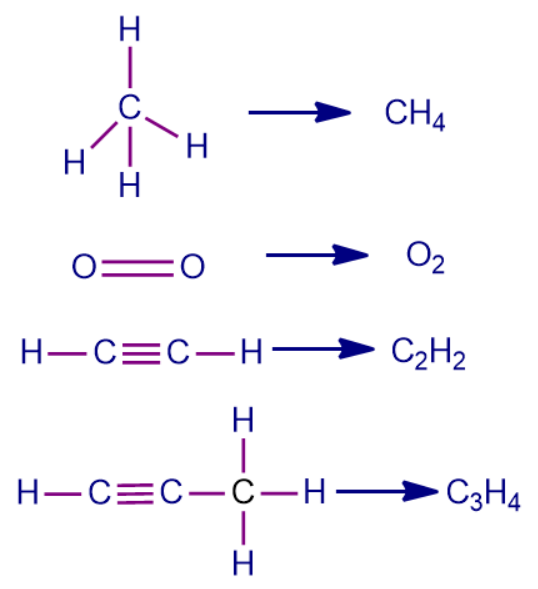
So, option A is the correct answer.
Q2. How many sigma bonds are there in a water molecule?
A. 1
B. 2
C. 3
C. 4
Answer: (B)
Solution: If we look at the structure of a water molecule, we can clearly see that a water molecule has 2 sigma bonds.
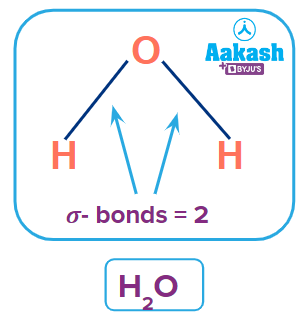
So, option B is the correct answer.
Q3. In a triple bond, how many sigma bonds are there?
A. 0
B. 1
C. 2
D. 3
Answer: (B)
Solution: Every single bond is made up of a sigma bond, while the double bond is formed of one pi and one sigma bond. A triple bond, on the other hand, is made up of two pi bonds and one sigma bond.
So, option B is the correct answer.
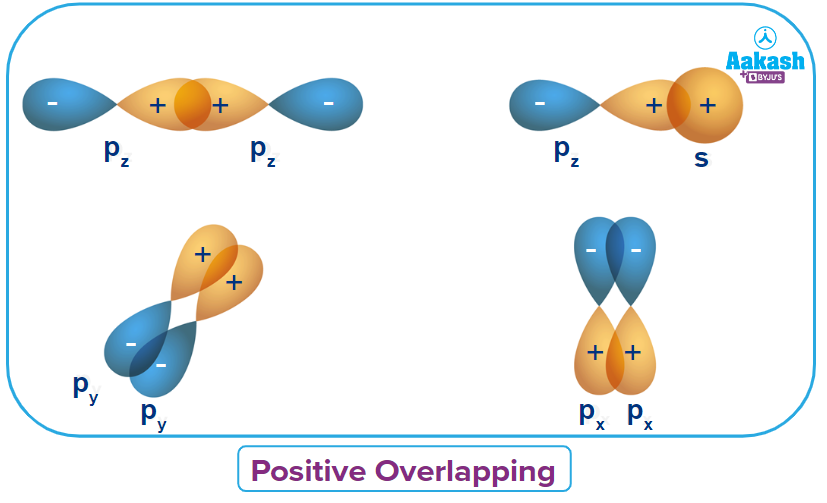
Q4. Which of the following statements is correct about the positive overlapping?
A. It involves the overlap of the lobes of the same sign (phase).
B. It has an inability to interact.
C. It involves the overlap of the lobes of the opposite sign (phase).
D. None of the above
Answer: (A)
Solution: Positive overlapping takes place due to the overlap of the lobes of the same sign (phase). It leads to attractive interactions. Given below are some of the examples of positive overlapping:
Frequently Asked Questions - FAQs
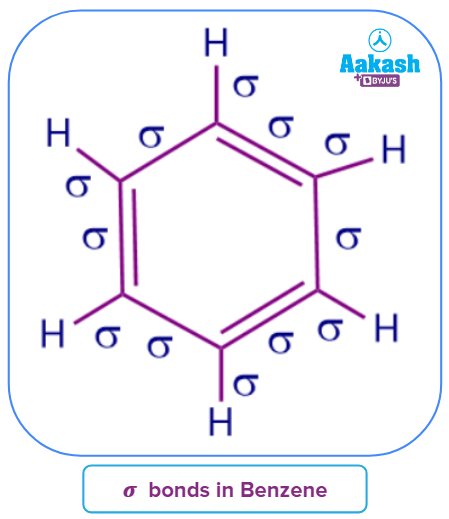
Q1. In a benzene molecule, how many sigma bonds are there?
Answer: Six carbon-carbon single bonds, all of which are sigma bonds, make up the benzene ring. There are also six sigma bonds between carbon and hydrogen. As a result, a benzene molecule has a total of 12 sigma bonds.
Q2. Which bond is more powerful, the sigma or the pi bond?
Answer: In contrast to sigma bonds, pi bonds are created by sideways overlapping of atomic orbitals. Because of the large difference in orbital overlap, the sigma bond is stronger than the pi bond as the head-on overlapping of orbitals is stronger.
Q3. What orbital combinations are possible in sigma bonds?
Answer: The following are the three most prevalent overlap conditions that lead to sigma bonds:
- s-s overlapping
- s-p overlapping
- p-p overlapping
Q4. How is sigma bond different from the pi bond?
Answer: Below is a list of the key differences between sigma and pi bonds:
|
Sigma bond |
Pi bond |
|
The overlapping orbitals may be pure or hybrid. |
The overlapping orbitals need to be hybridized. |
|
These bonds are powerful and contain a significant amount of energy. |
These bonds are comparatively weak. |
|
These bonds can exist independently. |
These bonds must exist along with the sigma bond. |
|
These bonds can impact the shape of the molecules. |
The shape of molecules is unaffected by these bonds. |


