-
Call Now
1800-102-2727
Sequential First Order Reactions - Definition, Examples, Concentration of the Intermediates, Practice Problems and FAQs
Do you know how a car is manufactured in the car factory?
Different parts are assembled on the assembly line. A car is assembled in the multiple steps for example lets start with chassis, then tyres will be fixed, then seats will be fixed, in the next step engine will be mounted, then there will be a set up of gear box and steering wheel. So, a car is manufactured in the multiple steps and to perform the whole job every step is to be performed in the stipulated time. Let’s suppose if one step is delayed, the whole process will be delayed. So, it can be said that the ultimate product depends on all the previous steps and the rate at which these steps are occurring.
Similarly, there are sequential reactions, where the concentration of the final product depends on the concentration of the initial reactant and all the intermediates. So, let’s discuss the kinetics of sequential first order reactions.
Table of Contents
- What is a Sequential First Order Reaction?
- Concentration of the Intermediate
- Maximum Concentration of the Intermediate
- Practice Problems
- Frequently Asked Questions – FAQs
What is a Sequential First Order Reaction?
Reactions which proceed from reactants to final products through one or more intermediates are known as sequential or consecutive reactions. All the steps of these reaction to form the final product should be first order reaction to call it as a sequential first order reaction.
For example:
A → B → C → D
Here, A is the reactant, B & C are the intermediates, D is the final product.
A → B, B → C,C → D all these three reactions follow first order kinetics.
Concentration of the Intermediate
Consider the following sequential first order reaction;
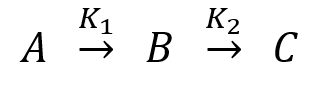
Where, B is the intermediate, K1 & K2 are the rate constant of A → B, B → C reactions respectively.
The differential rate equations can be written as:
Rate of disappearance of
Rate of change of
Rate of formation of
Integrating the equation (i) within the limits, we get;
Where, Ao is the initial concentration of A.
Now to determine the concentration fo the intermediate i.e., B, we need to integrate the equation (ii). But equation (ii) is a differential equation so we have to solve it by the method of linearisation of the differential equation.
Equation (ii) can be written as;
Now, in (v), we can put the value At using equation (iv).
Now, equation (vi) can not be solved by directly integrating it, as we are unable to separate the two variables B & t. So, we multiply both sides by an integrating factor i.e., eK2t. The equation (vi)becomes:
The L.H.S of the equation (vii) is equal to (which is a differential of ).
Equation (vii) now becomes:
Equation (viii) now becomes:
Integrating the equation (ix) between the limits, 0 to t.
⇒
From equation (x), we can find out the concentration of the intermediate B at any instant of time.
So, here, the concentration of A decreases exponentially. The concentration of B first increases and reaches a maximum and then decreases thereafter. The concentration of C increases gradually. This can be represented graphically.
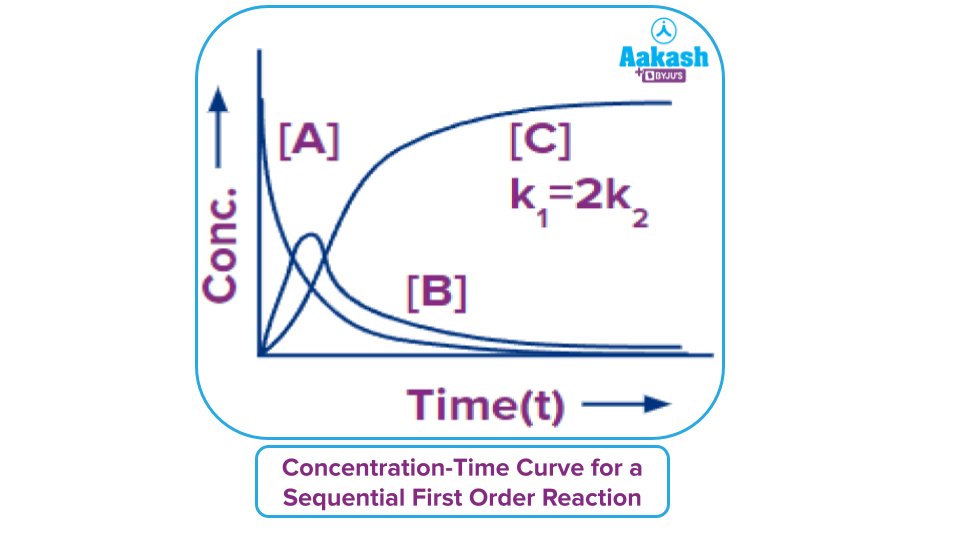
Maximum Concentration of the Intermediate
To find out the maximum concentration of the intermediate B, first we need to calculate the time at which the concentration of B is maximum.
We know at maxima, the slope of the curve would be zero. So, at that instant of time dBdt would be equal to zero.
So from the equation (ii), we can write;
Substituting the values of A and B in the above equation;
Reaaranging the above equation, we get;
Now, by putting this value of tmax in the equation (x), we get the maximum concentration of B.
Rearranging the above equation we get;
⇒
Practice Problems
Q1. Consider the following sequential first order reaction;
![]()
The time at which the concentration of B would be maximum is:
(Given: K1=20 & K2=10)
a.
b.
c.
d.
Answer: (A)
The time at which the concentration of B would be maximum is:
Putting the given values of K1=20 & K2=10, we get;
Q2. Consider the following sequential first order reaction;
![]()
The maximum concentration of B would be:
(Given: K1=20, K2=10 & the concentration of A is AO)
a.
b.
c.
d.
Answer: (B)
We know the formula to calculate the maximum concentration of B.
Putting the given values of K1=20 & K2=10, we get;
Q3. Consider the following sequential first order reaction;
![]()
The concentration of B at the 5th second would be:
(Given: K1=30, K2=10 & the initial concentration of A is 5 mol L-1)
a.
b.
c.
d.
Answer: (A)
We know the formula to calculate the concentration of B at any instant of time.
Putting the given values of K1=30, K2=10,[A]O=5 mol L-1 & t=5, we get;
Frequently Asked Questions-FAQs
Q. What is meant by the differential rate equation?
Answer: Differential rate laws are used to express the rate of a reaction in terms of the changes in reactant concentrations over a small interval of time.
For a chemical reaction; A +B → C+D, It can be expressed as:
R=-d[A]dt=k[A]x[B]y
Where, k is the rate constant, x & y are the order with respect to A & B respectively.
Differential rate law can also be used to calculate the instantaneous rate of a reaction, which is the rate of reaction defined for an infinitesimally small interval of time.
Q. In the sequential reactions, does the concentration of the initial reactant depends on the concentration of intermediates and product which form later?
Answer: In the sequential reactions, the concentration of the initial reactant does not depend on the concentration of intermediates and products. But concentration of intermediates and product does depend on the concentration of the reactant at any instant of time.
Q. How the rate of change of concentration of the intermediates depends on the concentration of the reagent before in the sequential reaction?
Answer: Consider the following sequential first order reaction;
![]()
Here, the concentration of intermediate depends on both the concentration reactant A and the concentration of B itself.
So, the rate of change in the concentration of B can be written as:


