-
Call Now
1800-102-2727
Revisiting Protein- Definition, Classification, Source, Importance, Test for Proteins, Denaturation of Protein, Amino Acid, Practice Problems, FAQs
Are you one of those, health-conscious doing regular daily workouts to keep yourself fit and maintaining body weight? If so you would have been advised to consume more protein-rich food and less carbohydrate-rich food. Do you know the reason?
Every single person requires protein on a daily basis. Protein becomes even more important as we become more active, and while most people understand that it is important, we often don't understand why.
Proteins are made up of amino acids and are the primary building blocks of our muscles, bones, skin, tissues, and organ systems. When we eat protein, our bodies digest it and break it down into individual amino acids, which are then used to develop new proteins across the body. It is critical to consume enough protein; otherwise, the body will have to break down muscle to obtain the amino acids it requires to function.
Proteins were recognised as important by chemists in the early nineteenth century, including Swedish chemist Jöns Jacob Berzelius, who coined the term protein in 1838, a word derived from the Greek “proteios”, which means "first place."
Let us have a tour of this article to know more about protein.

Table of Content
- Definition of Protein
- Structure of Protein
- Classification of Protein
- Sources of Protein
- Importance of Protein
- Test for Protein
- Denaturation of Protein
- Amino Acid
- Practice Problem
- Frequently Asked Questions-FAQs
Definition of Protein
Proteins are very large molecules made up of basic units known as amino acids. Proteins are made up of carbon, hydrogen, oxygen, nitrogen, and sulphur.
Protein molecules are large, complex molecules made up of twisted and folded amino acid strands. Proteins are extremely complex molecules that play an active role in the most fundamental and important aspects of life. Metabolism, movement, defence, cellular communication, and molecular recognition are some important proteins.
Structure of Protein
Protein structures is understood at four levels based on their complexity.
Protein Primary Structure
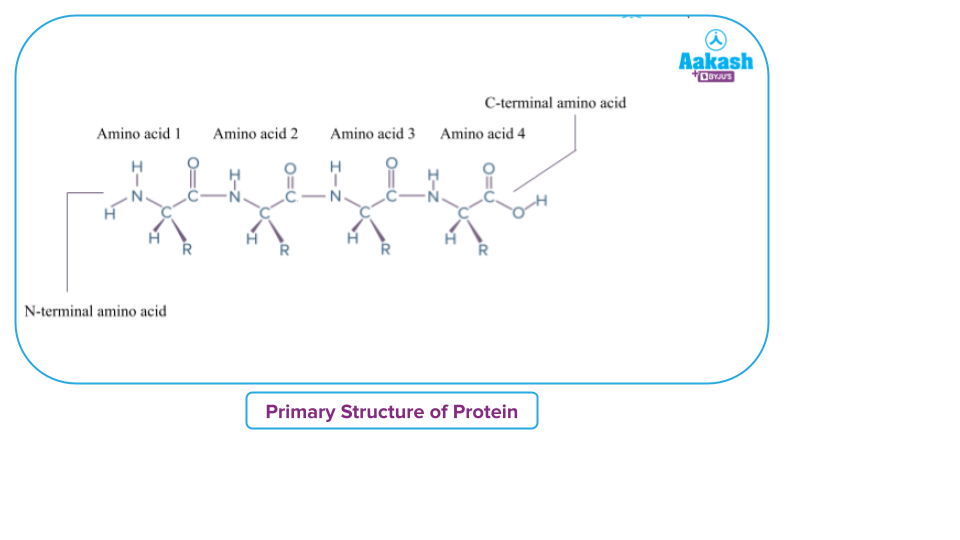
The primary structure of proteins is a linear chain of amino acid residues linked together by covalent peptide bonds. It gives amino acid positional information, such as which amino acid is in the first position, which one is in the second position, and so on.
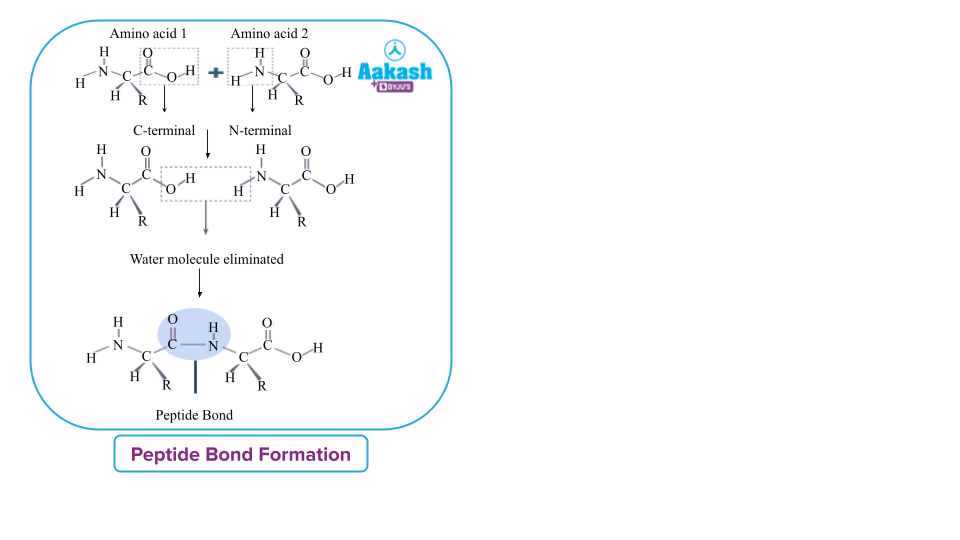
The primary structure of the protein can be represented as a line, with the first amino acid on the left and the last amino acid on the right.
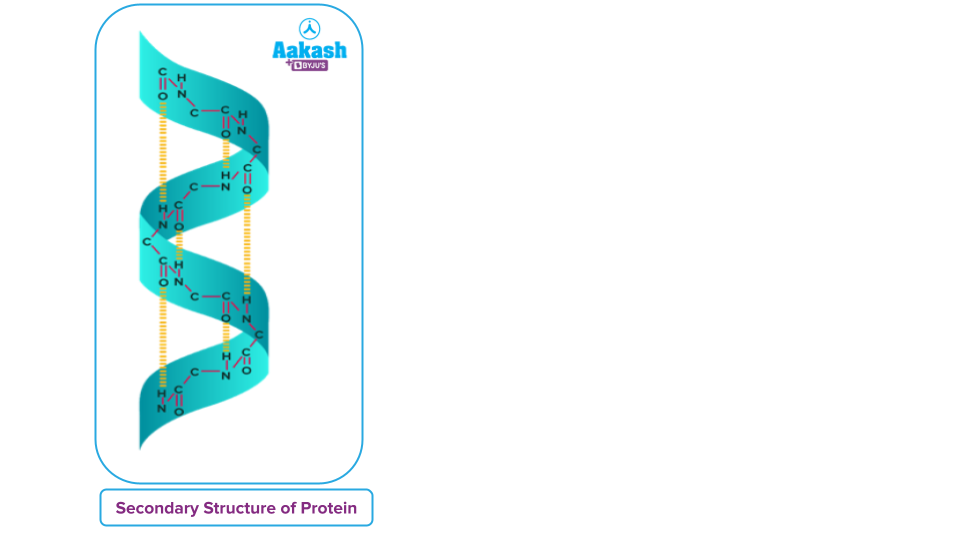
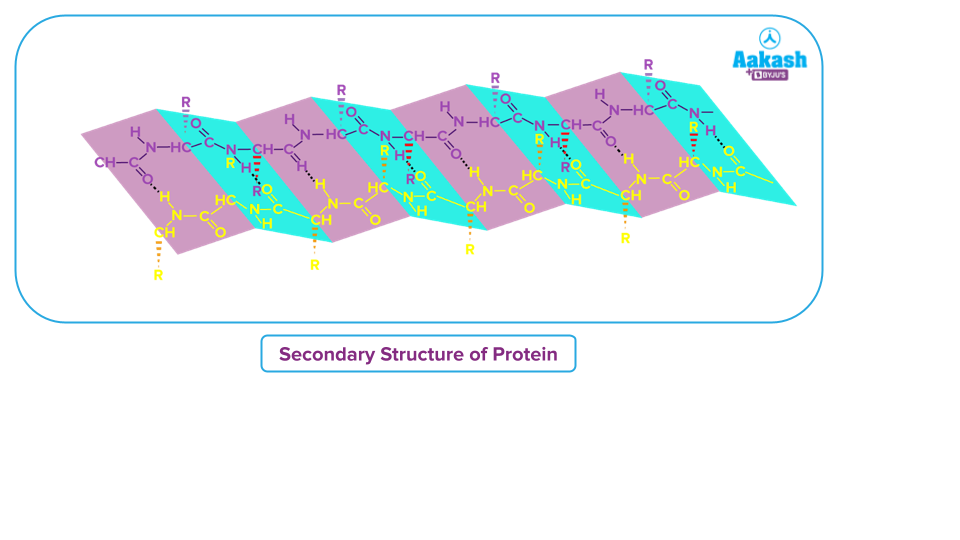
Protein Secondary Structure
Refers to extremely stable amino acid residue configurations that result in repeating structural patterns. It is created by the non-covalent interaction of amino acids, which results in the folding of a polypeptide. Peptide bonds and hydrogen bonds are the only interactions that keep a protein's secondary structure together. Protein secondary structure can be divided into two types:
- Alpha Helix
- Beta-pleated sheet
Tertiary Structure of the Protein
Proteins has a three-dimensional structure because the long polypeptide chain folds like a hollow woollen ball. This is referred to as protein tertiary structure. Peptide bonds, hydrogen bonds, and disulfide bonds are the interactions that makeup proteins' tertiary structure. The tertiary structure of proteins is required for many of their biological functions.
Quaternary Structure of the Protein
It is made up of two or more polypeptide chains. Each polypeptide chain is referred to as a subunit. Weak Van der Waal forces hold these polypeptide chains together. These polypeptides or subunits are arranged in a specific way with respect to one another to form a protein's quaternary structure.

Classification of Proteins
It can be classified into two types based on their molecular structure
- Fibrous proteins: Fibrous proteins are formed when polypeptide chains run parallel to each other and form a fibre-like structure. These proteins are found in animals and are insoluble in water. Keratin and myosin contain this type of protein. Hydrogen and disulfide bonds hold fibrous protein chains together.
- Globular proteins: A globular protein is formed when a folded polypeptide chain forms a spheroidal shape. These proteins, unlike fibrous proteins, are water-soluble. Insulin and albumins contain this type of protein.
On the basis of the nature of molecules, protein is classified into two types:
- Acidic proteins: acidic proteins exist as anions and contain acidic amino acids, such as blood groups.
- Basic proteins: basic proteins exist as cations and are high in basic amino acids such as lysine and arginine.
Based on the composition, protein is classified into two types:
- Simple proteins: These proteins are made up entirely of amino acids. Albumins, globulins, prolamins, and so on.
- Conjugated Proteins: These are complex proteins that have a non-amino acid substance called a prosthetic group attached to them. These are classified as follows:
Nucleoproteins are a protein-nucleic acid hybrid.
Mucoproteins are a combination of proteins and carbohydrates (>4%).
Glycoproteins are a combination of proteins and carbohydrates (4%).
Chromoproteins are proteins that have been combined with coloured pigments.
Lipoproteins are a protein-lipid complex.
Metalloprotein: A protein that has been combined with metal ions.
Phosphoprotein: A protein-phosphate group complex.
Sources of Protein
Proteins are the basic building blocks of our bodies. They are abundant in almost all foods, including fruits and vegetables. Protein-rich foods include eggs, milk, cheese, butter, yoghurt, chicken, soybeans, fish, paneer, beans, pulses, and meat. Although there are debates about the intake of carbohydrates and fats in order to maintain proper health, a doctor's first recommendation is always a minimum amount of daily protein intake.

Importance of Protein
- Enzymes are proteins that are responsible for the majority of the chemical reactions that occur within a cell. Protein aid in the regeneration and creation of DNA molecules, as well as the execution of complex processes.
- Hormones are proteins that aid in the balancing of the body's components. Hormones such as insulin, which aids in blood sugar regulation, and secretin are examples. It is also involved in digestion and the production of digestive juices.
- An antibody also referred to as an immunoglobulin, is a type of protein that the immune system primarily uses to repair and heal the body from foreign bacteria. They frequently collaborate with other immune cells to identify and separate antigens from increasing until they are completely destroyed by white blood cells.
- Proteins are a major source of energy that aids in our body's movements. It is critical to consume the proper amount of protein in order to convert it into energy. When consumed in excess, protein is converted into fat and becomes a component of fat cells.
Tests for Protein
Proteins are long chain polymers with a high molecular mass that are made up of -amino acids and contain carbon, hydrogen, nitrogen, oxygen, and sometimes phosphorus and sulphur.
The simple tests performed to determine the presence of proteins in the given food samples are as follows.
- Biuret test: This is a tes for the presence of compounds with peptide-links like proteins. The test solution is made alkaline with sodium or potassium hydroxide solution and treated with few drops of aqueous copper sulphate. Proteins presence is indicated by the change of the blue colour of copper supahte to violet colour.
- Xanthoproteic test: Proteins turn yellow or orange when exposed to concentrated nitric acid. Proteins reacts with nitrica cid and form a yellow precipitate Protein presence can be further confirmed by the changes of yellow colour to orange on treatment with a base.
- Millions test: Tyrosine's phenolic group in proteins reacts with mercuric sulphate, sodium nitrite and sulfuric acid.to produce a red colour.
Proteins on treatment with millions of reagents, initially produces a white precipitate. Which on heating changes to a brick-red colour upon boiling,.
- Ninhydrin test: Proteins react with a pyridine solution torm variety of colours from blue to red. However, this ninhydrin solution has to be prepared fresh as it is unstable.ac.
Denaturation of Protein
pH and temperature affect the structural forces to make the protein lose its structure, which is responsible for the activity. H-bonds are disrupted, globules unfold, helix uncoils as a result of temperature and pH changes, and proteins lose their biological activity, which can be defined as the process of protein denaturation.
Only secondary and tertiary structures are destroyed during denaturation, while primary structures remain intact.
In some proteins, denaturation is irreversible, and the protein is permanently destroyed. Other proteins' denaturation can be reversed in some cases by removing the denaturing agent, a process known as renaturation.
A few examples of denatured proteins are-
- Some common examples of protein denaturation include the coagulation of egg white when an egg is heated. Denaturation occurs due to temperature changes in this case.
- The Curdling of milk is another example of protein denaturation caused by the formation of lactic acid by microbial action.
Amino Acids
Amino acids are chemical substances that combine to form proteins, which is why they are referred to as protein building blocks. An amino acid is an organic molecule composed of a basic amino group (NH2) an acidic carboxyl group (COOH), and a unique organic R group (or side chain). These biomolecules play a variety of biological and chemical functions in the human body and are necessary for growth and development. Nature contains approximately 300 amino acids.
The human body can synthesize some amino acids out of a total of 20 amino acids known as non-essential amino acids which include aspartic acid, glutamic acid, alanine, asparagine, proline, glycine, arginine, cysteine, glutamine, serine, and tyrosine are all examples of amino acids.
Aside from these, ten additional amino acids are required because our bodies are unable to synthesize them and are known as essential amino acids. Isoleucine, phenylalanine, tryptophan, histidine, lysine, leucine, methionine, threonine, and valine are all important amino acids.
We know that amino acids are the building blocks of proteins, which play a critical role in almost all biological activities. As a result, in order to maintain a healthy and proper body function, we must include all nine essential amino acids in our daily diet. A lack of amino acids can cause a variety of pathological illnesses, including
- Oedema.
- Anemia.
- Insomnia.
- Diarrhea.
- Depression.
- Hypoglycemia.
- Appetite Deficiency
- Fat accumulates in the liver.
- Skin and hair issues
Some of the symptoms include headaches, weakness, irritability, and exhaustion.
Practice Problems
Q1. Which colour change takes place when nitric acid is added to the protein?
- It produces a coloured solution that ranges from deep blue to violet-pink to red
- The presence of proteins is confirmed by the appearance of a yellow-coloured solution.
- The appearance of a brick red colour confirms the presence of proteins
- The presence of Proteins is confirmed by the formation of violet colouration.
Answer: (B)
Solution: In the xanthoproteic test, proteins turn yellow or orange when exposed to nitric acid. For nitration, concentrated nitric acid is used. Proteins produce a yellow precipitate when exposed to nitric acid, which turns orange when exposed to alkali. Therefore option (B) is correct.
Q2. Select the correct option with respect to the denaturation of protein.
- Only secondary and tertiary structures are destroyed during denaturation, while primary structures remain intact.
- Only tertiary are destroyed during denaturation, while primary structures and secondary structures remain intact.
- Only primary are destroyed during denaturation, while secondary structures and tertiary structures remain intact.
- Primary, secondary and tertiary structures are destroyed during denaturation.
Answer: (A)
Solution: H-bonds are disrupted, globules unfold, helix uncoils as a result of temperature and pH changes, and proteins lose their biological activity, which can be defined as the process of protein denaturation. Only secondary and tertiary structures are destroyed during denaturation, while primary structures remain intact. Therefore option (A) is correct.
Q3. Which among the given option is an example of essential amino acid?
- Arginine
- Aspartic acid
- glutamic acid
- Methionine
Answer: (D)
Solution: Ten additional amino acids are required externally from food sources because our bodies are unable to synthesize them and are known as essential amino acids. Important amino acids include isoleucine, histidine, lysine, leucine, phenylalanine, tryptophan, methionine, threonine, and valine. Whereas Arginine, Aspartic acid and glutamic acid is a non-essential amino acid produced by the body. Therefore option (D) is correct.
Q4. Select the correct option with respect to the structure of the protein.
- Peptide bonds, hydrogen bonds, and disulfide bonds are the interactions that makeup proteins' quaternary structure.
- Peptide bonds and hydrogen bonds are the only interactions that keep a protein's primary structure together.
- The primary structure of proteins is a linear chain of amino acid residues linked together by peptide bonds
- All the above option is correct
Answer: (C)
Solution: The primary structure of proteins is a linear chain of amino acid residues linked together by peptide bonds Peptide bonds and hydrogen bonds are the only interactions that keep a protein's secondary structure together. Peptide bonds, hydrogen bonds, and disulfide bonds are the interactions that makeup proteins' tertiary structure. Therefore, option (C) is correct.
Frequently Asked Questions-FAQs
Q1. What is the difference between denaturation and renaturation of protein?
Answer: H-bonds are disrupted, globules unfold, helix uncoils as a result of temperature and pH changes, and proteins lose their biological activity, which can be defined as the process of protein denaturation.
In some proteins, denaturation is irreversible, and the protein is permanently destroyed. Other proteins' denaturation can be reversed in some cases by removing the denaturing agent, a process known as renaturation.
Q2. What is the difference between the xanthoproteic test and the million’s test?
Answer: In the xanthoproteic test, proteins turn yellow or orange when exposed to nitric acid. For nitration, concentrated nitric acid is used. Proteins produce a yellow precipitate when exposed to nitric acid, which turns orange when exposed to alkali. Whereas, in the case of the Millon test In the presence of sodium nitrite and sulfuric acid, Tyrosine's phenolic group in proteins reacts with mercuric sulphate to produce a red colour. Proteins containing phenolic amino acids perform Millon's test. In the presence of sodium nitrite and sulfuric acid, Tyrosine's phenolic group in proteins reacts with mercuric sulphate. to produce a red colour. Proteins containing phenolic amino acids perform Millon's test.
Q3. List some important functions of amino acids.
Answer: Some important function of amino acids includes:
- Glycine aids in the maintenance of proper cell growth and function, as well as the healing of wounds. It performs the function of a neurotransmitter.
- Valine is a key component in promoting muscle growth.
- Threonine aids in the promotion of immune system functions.
- Lysine is required for the formation of antibodies, hormones, and enzymes, as well as for the development and fixation of calcium in bones.
- Aspartic acid is essential for metabolism and the synthesis of other amino acids.
Q4. What is the difference between essential amino acids and non-essential amino acids? Give some examples.
Answer: Ten additional amino acids are required externally from food sources because our bodies are unable to synthesise them and are known as essential amino acids. Important amino acids include isoleucine, histidine, lysine, leucine, phenylalanine, tryptophan, methionine, threonine, and valine. Whereas Amino acid which is synthesised by the body is known as a non-essential amino acid. For example-Arginine, Aspartic, glutamic acid etc. is a non-essential amino acid produced by the body.


