-
Call Now
1800-102-2727
Resonance Energy - Resonance, Resonance Energy, Examples, Practice Problems and FAQ
Aloha is a fourth-grade kid studying in a School. One fine day, her science teacher took the grade four kids to a smart class and showed them how a rhinoceros has the features of both a unicorn and a dinosaur to make the concept interesting. Post-school time, Aloha reaches her home and explains to her parents how a rhinoceros is a combination of a unicorn and a dinosaur. She says that the rhinoceros has the distinct features of two mythological creatures and can be considered a hybrid of the two.

![]()
Similarly in chemistry, there are compounds (rhinoceros) that have properties (features) that cannot be explained using a single structure but need multiple structures (unicorn and dinosaur) that either does not exist or have a very little short life. These structures that explain the properties of a compound are called the resonance structures and the culmination of all the structures is called the resonance hybrid. The energy difference between the most stable resonance structure and the resonance hybrid is called resonance energy.
We shall learn more about resonance energy and examples of it in this article.
Table of Contents
- Resonance and Resonance Energy
- Examples of Resonance and Resonance Energy
- Determination of Resonance Energy
- Practice Problems
- Frequently Asked Questions-FAQs
Resonance and Resonance Energy
The atoms in molecules are supposed to attain the stable eight electron configuration by adopting electron rearrangement to form any one of ionic or covalent, coordinate or hybridisation bonding. But, the above bondings individually could not explain the properties of some molecules or ions with unsaturation and necessitates the presence of multiple bondings or multiple structures for the same molecule or ions.
Delocalization of pi-electrons can result in structures called canonical structures contributing to the various properties of the molecule.
Canonical structures satisfy the following conditions.
i) All the structures should have the structures satisfying Lewis distribution of electrons.
ii) All bonded atoms should be the same in all structures.
iii) All atoms involved in pi-bonding should be in the same plane
iv) Number of bonding and non-bonding electrons must be the same
v) All have similar energy
Structures pertaining to all these conditions are called resonance or canonical structures. All these resonance structures contribute to the properties of the molecule such that the real structure is a hybrid of all these resonance structures.
Resonance structures have fractional bonding due to delocalisation extending over multiple atoms in the molecule.
The phenomenon of contribution by such multiple structures to a single molecule's properties is called resonance.
The difference in energy between the actual (resonance hybrid) or experimental determination of the molecule to the most stable (or lowest energy) structure of any one of the structures is called resonance energy.
Resonance energy/enthalpy = Experimental enthalpy - theoretical enthalpy of the most stable structure
Examples of Resonance and Resonance Energy
1. Carbonate ions can be represented by three canonical structures.
Lewis structure satisfying the octet configuration of carbon gives three similar structures where two oxygen atoms have one negative charge each and are connected by a single bond to carbon, while the third oxygen has a double bond. All of them do not represent the real structure. The resonance hybrid from these three structures has delocalized electrons over all the atoms with fractional charges of -23 on each oxygen atom with the O-C bond order being between one and two. The resonance hybrid of carbonate ion is-
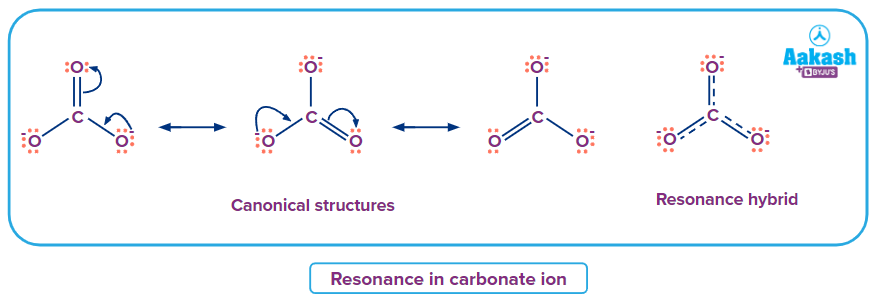
The energy difference between this resonance hybrid structure and any of the canonical structures is the resonance energy of carbonate ion.
2. Benzene molecule contains three double bonds in conjugation within the ring structure. The molecule is planar because of the conjugated pi-bonds. The structure of benzene can be written in two equal non-ionic structures. But the true structure will be a combination of these two. The canonical and hybrid structures of benzene can be represented as
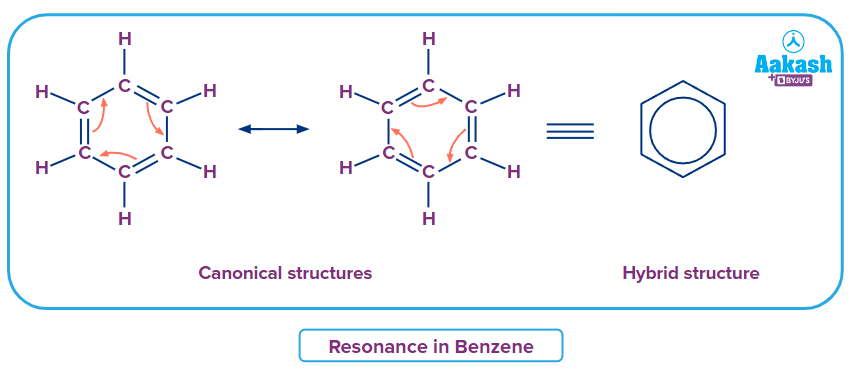
The difference in the energies of the resonance hybrid and the similar canonical structure is the resonance energy of benzene.
Determination of Resonance Energy:
Resonance energy is calculated as the difference between the experimental energy of the formation of the compound to the theoretically calculated energy of the formation of the same compound from the available data.
Resonance energy = Experimental heat of formation - Theoretical heat of formation.
Where, fHexp0 is the standard enthalpy of formation determined by experiment, which can be taken as that of the resonance hybrid,
fHtheo0 is the standard enthalpy of formation calculated from the available experimental values of the most stable canonical structures.
Resonance energy also can be calculated from the differences in the energy of conversion of the given conjugated system and theoretical non-conjugated molecule of the same molecular formula.
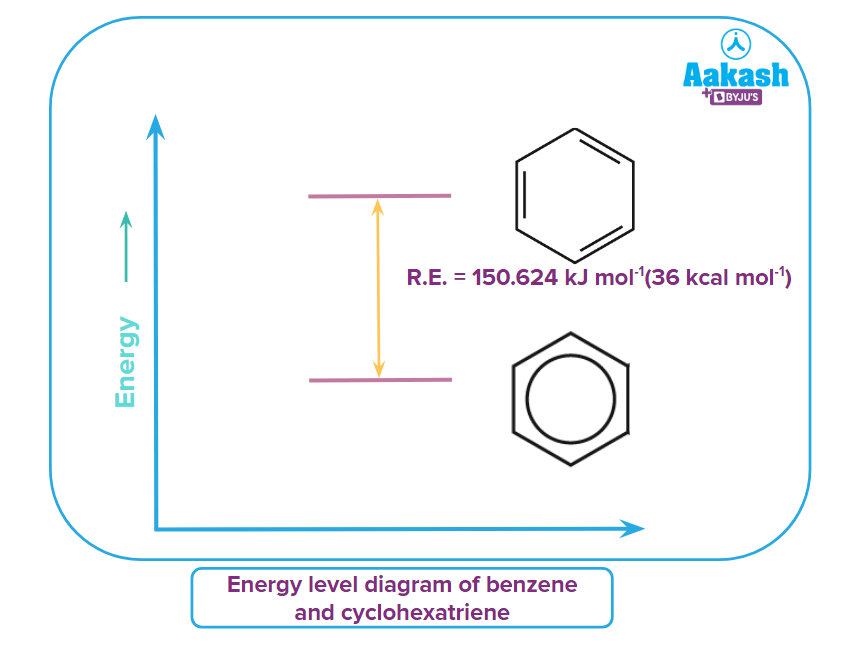
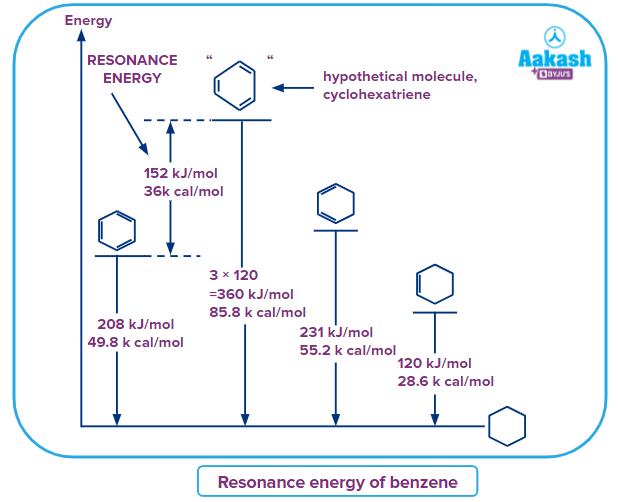
The resonance energy of benzene can be calculated from the enthalpy of hydrogenation of the conjugated benzene and a theoretical non-conjugated cyclohexatriene.
Benzene is fully conjugated and delocalised within the hexacyclic ring. The conjugation gives extra stabilisation energy called resonance energy compared to an unconjugated molecule. Hydrogenation of benzene with hydrogen to saturated cyclohexane is an exothermic reaction with 208kJmol-1 per mole and can be experimentally determined.
The unconjugated molecule with the same formula will be cyclohexatriene(imaginary) and also can be considered to be hydrogenated to the same saturated cyclohexane. The difference in enthalpy of hydrogenation between the benzene and cyclohexatriene will give the resonance energy of benzene.
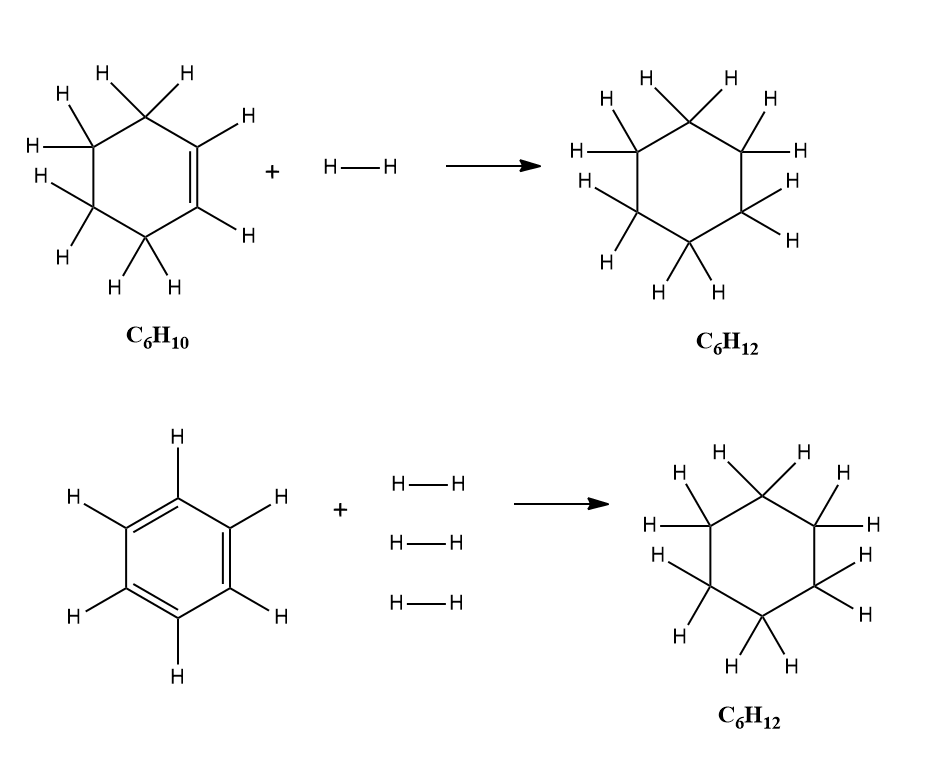
Since the cyclohexatriene is non-existence, ht energy of the formation of cyclohexatriene can be theoretically calculated from the available data as follows. Cyclohexatriene has three double bonds. So, the enthalpy of hydrogenation in an unconjugated molecule will be three times the enthalpy of hydrogenation of a single double bond, say in cyclohexene.
Enthalpy of hydrogenation of cyclohexene is 120kJmol-1. So, theoretically, the enthalpy of hydrogenation of three double bonds in nonconjugated cyclohexene will be
3 X 120 = 360kJmol-1.
The resonance energy of benzene = 360 - 208 = 152kJmol-1.
Resonating structure | CHEMISTRY | JEE | Concept of the Day | SM Sir - YouTube
Resonance - Chemical Bonding | Applying Concepts | JEE 2023 | Chemistry - YouTube
Practice Problems
Q1. The standard enthalpy of formation estimated experimentally and theoretically from the enthalpy of formation is 390kJmol-1 and 435kJmol-1 respectively. Calculate the resonance stabilisation energy of the molecule.
a. 390 kJ mol-1
b. -45kJ mol-1
c. 825kJ mol-1
d. 435kJmol-1
Answer: B
Solution: Given,
HHexp0=390kJ mol-1
HHtheo0 =435kJ mol-1
Standard resonance energy =
Jmol-1
Q2. Resonance energy will be?
a. Negative
b. Positive
c. Zero
d. Positive or negative
Answer: A
Solution:
Resonance energy is the additional stabilisation energy the molecule gains through the delocalisation of electrons through the conjugation. This energy will hence be liberated to make the change exothermic with enthalpy having a negative sign.
Q3. The most stable canonical structure will be
a. Neutral without charges
b. Opposite charges on the nearest neighbour atoms
c. Opposite charges separated as much as possible
d. All
Answer: Option A
Canonical structures can be neutral or with charges. But the presence of charges makes them unstable making them prone to reaction. Neutral canonical structures are the most stable.
Q4. Resonance structures are
a. More than one
b. Have the same number of paired and nonpairing electrons
c. Have almost similar energy
d. All the above
Answer: Option D
Resonance or canonical structures are more than one having similar structures involving the nucleus and bonding.
Frequently Asked Questions(FAQs)
Q1. What is the relation between the canonical structures?
Answer: Canonical structures have the nucleus and the same number of bonding and non-bonding electrons. They differ in the delocalization of electrons only and have the relatively same energy. Higher the number of canonical structures higher the stability of the molecule.
Q2. What is the difference between the canonical structures and the resonance hybrid?
Answer: Canonical structures are imaginary structures of the molecule, while the resonance hybrid is a combination of this structure and is close to the real structure to explain the properties of the molecule.
Q3. What causes the resonance in a molecule?
Answer: Resonance is caused by the delocalisation of pi-electrons in the molecules. Higher the conjugation, the higher the stabilisation.
Q4. How resonance and aromaticity are related?
Answer: Aromatic compounds also possess resonance. Aromaticity is an extension of the resonance phenomenon in a cyclic ring with (4n + 2) number of pi-electrons present in a planar structure.


