-
Call Now
1800-102-2727
Refining of Metals From Concentrated Ores - Distillation, Bessemerisation, Zone Refining, Liquation, Poling, Electrolytic Refining, Chromatography, Practice Problems, FAQs
Consider you are given an ore of a metal having impurities of other metals like
In order to separate metal from these impurities you are given two choices:

First is that you choose to treat the metal such that other impurities are collected together and pure metal is obtained.
Secondly you choose to treat all the impurities one by one and then after all the impurities will be removed we will be left with pure metal.
Which way would you like to extract the metal? First or second? The metal extracted from its ore is free from dust and other impurities. Now the metal contains impurities of other metals. So it will be better if we employ those methods that will give us pure metal. Since all the metals present as impurities have specific reactivity, therefore if you choose to treat all the impurities separately, it would increase the cost of extraction of metal. So the easier and cost effective way to get the pure metal is to treat the metal which is present in major portions.
Let's study more about such methods!
Table of contents:
- Introduction
- Distillation
- Bessemerisation
- Zone refining
- Liquation
- Poling
- Electrolytic Refining
- Vapour Phase refining
- Chromatography
- Practice problems
- Frequently Asked Questions-FAQs
Introduction:
A metal extracted by any method is usually contaminated with some impurities. To remove these impurities or obtain a metal of high purity, several techniques are required. Various techniques used are given as follows:
Distillation:
It is very useful for metals with low boiling points like An Impure metal is evaporated to obtain a pure metal (as distillate).

Bessemerisation:
In this method air is blown into the molten metal mat which is deposited in the Bessemer converter. In the final stage of smelting, other products like are oxidised and removed as slag
![]() .
.

Example: Sulphide of copper is converted to its corresponding oxide in an air blast. obtained reacts with the remaining molten
.
Zone refining:
It is based on the principle that impurities are more soluble in the molten state than in the solid metal. A circular mobile heater is fixed at one end of a rod of impure metal. The molten zone moves along with the heater that moves forward. As the heater moves, pure metal crystallises out of the melt. The impurities pass on into the adjacent molten zone. This process is repeated several times and the heater is moved in the same direction. The end at which impurities get concentrated is cut off.
Example: Semiconductors like Ge, Si, B, Ga, In, etc. and other metals.
Liquation: Liquation is used for metals with low melting points. Metal is made to flow on the sloping surface such that it separates from impurities with higher melting points.

Poling:
Electrolytic Refining:
Anode consists of impure metal and cathode consist of a strip of the same metal in its pure form. Both the electrodes are dipped in a suitable electrolytic bath containing soluble salt of the same metal.
Oxidation half cell reaction occurring at the anode:
Metal, M should be more basic.
Reduction half cell reaction occurring at the cathode:
![]()
Metal, M should be less basic.
Example: Consider electrolytic refining of copper, such that impure copper is taken as anode and pure copper strips are taken as a cathode, both dipped in acidified solution of
Reaction at the anode:
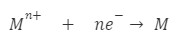
Reaction at the cathode:
![]()
Impurities from blister copper deposit as anode mud below the anode which contains metals like Se, Te, Ag, Au, Pt. These metals can be recovered to meet the cost of refining.
Other metals like zinc can also be obtained by this process.
Vapour Phase refining:
Chromatography:
In chromatography, a column of is prepared in a glass tube. If the moving medium containing a solution of components is in liquid form, it is referred to as column chromatography. Column chromatography is very useful for the purification of elements that are available in minute quantities and impurities that are not very different in chemical properties from the element to be separated. Several chromatographic techniques, such as paper, column, gas, etc., are used for the purification of metals.
Practice Problems:
Q1. The correct statement is:
A). Zone refining is used for the refining of titanium
B). Zone refining is used for the refining semiconductors like
C). Both of these
D). None of these
Answer: B
Semiconductors like Ge, Si, B, Ga, In, etc. and other metals can be purified using zone refining.
Q2. The refining method used when the metal and the impurities have low and high melting temperatures, respectively is:
A). Liquation
B). Vapour phase refining
C). Zone refining
D). Distillation
Answer: A
The liquation method is used when the melting point of metal is less in comparison to the melting point of the associated impurity.
Q3. The Mond's process is used for the:
A). Purification of
B). Extraction of
C). Purification of and
D). Extraction of
Answer: A
The Mond's process is used for the purification of nickel.
Q4. Which of the following statements is true regarding electrolytic refining?
A). Anode consists of impure metal
B). Anode consists of pure metal
C). Cathode consists of a strip of the same metal in its pure form
D). Both A and C
Answer: D
Anode consists of impure metal and cathode consists of a strip of the same metal in its pure form. Both the electrodes are dipped in a suitable electrolytic bath containing soluble salt of the same metal.
Frequently Asked Questions-FAQs:
Q1. What other metals can be refined using electrolytic refining?
Answer: In electrolytic refining anode consists of impure metal and cathode consists of a strip of the same metal in its pure form. Both the electrodes are dipped in a suitable electrolytic bath containing soluble salt of the same metal. Metals like can be purified usingelectrolytic refining.
Q2. What is anode mud?
Answer: Insoluble impurities present along with the metal get deposited at the anode, which is known as anode mud. Example- During electrolytic refining of lead, metals like are deposited as anode mud.
Q3. What is the principle on which chromatography is based?
Answer: Chromatography is based on the principle of adsorption. Different components of a mixture are adsorbed on an adsorbent in different ways. Several chromatographic techniques, such as paper, column, gas, etc., are used for the purification of metals.
Q4. What is the principle on which liquation is based?
Answer: Liquation is based on the difference between metal and impurity melting points. This procedure is used to purify metals that have a lower melting point than the impurities they contain. It can be used to purify impure tin metal, impure zinc (spelter), for removing from
metal.
Related topics:
| Extraction of Crude Metal from concentrated Ore | Extraction of iron |
| Occurrence of Metals | Thermodynamical principle of metallurgy |
| Concentration of ores | Ellingham Diagram |


