-
Call Now
1800-102-2727
Reduction of Isocyanides, Imines, Imides: Mendius Reduction, Gabriel Phthalimide Synthesis, Practice Problems and FAQs:
Ishita, a student in class 12th had a mild fever, so her parents decided to take her to the hospital. The doctor examined her and prescribed paracetamol for recovery. While returning back to home, unfortunately, the tires of their car got punctured. Such a bad day for her. Her father fixed the puncher and they headed back home safely. As Ishita is a curious child, she immediately searched the composition of tires and paracetamol.

One thing that was common in both was amine. So she immediately started learning about preparation, properties and other useful application of amines.
Table of content
- Definition of Amine
- Methods of Preparation of Amines
- From Nitriles and Isonitriles
- Reduction of Isocyanides or Carbylamines
- Practice Problems
- Frequently Asked Questions
Definition of Amine
Compound, the amine is a product of ammonia (NH3). In other words, amines are products of ammonia. Amines are organic nitrogen compounds with a nitrogen atom in a lone pair. The hydrogen atoms of ammonia are typically swapped out for an aryl or alkyl group in amines.
Methods of Preparation of Amines
Amines are typically made using the following techniques:
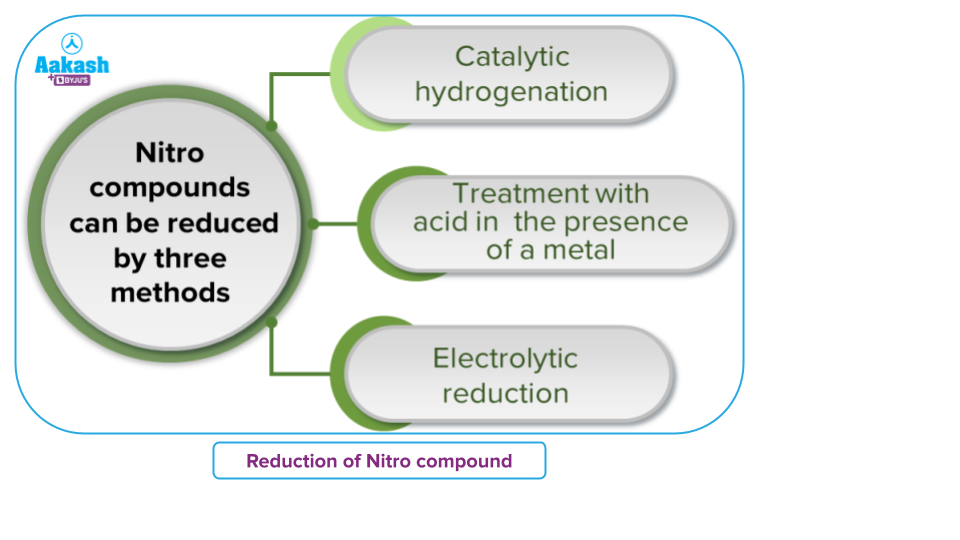
Reduction of Nitro Compounds
By reducing the corresponding nitro compounds, one can quickly prepare both aliphatic and aromatic primary amines. There are several ways to achieve this reduction.:
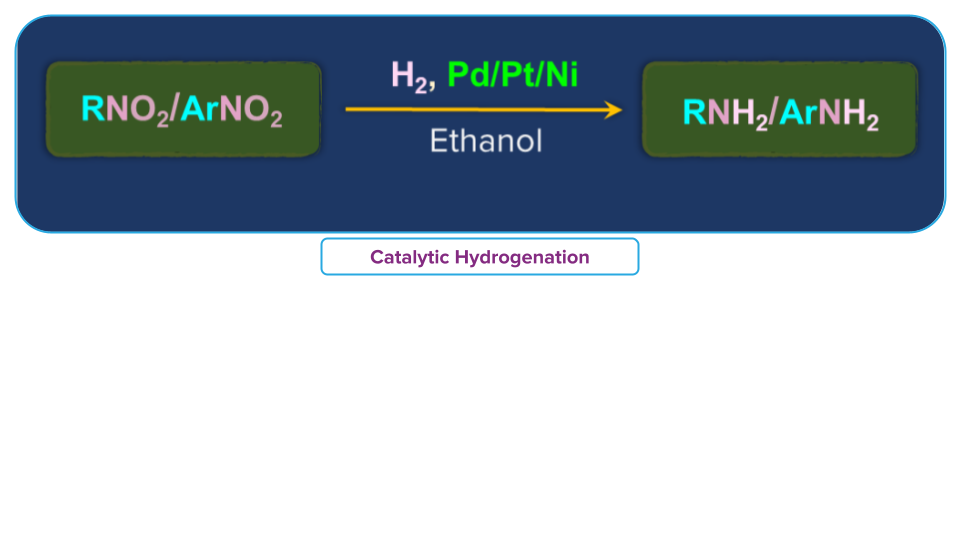
- Catalytic Reduction.
By, passing hydrogen through the nitro-substrate in the presence of a catalyst made of finely divided in the presence of Raney Ni at room temperature amines can be obtained
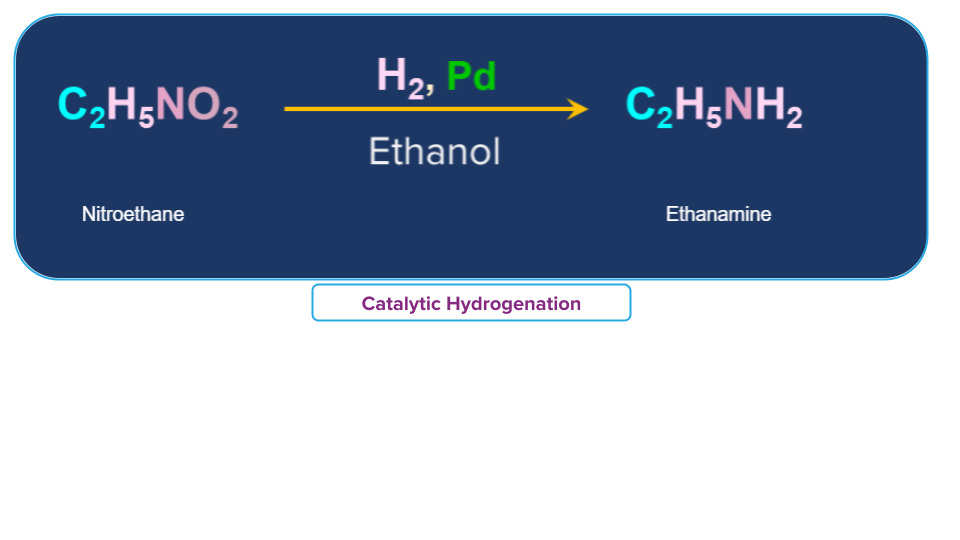
Example:
Nitro ethane can be converted to ethanamine in presence of H2, Pd and ethanol.
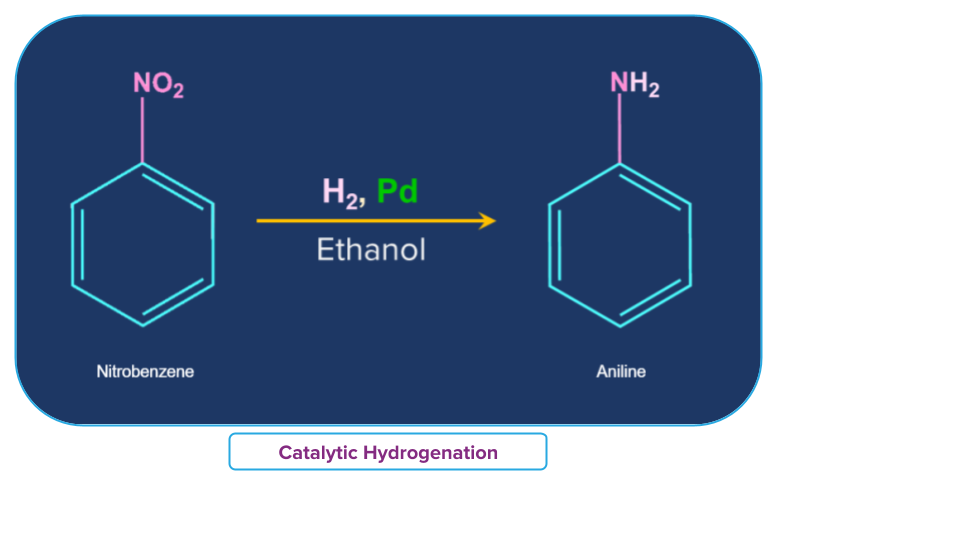
Nitro benzene can be converted to aniline in presence of H2, Pd and ethanol.
Treatment with Acid in Presence of a Metal
In the presence of concentrated hydrochloric acid and an active metal like Sn, or Zn, nitroalkane is transformed into a primary amine.
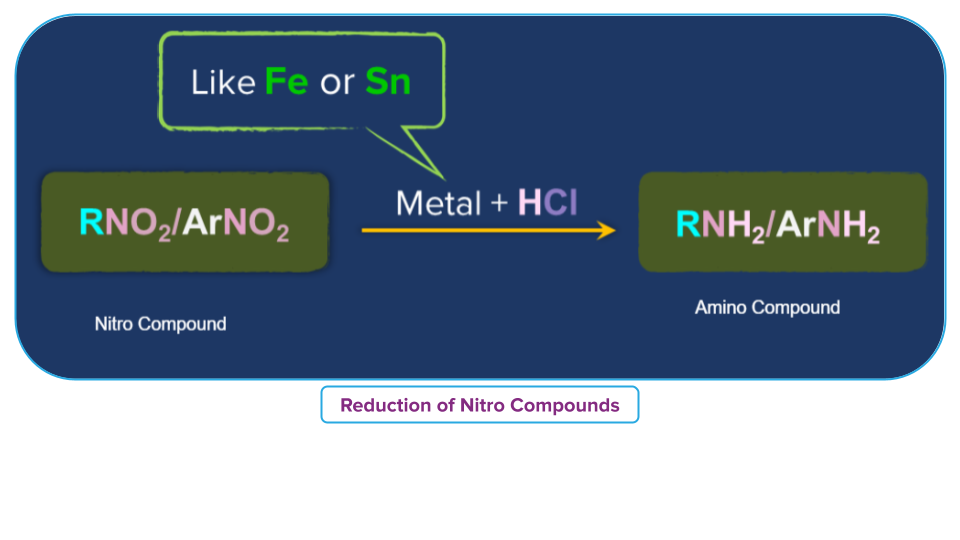
For example,
- Nitrobenzene in the presence of a metal such as tin or iron and with concentrated HCl reduces to aniline.
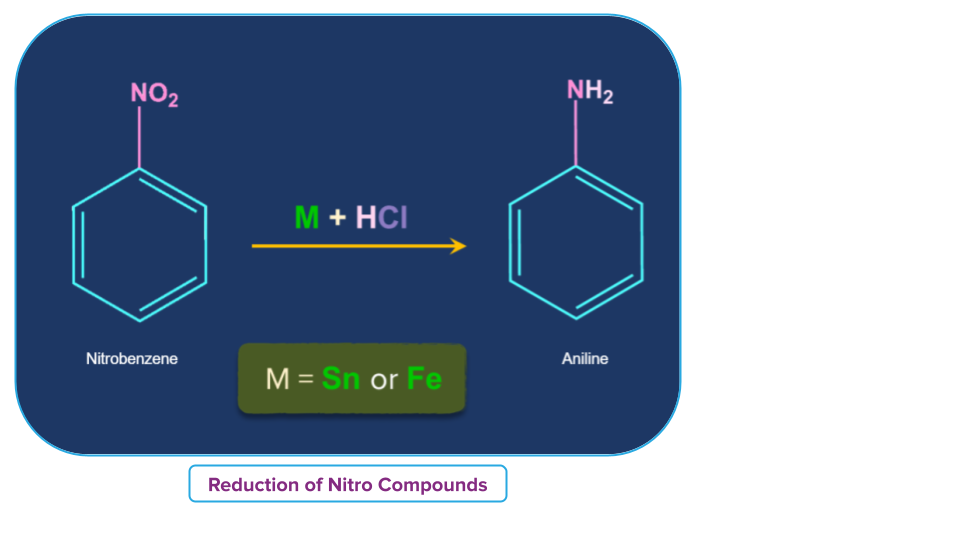
Conc.HCl and SnCl2 combined can also be used for reduction. Since FeCl2 formed during the reaction is hydrolysed to release hydrochloric acid, the reduction with Fe/HCl method is preferred to . As a result, only a small quantity of hydrochloric acid is needed to start the reaction.
Since primary aromatic amines cannot be made from the corresponding aryl halides on treatment with ammonia, one of the most practical ways to make them is through the reduction of nitro compounds to primary amines. The nitration of arenes is a simple method for producing the necessary nitro compounds.
With Lithium Aluminium Hydride (LiAlH4)
Nitroalkanes can be easily reduced to the corresponding primary amines with LiAlH4. In contrast, aromatic nitro compounds on reduction with LiAlH4 give azo compounds and not primary amines.
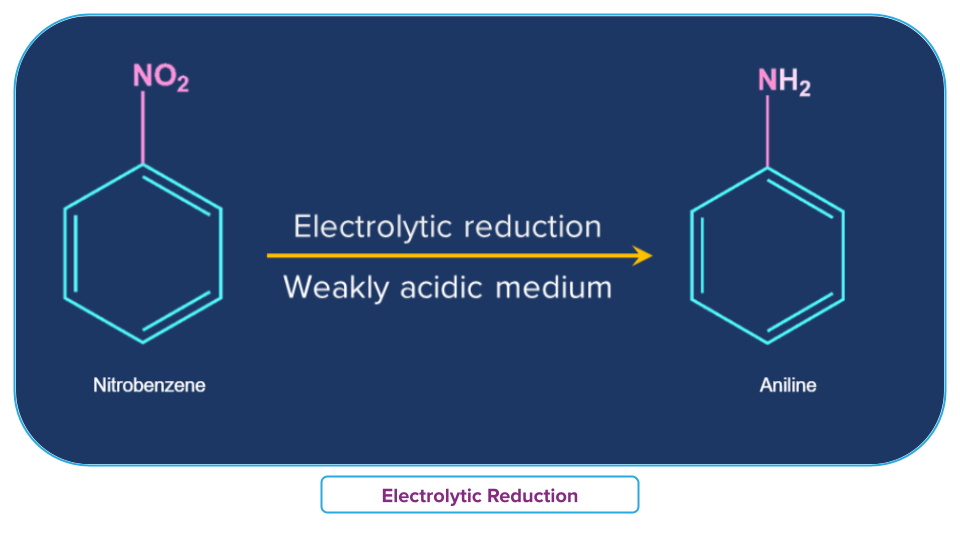
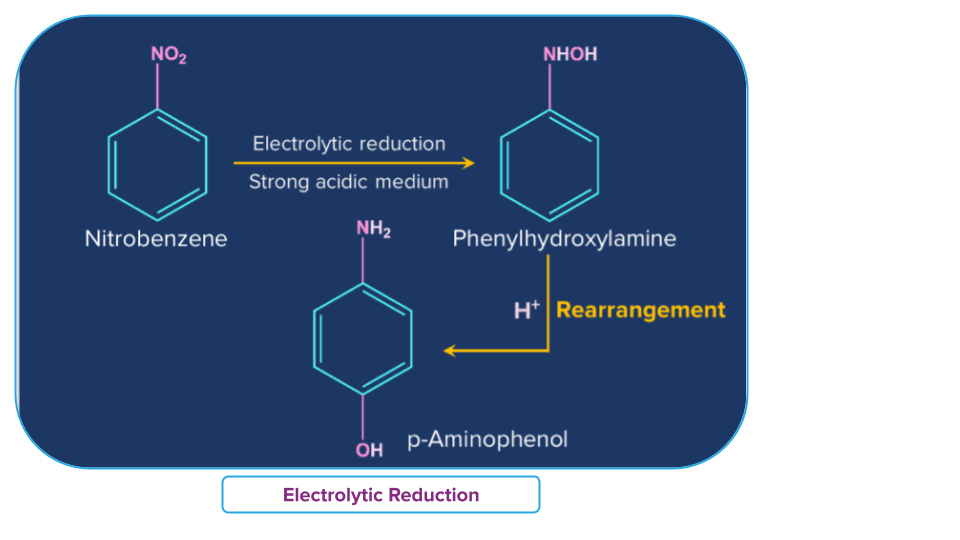
Electrolytic Reduction
In a weakly acidic medium, electrolytic reduction of nitrobenzene produces aniline, whereas, in a strongly acidic medium, it produces p-aminophenol. Azoxybenzene and azobenzene are two examples of the many mono- and di-nuclear reduction products that can be made in an alkaline medium.
From Nitriles
By the Reduction of Nitriles (or Cyanides)-
Aliphatic and aralkyl primary amines can be easily prepared by the reduction of the corresponding nitriles either catalytically with H2 and Raney nickel or chemically with lithium aluminium hydride or sodium and alcohol or sodium amalgam and alcohol (reduction with sodium and alcohol is called Mendius reduction).
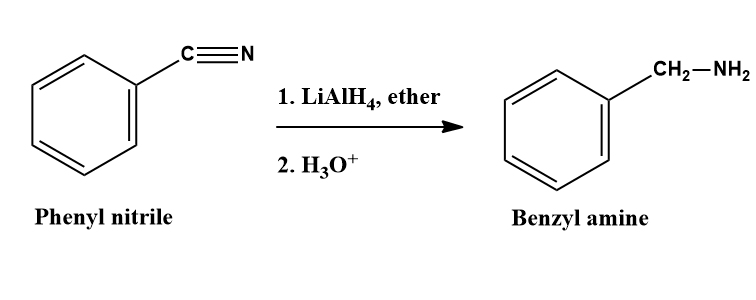
Since alkyl cyanides can be easily prepared by the action of alcoholic NaCN or KCN on alkyl halides, this reaction gives us an excellent method for converting alkyl halides into primary amines having one carbon atom more than the parent alkyl halide.
Reduction of Isocyanides or Carbylamines
Reduction of isocyanides or carbylamines gives secondary amines i.e., N- methylamines.
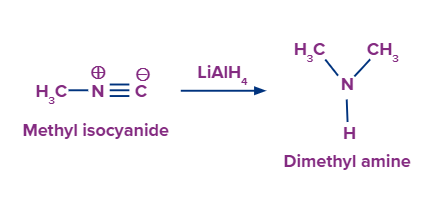
This method can be used only for the preparation of 2° amines in which one of the alkyl groups is always methyl.
Hydrolysis of Isocyanides or Carbylamines
When isocyanides or carbarylamines are hydrolyzed with diluted mineral acids (but not with alkalies), formic acid and primary amines (containing one less carbon than the initial isocyanide) are always produced.
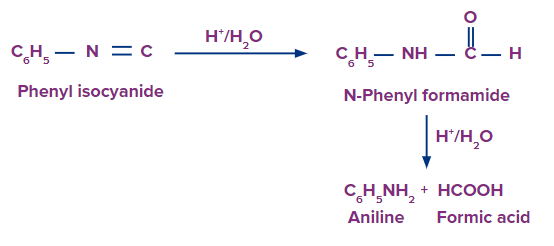
Isocyanides have a negative charge on the carbon atom, which at first attracts electrophiles like H+ but repels nucleophiles (OH- ion). As a result, alkalies cannot hydrolyze isocyanides; only acids can. The ability of the negatively charged carbon atom to attract a nucleophile increases once a proton is attached to it due to the presence of a positive charge on the nitrogen atom, which facilitates hydrolysis as shown below:
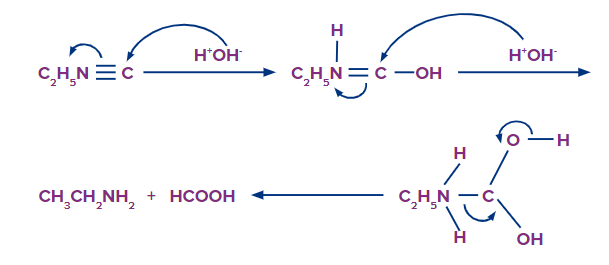
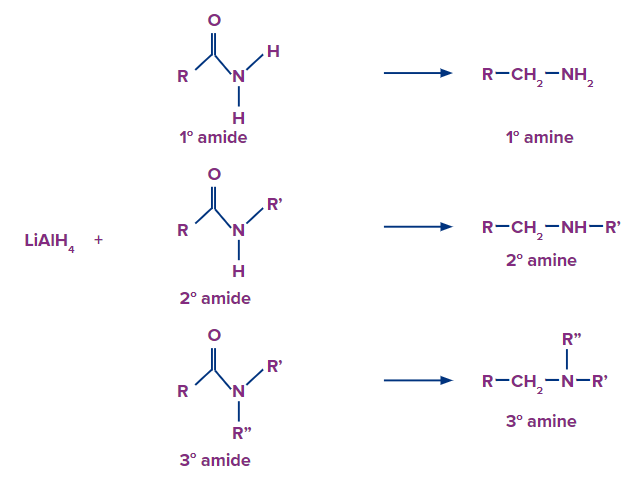
Reduction of Amides
Primary, secondary and tertiary amines can be prepared by the reduction of the corresponding amides with lithium aluminium hydride (LiAlH4). Secondary and tertiary amines can be prepared by the reduction of secondary and tertiary amides respectively, For example,
Gabriel Phthalimide Reaction
This is a very practical process for making pure aliphatic and aralkyl primary amines. Potassium phthalimide is produced when ethanolic KOH reacts with Phthalimide, which may then be heated with the appropriate alkyl or aralkyl halide to produce N-substituted phthalimides. These yield primary amines when further hydrolyzed with dilute HCl under pressure or with alkali.
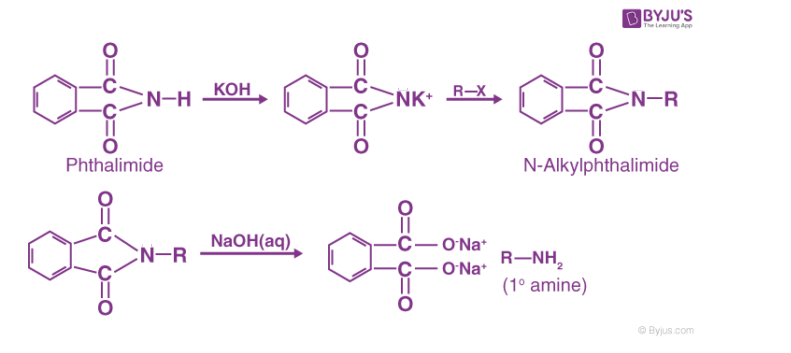
- The compound phthalimide can be produced from phthalic acid once more and used repeatedly. Ethyl chloroacetate can be used in place of ethyl iodide in the reaction described above to create benzylamine, which can then be produced using benzyl chloride and glycine (NH2CH2COOH).
- This method cannot be used to make aromatic 1° amines like aniline, toluidines, etc. because, under mild conditions, ary1 halides do not undergo a nucleophilic substitution reaction with potassium phthalimide.
- N-alkylphthalimides typically hydrolyze slowly in an acidic or basic environment. In order to obtain 1° amine using the Gabriel synthesis, hydrazinolysis of N-alkylphthalimides is a more practical and effective method. For instance,
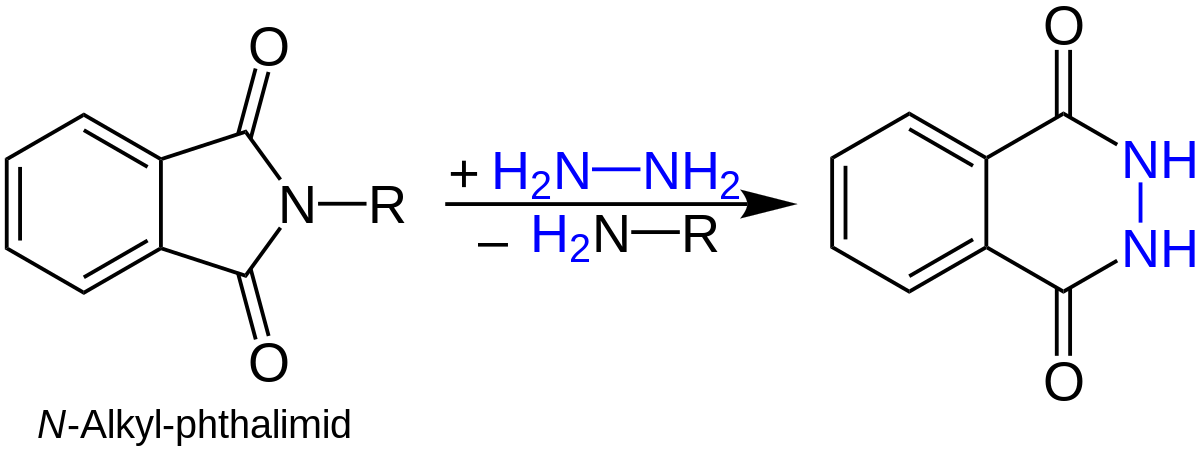
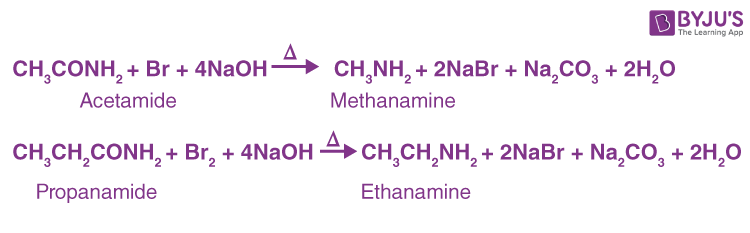
By Hofmann's Degradation of Primary Amides. (Hofmann Bromamide Reaction)-
A primary amide can be converted into a primary amine with one fewer carbon atom by reacting it with an aqueous or ethanolic solution of potassium hydroxide and bromine (or potassium hypobromite, KOBr). :
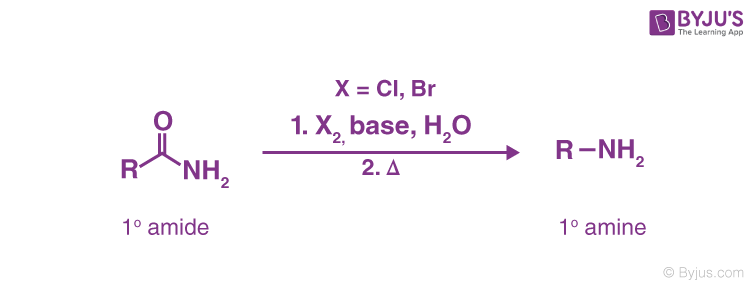
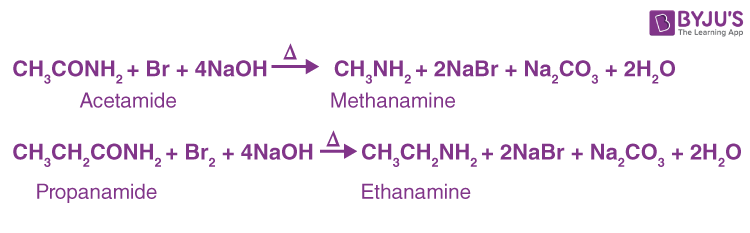
This reaction is frequently used for homologous series descent or stepping down.
By Reductive Amination of Aldehydes and Ketones,
Aldehydes and ketones react with ammonia in presence of a reducing agent such as Raney Ni or NaBH3CN (sodium cyanoborohydride) to form primary amines. The reaction, in fact, occurs through the intermediate formation of an imine. For example,
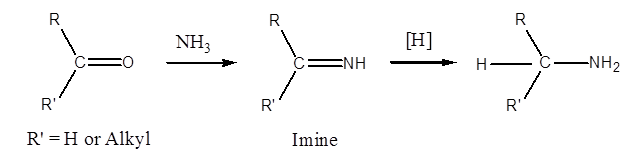
Reductive amination is the name of the reaction that occurs when an aldehyde or ketone is treated with ammonia in the presence of a reducing agent, such as , and results in the conversion of the two compounds into the corresponding amine. in place of NH3. In the reaction described above, a primary amine is used, and a secondary amine is produced. For instance,
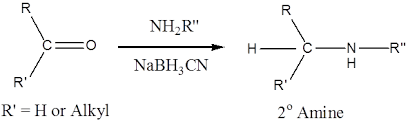
Practice Problems
Q1. The most convenient method to prepare an amine-containing one carbon atom less is
A. Gabriel phthalimide synthesis
B. reductive amination of aldehydes
C. Hofmann bromamide reaction
D. reduction of isonitriles
Answer: (C)
Solution: The Hofmann bromamide reaction is the most effective way to get one carbon atom less amine. When a primary amide is treated with an aqueous or ethanolic solution of potassium hydroxide and bromine (or potassium hypobromite, KOBr), it gives a primary amine that has one carbon atom less than the original amide.
Q2. Amine that cannot be prepared by Gabriel phthalimide synthesis is
A. Aniline
B. methyl amine
C. Benzylamine
D. iso-butylamine
Answer: (A)
Solution: This is a very practical process for making pure aliphatic and aralkyl primary amines. Potassium phthalimide is produced when ethanolic KOH reacts with Phthalimide, which may then be heated with the appropriate alkyl or aralkyl halide to produce N-substituted phthalimides. These yield primary amines when further hydrolyzed with dilute HCl under pressure or with alkali. Because aromatic halides do not undergo nucleophilic substitution from the phthalimide salt, Gabriel phthalimide cannot be used to make aromatic amines.
Q3. Which of the following method is suitable for the preparation of amines from nitro compounds?
A. Catalytic hydrogenation
B. Electrolytic reduction
C. Treatment with acid in presence of metal
D. All of the above
Answer: (D)
Solution: Both aliphatic and aromatic primary amines can be easily prepared by the reduction of the corresponding nitro compounds. This reduction can be achieved in a number of ways such as
- Catalytic hydrogenation
- Electrolytic reduction
- Treatment with acid in presence of metal
Q4. Instead of which of the following reducing agent can be used for the better yield of amine
Answer: (B)
Solution: Reduction of nitro compounds yields amine using several reducing agents. One of the common reducing agents that we use is raney Ni, Pd or Pt. A mixture of SnCl2 and Conc. HCl has also been used for the reduction of aromatic nitro compounds. Reduction with iron metal and hydrochloric acid forms ferrous chloride along with hydrogen. FeCl2 formed undergoes hydrolysis to form HCl so that HCl required for the entire reaction will be small..
Frequently asked questions (FAQs)
Q1. Why can't we reduce nitro compounds using Conc. HCl rather than conc. sulphuric acid?
Answer: In contrast to hydrochloric acid, sulfuric acid is an oxidising agent. As a result, conc. HCl is employed to decrease nitro compounds rather than Conc. sulphuric acid.
Q2. How basic or acidic are nitro compounds?
Answer: Acidic character: The -hydrogen linked to the carbon atom bearing the nitro group is acidic and can be readily extracted by a base due to the strong electron-withdrawing tendency of the nitro group (-I effect). Resonance stabilizes the resultant anion, which is frequently referred to as the nitronate ion.
Q3. Are amine and amino interchangeable terms?
Answer: An amino group is a functional group in chemistry that consists of one bond connecting a nitrogen atom to a hydrogen atom, an alkyl group, an aryl group, or a combination of these three groups. A chemical substance with an amino group is known as an amine.
Q4. Which are the common compounds made up of amines?
Answer: Amine functional groups are found in a wide variety of substances, including natural and synthetic colours, polymers, vitamins, and medications like penicillin and codeine. Additionally, they are present in a number of chemicals necessary for life, such as DNA, amino acids, hormones, and neurotransmitters.


