-
Call Now
1800-102-2727
Redox Reactions in Electrodes – Redox Reactions, Electrodes, Redox Reactions in Cells, Practice Problems and FAQ
Metals have a significant impact on our lives and they have to be refined from their natural forms. For instance, copper is used in electrical wiring in our homes and in many ornaments.

Do you know what the best method is to refine copper?
Pure copper is deposited at the cathode as a result of a redox reaction that occurs when an electric current is transmitted through the solution.
We will learn more about the redox processes that occur in the electrodes during electrolysis on this concept page.
TABLE OF CONTENTS
- Redox Reactions
- Electrodes
- Types of Electrodes
- Redox Reactions in Electrochemical Cells
- Redox Reaction in Electrolytic Cells
- Practice Problems
- Frequently Asked Questions – FAQ
Redox Reactions
A redox reaction is a chemical reaction in which electrons are exchanged between two distinct reactants. It is possible to determine the transfer of electrons between the relevant reactants by looking at the change in oxidation states of both the reacting species.
A reduction-oxidation or redox reaction is a chemical process in which both reduction and oxidation take place at the same time. While the reduced species gains electrons, the oxidised species loses electrons.
Let us take an example to understand this concept better.

The oxidation half-reaction and the reduction half-reaction are the two parts of every redox process. Each of these half-reactions must balance when stated separately in order for the number of electrons in each to be equal.
In this oxidation half-reaction, the oxidation state of lead changes from 0 in Pb(s) to +2 in PbSO4(aq). Since the oxidation state of lead increases, it has undergone oxidation i.e. Pb (s) acts as a reducing agent.
Similarly, the oxidation state of copper changes from +2 in CuSO4(aq) to 0 in Cu(s). Since the oxidation state of copper decreases, it has undergone reduction. i.e. CuSO4(aq) acts as an oxidising agent.

Electrodes
A form of electrical conductor known as an electrode comes into touch with non-metallic circuit elements such as an electrolyte and gas. Depending on their capacity to transfer or receive electrons in an electrochemical cell, electrodes can be referred to as either an anode or a cathode.
The anode is an electrode where oxidation takes place after electrons leave the cell and enter the circuit.
The cathode is an electrode where reduction takes place after electrons enter the cell and leave the circuit.
Because of its ability to function as either an anode or a cathode depending on its electrode potential with respect to the other electrode, a metal electrode is not permanently fixed as either an anode or a cathode.
Types of Electrodes
Electrodes are categorised into the following types according to their composition.
- Metal – Metal Soluble Salt Electrode
- Gas – Ion Electrode
- Metal – Metal Insoluble Salt Electrode
- Calomel Electrode
Metal – Metal Soluble Salt Electrode
This is the most fundamental kind of electrode, where each metal is immersed in a solution of its soluble salt. A pure metal (M) in contact with its cation solution (Mn+) makes up these electrodes.
For example, a copper rod dissolved in copper sulphate. The symbol for the copper-copper ion electrode is Cu|Cu2+.
Half–cell reaction at the anode:
Half–cell reaction at the cathode:
Gas – Ion Electrode
A gas – ion electrode is the one that involves the exchange of electrons between an ion and a gaseous species. Example: Standard hydrogen electrode, (SHE).

Half–cell reaction at the anode:
Half–cell reaction at the cathode:
Metal – Metal Insoluble Salt Electrode
They contain a metal (M) that is enclosed in a thin layer of the metal salt (MX), which is sparingly soluble, and immersed in a solution of common ions (X-). It is depicted as an anion electrode or an anode.
These are composed of a silver metal (Ag) that is enclosed by a thin layer of silver chloride salt (AgCl), which is sparingly soluble, and immersed in a solution of common ions (Cl-) in ionic electrolyte such as KCl.
The standard reduction potential of the cation in an electrolyte should be lower than that of metal salts.

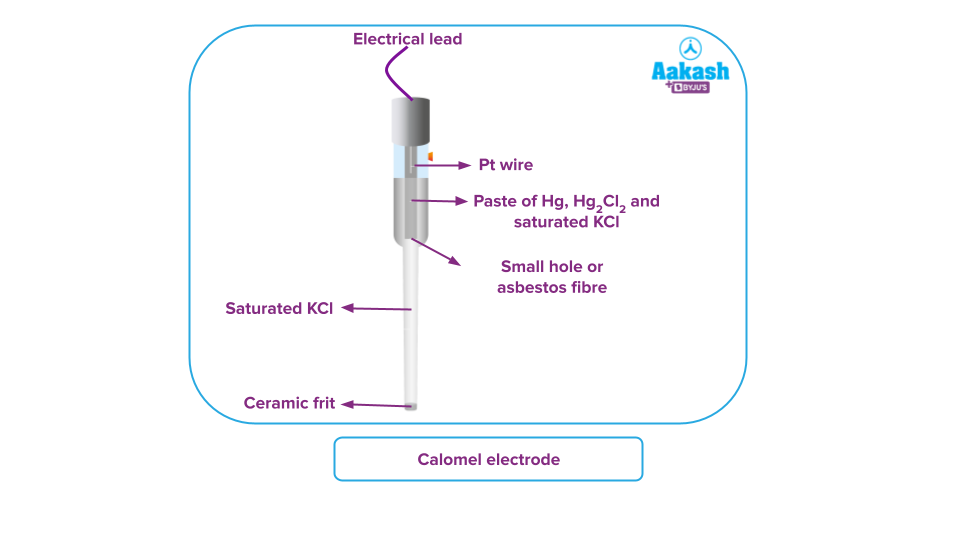
Calomel Electrode
The name of this electrode is derived from mercury chloride. Where SHE can not be used, calomel electrodes are employed as a standard electrode. Here, mercury in contact with its salt mercury chloride serves as the electrode. These calomel reference electrodes are used in chemical laboratories with voltmeters and pH metres.
An outstanding non-polarizable calomel electrode is commonly used in two-electrode systems with a supporting electrolyte that is a non-reactive chloride salt because of its exceptional durability. Calomel electrode consists of two glass tubes. The electrode is housed inside the inner tube, and the mercuric chloride used as the electrolyte is disseminated in an aqueous KCl solution.
The inner and outer liquids are electrically connected by the outer tube.
Half–cell reaction:
……………(i)
…………………(ii)
Combining equations (i) and (ii),
Redox Reactions in Electrochemical Cell
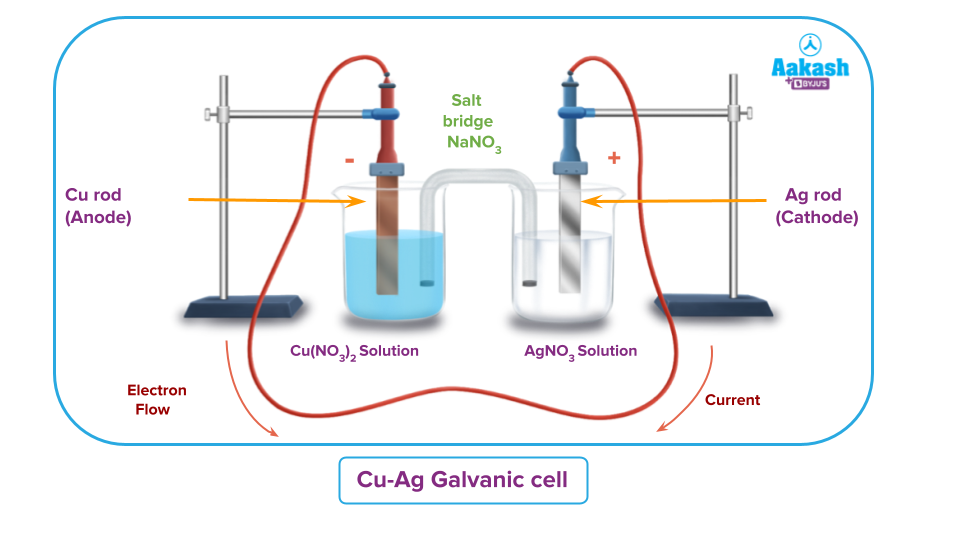
Redox Reaction Between Copper and Silver Nitrate
An electrochemical cell is a device that converts chemical energy produced in a redox reaction into electrical energy.
If a redox reaction is allowed to occur in such a way that the oxidation half-reaction occurs in one beaker and the reduction half-reaction occurs in another, the electrons given out by the former are taken by the latter and a current flows. As a result, the cell comprises two half-reactions or two half-cells.
The two half-reactions will be:
(Oxidation half-reaction)
(Reduction half-reaction)
The anode is the copper electrode where oxidation occurs. Because reduction occurs at the silver electrode, it serves as the cathode. The normal current flow is from silver to copper as opposed to electron flow from copper to silver.
The overall reaction is the result of combining the two half-reactions and gives you a redox reaction.
The reaction is
Or it may be written as
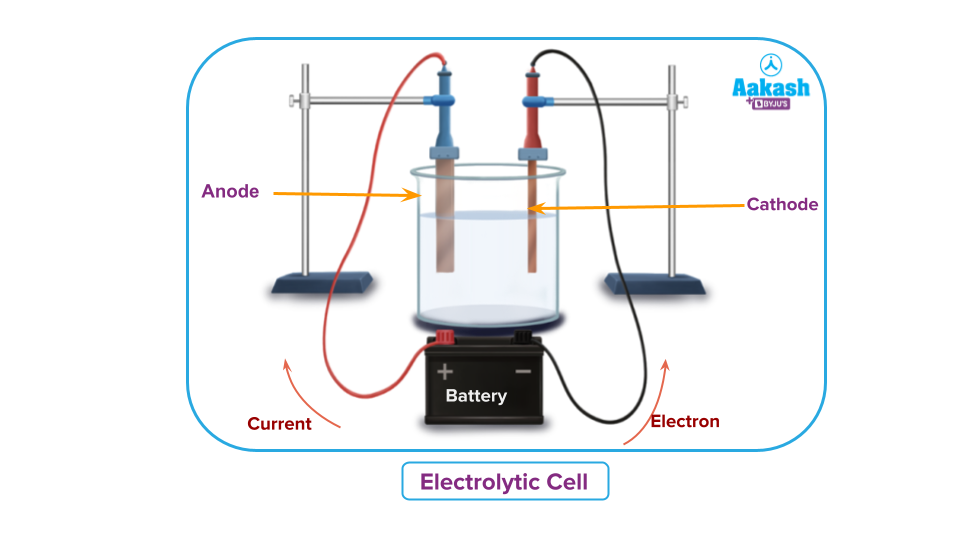
Redox Reaction in Electrolytic Cells
An electrolytic cell is used to cause a non-occurring redox reaction. If we connect a battery between two electrode half-cells and apply a voltage equal to or slightly greater than the cell EMF, the reaction occurs in the opposite direction of a galvanic cell, i.e. non-spontaneous redox reaction becomes feasible.
As a result, in an electrolyte cell, electrical energy is employed to generate chemical energy.
For example, molten sodium chloride (NaCl) can be electrolysed into its pure constituents of sodium and chlorine gas using an electrolytic cell.
When an electric current passes through the cathode, it becomes rich in electrons and attracts sodium cations. At the cathode, the sodium ions accept an electron and are neutralised, resulting in metallic sodium.
The anode attracts the chloride ions, which release their electrons in the circuit. Two of these neutral chlorine atoms combine to form a chlorine molecule, which then escapes at the anode.
Anode:
Cathode:
Overall Redox reaction:
Hence, Molten sodium chloride can be electrolysed in an electrolytic cell to create metallic sodium and chlorine gas as a product.
Recommended Videos
ElectroChemistry Class 12 Chemistry (Chapter-3): Important Topics for JEE Main | JEE 2022 Exam Prep
Practice Problems
1. Pick out the redox reaction among the given reactions.
a.
b.
c. N
d. All of these
Answer: A
In the reaction given above, the oxidation state of iron changes from +2 in FeO to 0 in Fe(s). Iron has undergone reduction because its oxidation state decreases from +2 to 0. Similarly, the oxidation state of hydrogen changes from 0 in H2(g) to +1 in H2O(aq). Hydrogen has undergone oxidation since its oxidation state has increased from 0 to +1.
As oxidation and reduction are taking place simultaneously in this reaction, it is a redox reaction.
In the reaction given in option B, the oxidation states of the elements remain the same. Therefore, it is not a redox reaction.
In the reaction given in option C, the oxidation states of the elements remain the same. Therefore, it is also not a redox reaction.
So, option A is the correct answer.
2. Select the correct statement about redox reactions.
a. Oxidation is the removal of an electropositive element.
b. Not all decomposition reactions are redox reactions.
c. Reduction is the addition of an electropositive element.
d. All of the above
Answer: D
- The removal of an electropositive element is referred to as oxidation.
(oxidation of iodine: (-10))
Thus, the statement given in option A is correct.
- All decomposition reactions are not redox reactions.
For example, the decomposition of sodium bicarbonate into sodium carbonate, water and carbon dioxide is not a redox reaction.
Since the oxidation states of the elements remain the same, it is not a redox reaction.
Thus, the statement given in option B is correct.
- The addition of an electropositive element is referred to as reduction.
(Reduction of oxygen :(0-2))
Thus, the statement given in option C is correct.
So, option D is the correct answer
3. Which of the following statements is correct about Redox reactions in cell?
a. In an electrochemical cell, there is the occurrence of a non-spontaneous redox reaction and electrical energy is converted into chemical energy.
b. In an electrochemical cell, there is the occurrence of a spontaneous redox reaction and chemical energy is converted into electrical energy.
c. In an electrolytic cell, there is the occurrence of a non-spontaneous redox reaction and chemical energy is converted into electrical energy.
d. In an electrolytic cell, there is the occurrence of a spontaneous redox reaction and electrical energy is converted into chemical energy.
Answer: B
Solution: In an electrochemical cell, there is the occurrence of a spontaneous redox reaction and chemical energy is converted into electrical energy. In an electrolytic cell, there is the occurrence of a non-spontaneous redox reaction and electrical energy is converted into chemical energy.
So, option B is the correct answer.
4. Which of the following statements is correct about electrodes?
a. Oxidation occurs at anode and reduction occurs at cathode.
b. Oxidation occurs at cathode and reduction occurs at anode.
c. Oxidation and reduction both occurs at cathode.
d. Oxidation and reduction both occurs at anode.
Answer: A
Solution: An electrode may be referred to as either a cathode or an anode depending on whether it has the ability to transfer or receive electrons. Oxidation occurs at the anode and reduction occurs at the cathode.
For example, in the electrolysis of molten sodium chloride (NaCl), when an electric current passes through the cathode, it becomes rich in electrons and attracts sodium cations. At the cathode, the sodium ions accept an electron and are neutralised, resulting in metallic sodium.
The anode attracts the chloride ions, which release their electrons in the circuit. Two of these neutral chlorine atoms combine to form a chlorine molecule, which then escapes at the anode.
Anode:
Cathode:
So, option A is the correct answer.
Frequently Asked Questions – FAQ
1. Can a platinum electrode be used as a reference electrode?
Answer: The findings suggest that Pt can be utilised as a reference electrode in some circumstances where conventional reference electrodes cannot. The application of conventional reference electrodes such as the calomel electrode, is constrained.
2. In a redox process, how do electrons flow between elements?
Answer: Reducing agents are also known as electron donors, whereas oxidising agents are known as electron acceptors. Electrons transfer from a set of orbitals on the electron donor called the donor orbitals to a set of electron shells on the recipient called the acceptor orbitals in an electrochemical reaction.
3. What role does redox reaction have in human lives?
Answer: Cellular respiration, the ultimate source of energy in humans, consists of a sequence of redox processes. As a result, the food we eat is only turned into energy through redox reactions. Carbon dioxide is decreased during aerobic respiration, while water is oxidised to generate oxygen.
4. Is iron rusting a redox reaction?
Answer: An illustration of a redox or oxidation-reduction reaction is the rusting of iron. Iron is used to combine with oxygen in the presence of water in the rusting process. In this instance, oxygen serves as the oxidising agent and the metal iron acts as a reducing agent.


