-
Call Now
1800-102-2727
Real Gas - Deviation of Gas From Ideal Behaviour, Compressibility Factor & Boyle’s Temperature
Do you think if you throw a ball in the air and it will never come back? Well! It is possible under certain conditions if you can throw a ball with an escape velocity that the ball leaves the earth’s gravity and escaped out. Now, this can happen only under certain conditions, which practically is impossible for a human to throw a ball with such a force. So, what we are talking about is a hypothetical scenario. In a real scenario, a thrown ball will surely come back due to earth’s gravity. Similarly, if we talk about gases, it is impossible to find ideal and perfect gases, there will always be deviations from such behaviours. So, what do you think, is it possible for a gas to behave ideally under certain conditions?
Let’s discuss in detail, how gases behave under different conditions and is it even possible for a real gas to behave like an ideal gas?
Table of contents
- Deviation from ideal behaviour
- Compressibility factor
- Modifications of van der Waals equation
- Comparison of Z vs P curves of same gas at different temperatures
- Boyle’s temperature
- Practice problems
- Frequently asked questions-FAQs
Deviation from ideal behaviour
At a constant temperature and at different pressure and volume, the gas was seen to deviate from the ideal gas curve. However, no gas is ideal and almost all the gases show significant deviation from the ideal behaviour. Thus gases like H2 , N2, CO2 etc that fail to obey the ideal gas equation are termed as non-ideal or real gases.

Few assumptions in the kinetic theory of gas for ideal gases:
- Volume of gas molecules is negligibly small in comparison to the total volume of the gas (i.e., the volume of the container in which it is kept).
- There are no intermolecular interactions between the gas molecules.
Reasons for the deviation of real gases from the ideal behaviour:
- Volume of gas molecules cannot be neglected in comparison to the total volume of the gas.
- There will be intermolecular interactions between the gas molecules.
Compressibility factor
Compressibility factor determines the relationship between the molar volume of the real gas and the molar volume of the ideal gas.
At constant temperature and pressure, the ratio of the molar volume of the real gas to the molar volume of the ideal gas determines the compressibility factor.
Compressibility factor (![]()
Compressibility factor (![]()
Here, Molar volume of the gas (Vm) is:
(Vm) ![]()
As we know, the ideal gas equation is represented as PV = nRT.
Therefore, the ideal gas equation can be written as:
PVm = RT
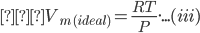
Putting the value of the equation (iii) in the equation (i), we get;
Compressibility factor ![]()
Where,
Vm represents the molar volume of the gas
P represents the pressure of the gas
T represents the temperature of the gas in kelvin scale
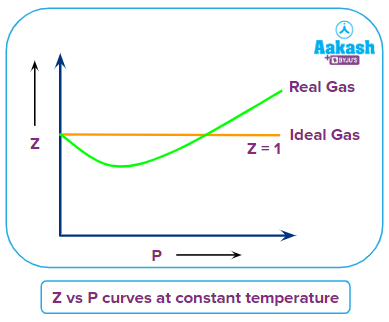
- For an ideal gas, Z=1 because PVm = RT and the compressibility factor is independent of temperature, pressure and volume. The deviation from the ideal behaviour of a gas is determined by the value of Z being greater or less than 1.
- If Z < 1, the gas shows negative deviation because the pressure of the real gas is less than the pressure of the ideal gas and non-ideality is largely due to the attractive forces between the molecules.
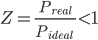
⇒Preal < Pideal
- If Z > 1, the gas shows positive deviation from the ideal behaviour because the pressure of the real gas will be more than pressure of the ideal gas and non-ideality is largely due to repulsive forces between the molecules.
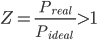
⇒Preal >Pideal
Modifications of van der Waals equation

For one mole of a gas van der Waals equation can be represented as:
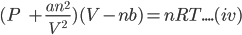
Putting the value of n=1 in the equation (iv), we get;
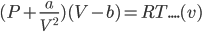
- In extremely low pressure regions: In extremely low pressure regions,
 can be neglected with respect to the pressure in the equation (v) because if the pressure is low volume occupied by the gas would be too large and
can be neglected with respect to the pressure in the equation (v) because if the pressure is low volume occupied by the gas would be too large and  factor will be too small in comparison to pressure term.
factor will be too small in comparison to pressure term.
At the same time, since the volume is very large so b can be neglected in comparison to the volume term in equation (v). Thus, at extremely low pressures, the equation (v) can be represented as:
PV=RT
Dividing the above equation with RT we get,

But we know compressibility factor (for one mole of a gas)
(for one mole of a gas)
Therefore, it can be said that:
Z = 1
So, at extremely low pressures, the gas would behave ideally. - In moderately low pressure regions: In moderately low pressure regions,
 cannot be neglected with respect to the pressure in the equation (v) because if the pressure is moderately low, the volume occupied will be high and
cannot be neglected with respect to the pressure in the equation (v) because if the pressure is moderately low, the volume occupied will be high and  will be small and
will be small and 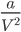 will be comparable to pressure.
will be comparable to pressure.
At the same time, since volume is large, b can be neglected with respect to volume in equation (v) i.e., (V-b) ≅ V
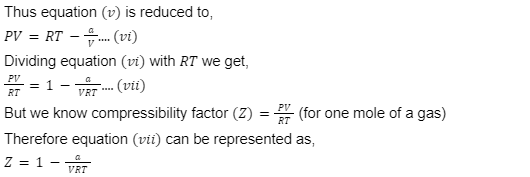
At moderately low pressure, the compressibility factor ( Z) is less than 1 and the gas shows negative deviation because the pressure of the real gas is less than the pressure of the ideal gas and non-ideality is largely due to attractive forces between the molecules. - In high-pressure regions: When the pressure is high, the volume would be small but
 term will be large and
term will be large and 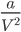 term can be neglected in the equation (v) with respect to the pressure as the repulsive force dominates in this region and molecules are brought closer by increasing the pressure.
term can be neglected in the equation (v) with respect to the pressure as the repulsive force dominates in this region and molecules are brought closer by increasing the pressure.
⇒ (P +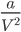 ) ≅ P
) ≅ P
At the same time volume is small and comparable to b and cannot be neglected in the equation (v),
Now, van der Waals equation is reduced to:
P(V-b) = RT
PV = RT + Pb....(viii)
Dividing equation (viii) with RT we get,
![]()
But we know compressibility factor ![]() (for one mole of a gas)
(for one mole of a gas)
Therefore equation (ix) can be represented as,
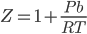
At high pressure, the compressibility factor (Z) is more than 1 and repulsive forces are
dominating between the molecules.
- At high temperature: At high temperature, the kinetic energy of the molecule will be higher and attraction force will be very low and the value of a→0 and therefore
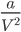 term will be zero.
term will be zero.
On the other hand if the temperature increases, the volume of the gas also increases and b can be neglected with respect to the volume of the gas. i.e., (V-b) ≅ V
Therefore equation (v) and can be reduced to,
PV = RT ..... (x)
Dividing equation (x) with RT we get,

But we know compressibility factor (for one mole of a gas)
(for one mole of a gas)
Therefore, it can be said that:
Z=1
So, at extremely high temperatures, the gas would behave ideally. - Exceptional behaviour of hydrogen and helium:
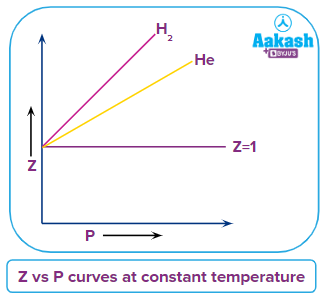
- Force of attraction

Because of low molecular mass of H2 and He forces of attraction between molecules will be small i.e., the value of van der Waals constant (a) will be small as it represents the attraction forces present between the molecules. Therefore the value of a→0 for H2 and Heand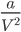 term will be zero.
term will be zero.
⇒(P +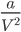 ) ≅ P
) ≅ P
van der Waals equation (v) can be represented as:
P(V - b) = RT
=> PV - Pb = RT ..... (xii)
Dividing equation (xii) with RT we get,

we know that compressibity factor (for one mole of a gas)
(for one mole of a gas)
Therefore equation (xiii) can be represented as,
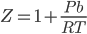
If this equation is compared with a straight line Y=mX+C, where, Y represents the Y- axis, X represents the X-axis, m represents the slope of the straight line, C represents the intercept on Y-axis.
So, Y would be Z, X would be P, C would be 1 and m would be .
.
Therefore, its a straight line on Z vs P curve and Z is always greater than 1.
Comparison of Z vs P curves of same gas at different temperatures
When the temperature is changed the nature of Z vs P curve also changes and at lower temperature the kinetic energy of the gas molecules is less due to which there are fewer collisions between the molecules and attraction forces will be dominant but when temperature increases the collision between the molecules increases and due to which repulsion forces become dominant and the dip in the curve decreases, compressibility factor increases and ease of liquefaction of gas becomes difficult.
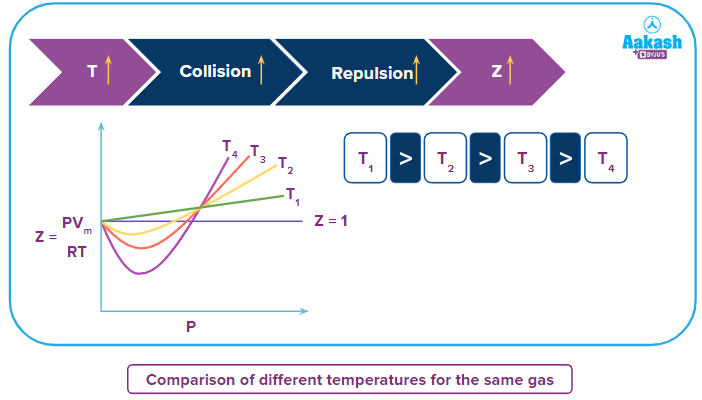
Boyle’s temperature: As the temperature is raised the dip in the curve decreases, at a certain temperature the minimum in the curve vanishes and the curve becomes horizontal for an appreciable range of pressure. At this temperature PV/RT is almost equal to unity and Boyle’s law is obeyed. Hence, this temperature of the gas is known as the Boyle’s temperature.
Mathematically boyle’s temperature of the gas can be calculated by using the virial equation of state for a real gas.
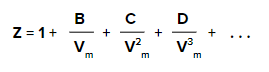
Here,
'Z', represents the compressibility factor
B, C, D represent the first, second and third virial coefficient respectively
The virial coefficient can be expressed in terms of van der Waals equation which is represented as,

Comparing the above equation with Virial equation we get,
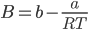
Putting the value of B in the virial equation we get,
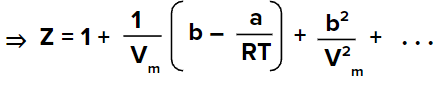
The above equation represents the decreasing series and can be approximated as,
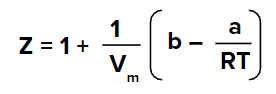
We know that for the gas to behave ideally Z=1 and considering the ideal behaviour of gas we get,
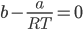
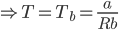
Now, this temperature is the boyle’s temperature because, at this temperature, gas will behave ideally.
Here,
Tb represents the Boyle’s temperature of the gas
“b" is the excluded volume of molecules per mole of gas
“R” represents the universal gas constant
“a” represents the proportionality constant in van der Waals gas equation
The boyle’s temperature is a characteristic property of the gas.

In the above, Z vs P curve the temperature TB represents the Boyle’s temperature over a particular range of pressure when the curve is almost horizontal and Z=1.
Recommended video
Practice problems
Q1. Select the correct option if 3.2 g of SO2 at 0℃ and 1 atm pressure occupies a volume of 2.24 L.
- The gas behaves ideally
- The gas is real with positive deviation
- The gas is real with negative deviation
- The gas may be ideal or real at a 0℃ temperature
Answer: (B)
Solution: According to the given data,
Number of moles of gas = ![]()
Temperature of gas = 0℃= 273 K
Pressure of the gas = 1 atm
Real volume of the gas (Vreal) = 2.24 L
Using the ideal gas equation PV=nRT, we get;
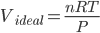
Putting the value in the equation, we get;
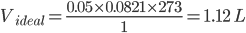
We know that,
Compressibility factor (![]()
Putting the values of Vreal and Videal.
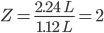
therefore, Z > 1 and it will show positive deviation from ideal behaviour.
Q2. At low pressure, the van der Waals equation for 1 mole of a gas can be represented as:
- PV = RT
- PV = RT + Pb
- P + a/V2 = RTV
- PV = RT - a/V
Answer: (C)
Solution: For one mole of a gas, van der Waals equation can be written as: ![]()
In the low pressure region, the volume will be very large so “b” can be neglected with respect to the volume of the gas and van der Waals equation takes the form:
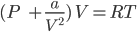
⇒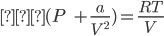
Q3. Select the correct option for van der Waals equation for 1 mole of a gas in terms of the compressibility factor and type of deviation that gas will show at extremely high pressure.
- Z = 1, No deviation from ideal gas behaviour
- Z = 1 + Pb/RT, Negative deviation from ideal gas behaviour
- Z = 1 - a/VRT, Positive deviation from ideal gas behaviour
- Z = 1 + Pb/RT, Positive deviation from ideal gas behaviour
Answer: (D)
Solution: For one mole of a gas, van der Waals equation can be written as:
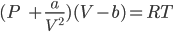
In high pressure regions:
P >> 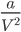
So, (P + 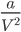 ) ≅ P
) ≅ P
Now, the van der Waals equation becomes:
P(V-b)=RT
⇒PV=RT+Pb
Dividing the above equation by RT, we get;
![]()
⇒ 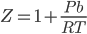
(∵ Compressibility factor ![]() )
)
From the above equation we can see that Z> 1 and if Z> 1 then the gas will show a positive deviation.
Q4. Select the correct option for the compressibility factor (Z) vs pressure (P) curve at a constant temperature.
- Horizontal line in the curve represents the non-ideal behaviour of the gas with positive deviation
- H2 will always show negative deviation at all temperatures and pressures
- H2 will always show positive deviation at all temperatures and pressures
- Deviation of a gas is dependent only on temperature and independent of pressure.
Answer: C

Solution: Horizontal line in (Z) vs (P) curve at a constant temperature represents the ideal behaviour of the gas (i.e., Z=1) . H2 gas always shows a positive deviation due to the lower molecular mass, hence, the force of attraction between the molecules will be small and results in a positive deviation of gas from the ideal behaviour.
Compressibility factor (Z) = 1+Pb/RT
Frequently asked questions-FAQs
Question 1. Why the ideal gas equation is modified to show the behaviour of real gases?
Answer: As we know that in the case of real gases the force of attraction between the molecules cannot be neglected and the volume of gas molecules can not be neglected in comparison to the volume of the container. Therefore the ideal gas equation needs to be modified by correcting the pressure and volume terms and a new equation was proposed known as the van der Waals gas equation.
Question 2. What does it indicate when the compressibility factor of gas is greater than one?
Answer: The compressibility factor of gas represents the ratio of the molar volume of real gas to the molar volume of an ideal gas. When the compressibility factor of gas is greater than one it means the molar volume of real gas will be greater than the molar volume of an ideal gas and it will show a positive deviation from the ideal behaviour at a constant temperature. Repulsion forces between the molecules of gas are dominating here.
Question 3. What will be the nature of the gas at high temperature and low pressure?
Answer: At high temperature and low pressure, the compressibility factor of a gas (Z) is one. It means that at high temperature and low pressure region the attraction forces present between the molecules is balanced by the repulsion forces and gas tends to behave ideally.
Question 4. What can be said for a gas showing negative deviation and liquefaction of gas?
Answer: If compressibility factor (Z<1) , the gas shows negative deviation and non-ideality is largely due to attractive forces between the molecules. The higher the attraction forces between the molecules more will be the ease of liquefaction of gas.
Related topics
|
Charles's Law |
Liquefaction of gases |
|
Van der Waals equation |
Dalton's Law of Partial Pressure |
|
Ideal Gas Equation |
Kinetic Theory Of Gases |


