-
Call Now
1800-102-2727
Reaction of Alkenes - Addition of Hypohalous Acids, Acid-Catalyzed Hydration, Hydroboration-Oxidation Reaction and Oxymercuration-Demercuration Reaction, Hydrogenation Reaction of Alkenes and Their Mechanisms, Addition of HX, Hydroxylation of Alkenes,
Polyethene is a typical plastic that is both functional and cost-effective. Plastic shopping bags, plastic wraps, drain pipes, milk cartons, and garbage cans are all examples of where they can be found today. It's a thermoplastic that's simple to work with and can be molded into a variety of shapes and forms, including tubing. Do you know what the initial material is?
Alkene is the answer. Polyethene is a polymer made from alkene as a starting material. Similarly, an alkene is employed as a starting component in a variety of reactions, some of which are discussed below.

Table of Contents
- Addition of Hypohalous acid to alkene- Halohydrin
- Acid-catalyzed hydration
- Hydroboration-Oxidation Reaction
- Hydration by Oxymercuration-Demercuration
- Hydrogenation reaction of alkenes
- Addition of Hydrogen Halides (HX)
- Oxidation of Alkenes-Hydroxylation of Alkene
- Practice Problems
- Frequently Asked Questions
Addition of Hypohalous acid to alkene- Halohydrin
Other halogen-containing reagents, such as hypohalous acids, HOX, are electrophilically added to an alkene to generate halohydrins. In each of these reagents, the electrophilic element is halogen. Oxygen has a stronger electronegativity than chlorine, O has a δ− charge and Cl has a δ+ charge.
Example 1: 2-Chloroethanol is formed by adding HOCl to ethene.
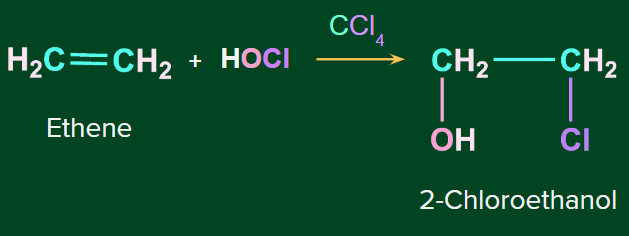
Example 2: Addition of HOCl to propene to form 1-Chloropropan-2-ol
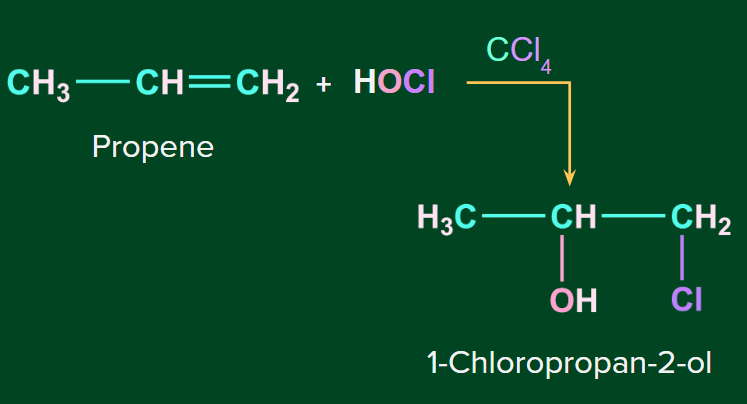
Halohydrins, on the other hand, are not generated by adding a hypohalous acid directly to an alkene; instead, the alkene is reacted with Br2 or Cl2 in the presence of water. Let us discuss in detail by mechanisms.
Mechanism
Step-1:When alkenes are halogenated, the first process is the same. When a halogen, in this case bromine, reacts with an alkene, a cyclic bromonium ion is generated.

Step-2: In step two, the bromine ion is not attacked; instead, water serves as the nucleophile. Water is more common than bromine ion, thus it's more likely to be in the right location for nucleophilic attack. Water has a better nucleophile characteristic than bromine. A nucleophile's assault probability increases as its strength grows. On more replacement carbon, the Nucleophille attack is favoured.
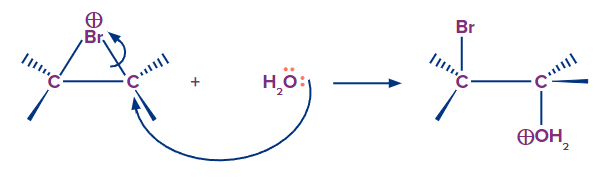
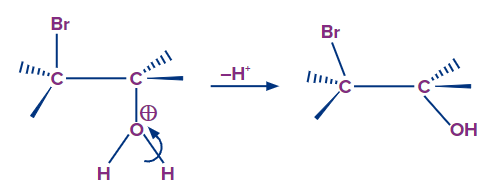
Step-3: It is the final acid-base step in which a proton is transferred from oxygen to the solvent (water), resulting in neutral halohydrin. In this case, the addition reaction produces bromohydrin.
Addition of Water
There are three methods by which water can be added to an alkene
- Acid-catalyzed Hydration
- Hyboration-Oxidation Reaction
- Oxymercuration-Demercuration Reaction
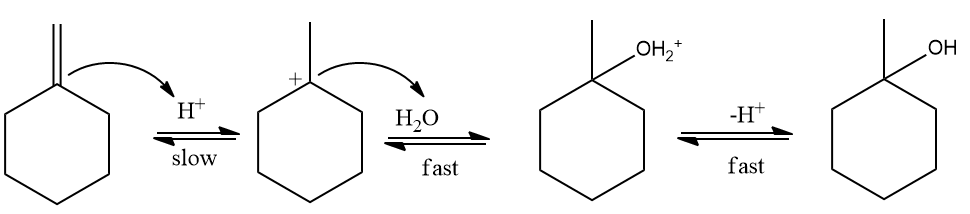
Acid catalyzed hydration:
This is also known as the hydration of alkenes. Addition of (acid catalyzed hydration) to alkenes is a Markovnikov addition reaction (Markovnikov rule states that the negative part of the addendum (adding molecule) gets attached to that carbon atom which possesses a lesser number of hydrogen atoms)
Example: Addition of H2O to isobutene
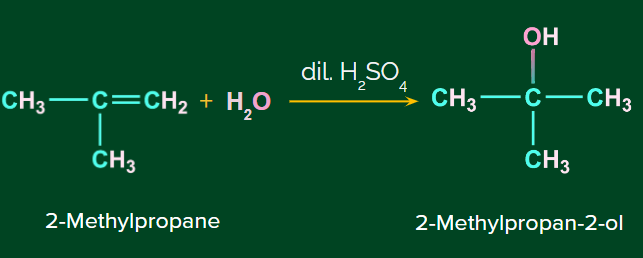
Mechanism of Acid catalyzed hydration:
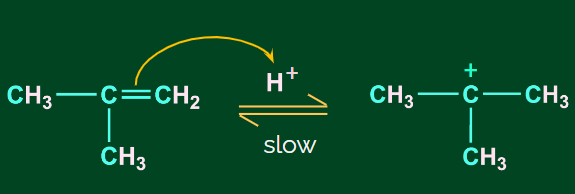
Step 1: Protonation and formation of carbocation
When the double bond breaks, it forms a stable carbocation. Here, a tertiary carbocation is formed
instead of a primary carbocation. This is because the tertiary carbocation is more stable than the
primary carbocation.
Step 2: Nucleophilic attack of H2O
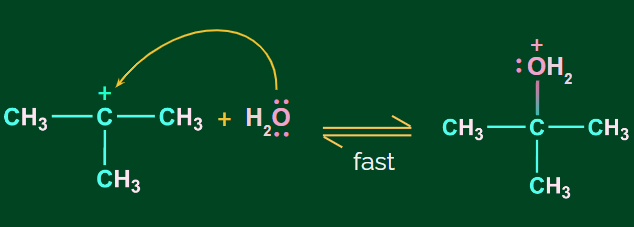
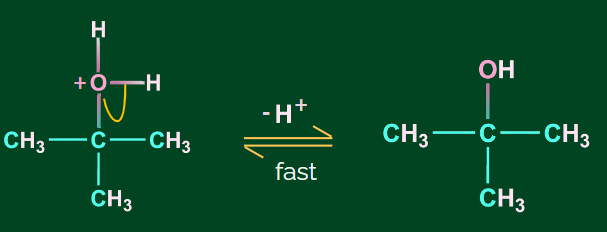
Step 3: Deprotonation to form alcohol
Examples:
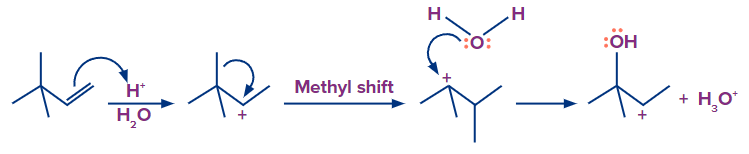
In the second example, the rearrangement by methyl shift will take place to form a stable tertiary
carbocation.
Hyboration-Oxidation Reaction:
The hydroboration oxidation reaction is an organic chemical reaction that is used to convert alkenes into primary alcohols or alkynes into ketones or aldehyde. This is accomplished through a net addition of water (across the entire double bond)using an anti-Markovnikov Rule.
Let us consider the example of Hyboration oxidation of alkene hex-1-ene.
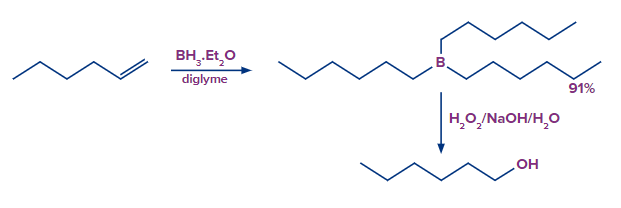
Mechanism of Alkene Hydroboration-Oxidation
The mechanism of hydroboration oxidation can be thought of as an anti-Markovnikov reaction in which a hydroxyl group attaches itself to the less substituted carbon.
Characteristics of Hydroboration
- Stereospecific -syn addition
- Anti Markovnikov mechanism
- Primary alcohol formation
The conversion of alkenes into neutral alcohols takes place here. The entire reaction can be broken down into two steps, as explained below.
- The Hydroboration Process
The first step is to add borane in the form of BH3 to the given double bond. This results in the transfer of a hydrogen atom to the carbon atom next to the carbon atom bonded with the boron atom. The hydroboration step is now repeated twice, yielding three alkenes attached to the boron atom from the starting BH3.
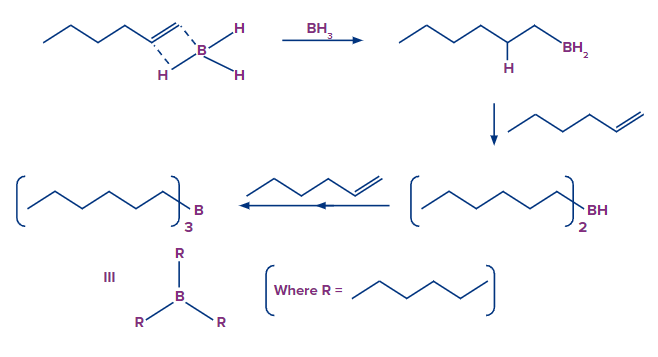
- The Oxidation Process
Now that the trialkyl borane has been produced, the second step in the hydroboration process can begin. The boron atom is attacked in this step by the hydroperoxide ion, which is nucleophilic in nature. The R group, along with its electron bond pair to the adjacent oxygen atom, is now rearranged.
The ion of hydroxide has now been removed. This process is repeated three times to produce trialkyl borate as the product. This trialkyl borate is now treated with water to produce the required primary alcohol. This step of the mechanism is illustrated below.
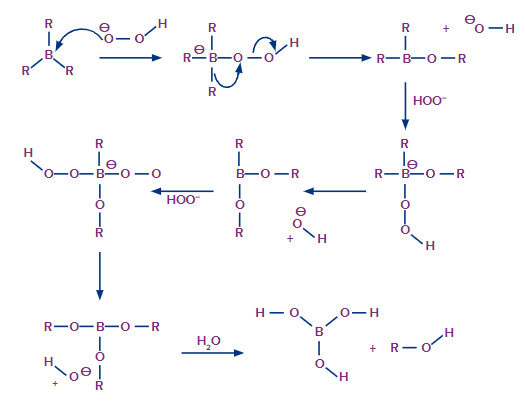
Hydration by Oxymercuration-Demercuration:
Alkenes can be converted to alcohol using the oxymercuration-demarcation reaction. An alkene is treated with mercuric acetate in a Tetrahydrofuran–water solution to produce a product, which can then be reduced with NaBH4 to produce alcohol. Markovnikov's rule operates in this reaction.
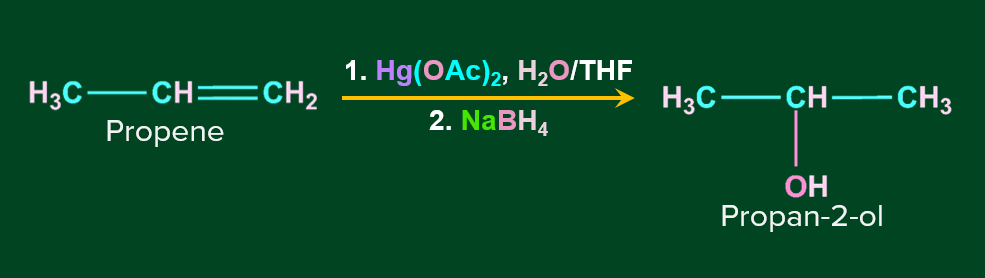
Mechanism of Oxymercuration-Demercuration
The mechanism of this reaction follows Markonikov's regioselectivity rule, with the hydroxyl group joining the most substituted carbon atom and the hydrogen atom joining the least substituted carbon atom.
Characteristics of Oxymercuration-Demercuration
- Stereospecific -syn addition
- Markovnikov mechanism
The reaction mechanism is depicted below.
Step 1: In this step, the nucleophile C=C bond attacks the electrophile Hg, resulting in the acetate ion leaving as the leaving group and the formation of cyclic mercurinium ion.
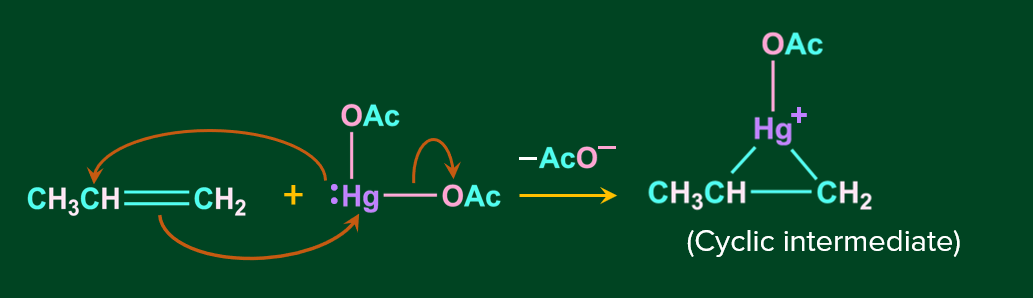
Step 2: In this step, the nucleophile attacks one of the carbons linked to Hg, causing the C-Hg bond to cleave.
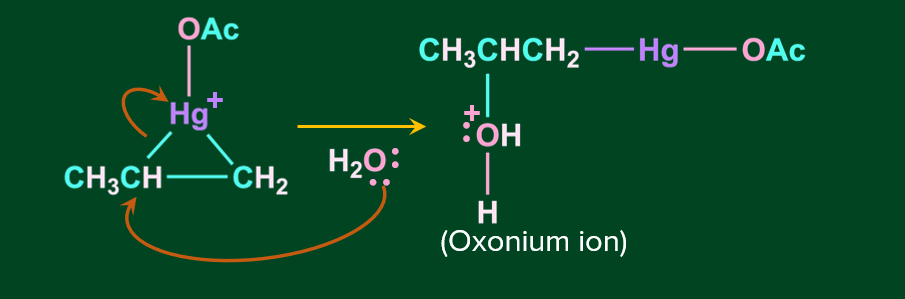
Step 3: In this step, oxonium ion is deprotonated in the presence of a base acetate ion, resulting in alcohol.
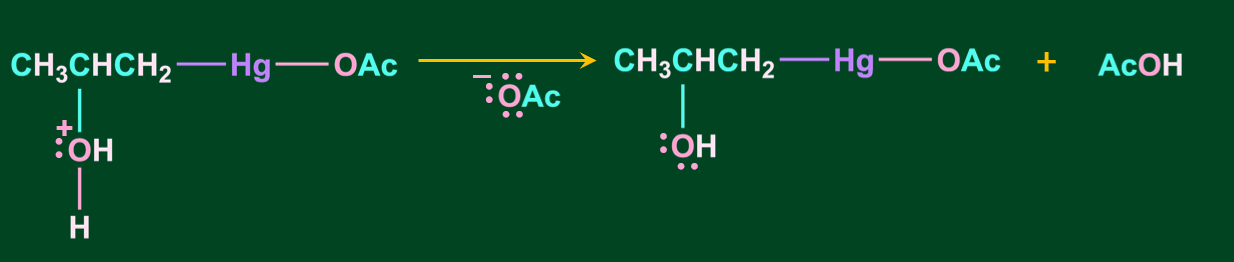
Step 4: In the final step, the addition of sodium borohydride replaces the acetyl mercury with a hydrogen atom, resulting in the formation of alcohol via a new C-H bond.
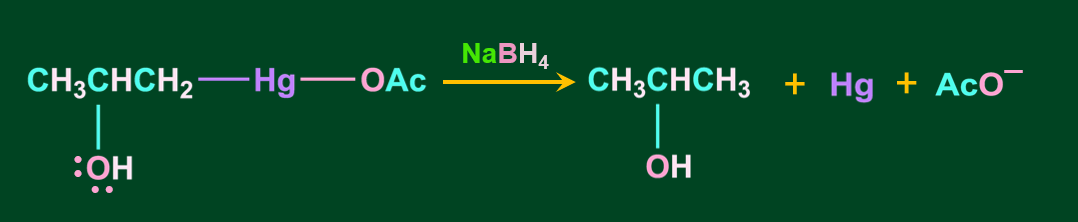
|
Reactions |
Stereochemistry |
Product |
Rule |
|
Acid catalyzed Hydration |
Stereospecific -anti addition |
Secondary, tertiary |
Markovnikov mechanism with rearrangement |
|
Hyboration-Oxidation Reaction |
Stereospecific -syn addition |
Primary |
Anti-Markovnikov mechanism |
|
Oxymercuration-Demercuration Reaction |
Stereospecific -syn addition |
Secondary, tertiary |
Markovnikov mechanism |
Hydrogenation reaction of alkenes
Alkenes adds up one molecule of dihydrogen gas in the presence of finely divided nickel, palladium, or platinum to form alkanes.

This reaction is known as the Sabatier-Sanderson reaction when Ni is used as a catalyst.
An illustration of the addition of dihydrogen to ethene in the presence of nickel is given, Ethene is adsorbed over the catalyst surface. Syn addition (addition of H2 from the same side) of dihydrogen happens and a four-membered high energy transition state is formed in the process. Finally, ethene is reduced to ethane.
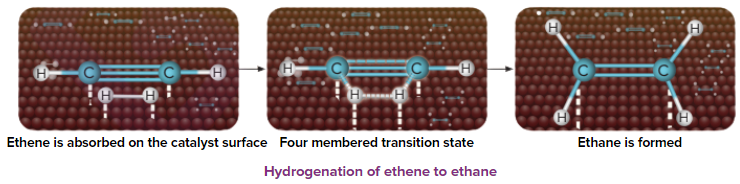
Addition of Hydrogen Halides (HX)
Hydrogen halides (HCl, HBr, HI) when added to alkenes give alkyl halides. This is known as the hydrohalogenation process. It is an electrophilic addition reaction.
Alkene + HX Alkyl halide
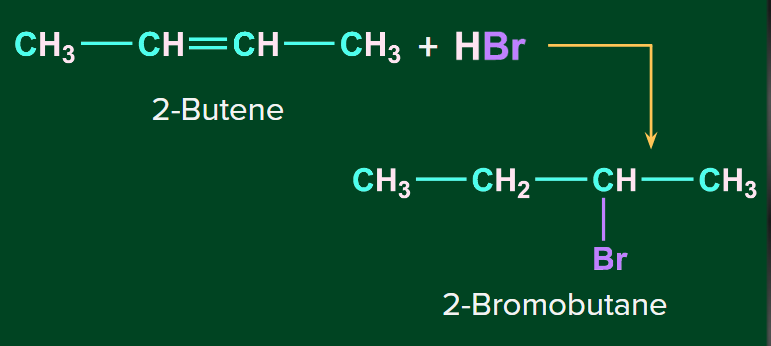
Addition of HBr to symmetrical alkenes
The addition reaction of HBr to symmetrical alkenes takes place by an electrophilic addition mechanism in which the hydrogen ion gets attached to one carbon and bromide gets attached to the second carbon of the double bond.
Example-1:
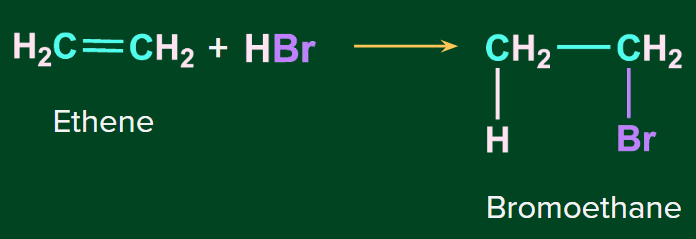
Example 2:
Addition of HBr to unsymmetrical alkenes
When HBr is added to the unsymmetrical alkenes, there are two possibilities of the addition of HBr to propene (unsymmetrical alkene) in which double bonds can break in such a way that one carbon gets the negative charge and the other gets the positive charge. The breaking of the bond takes place in such a way that the bond-forming carbocation should be the most stable.
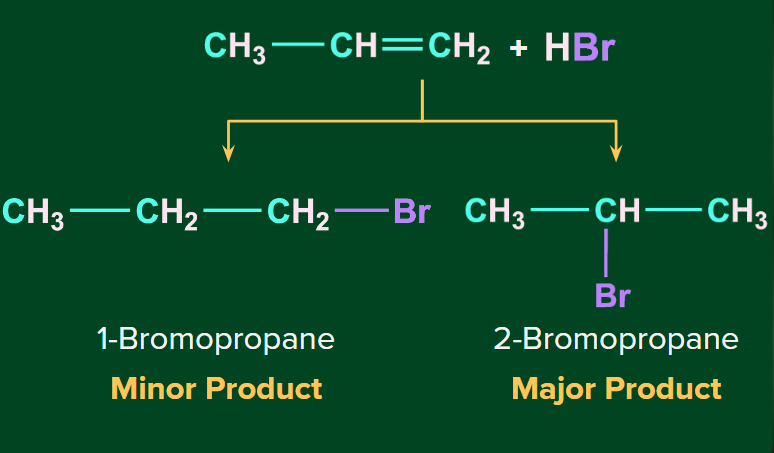
The major product formed upon the addition of HBr to an unsymmetrical alkene follows Markovnikov’s rule.
Mechanism (Markovnikov Addition)
Step 1: The H+ ion attacks the double bond (electrophilic addition of H+) and gets attached to the carbon having more hydrogen atoms.
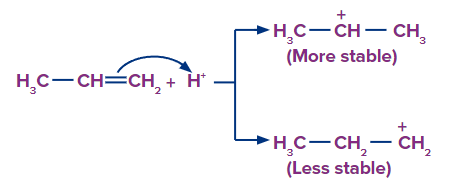
Step 2: Since, the 2o carbocation is more stable than 1o carbocation, so the stable carbocation will be available more for the attack of Br- and the product formed from 2o carbocation is the major product.
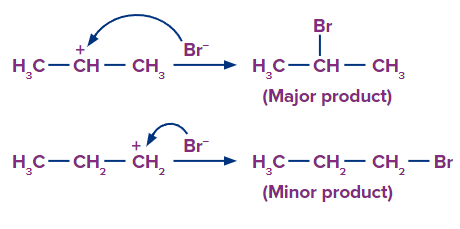
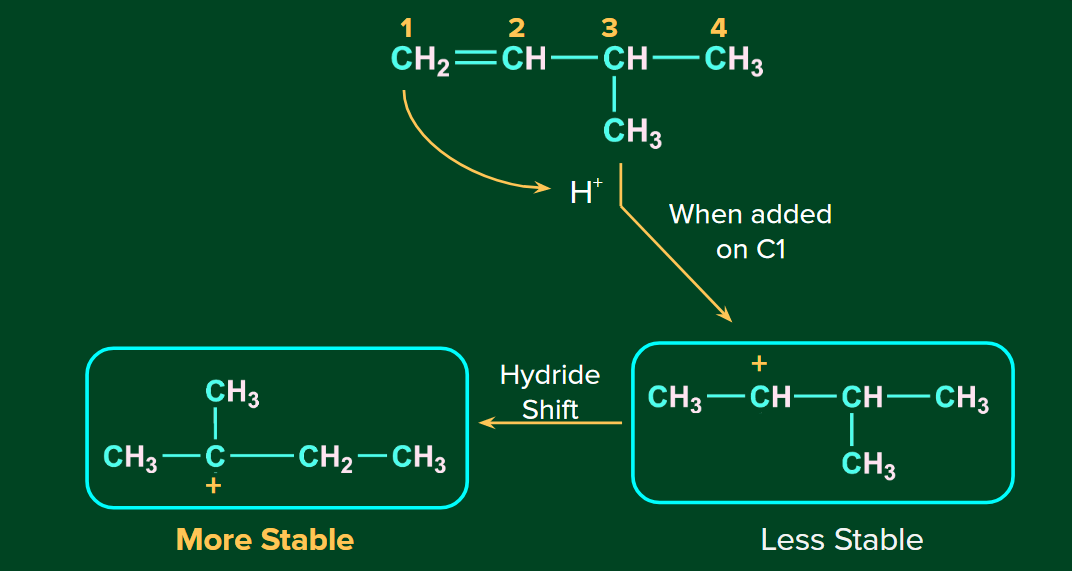
Rearrangement of Carbocation
The H+ ion attacks the double bond and it gets attached to the carbon having more hydrogen atoms. The 2o carbocation undergoes rearrangement (1,2-hydride shift) to form a more stable 3o carbocation. Finally, Br- ion attacks on the 3o carbocation to form the major product.
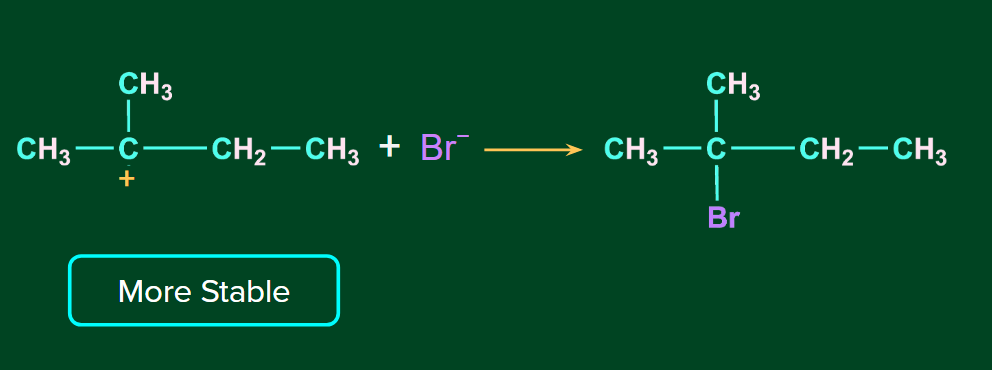
Order of reactivities of hydrogen halides
As we move down the group, the bond length of HX increases and the bond strength decreases (i.e., the H-X bond becomes weak and can be easily dissociated). Thus, the reactivity of hydrogen halides towards addition reactions increases down the group.
HF < HCl < HBr < HI
Mechanism (Anti-Markovnikov Addition)
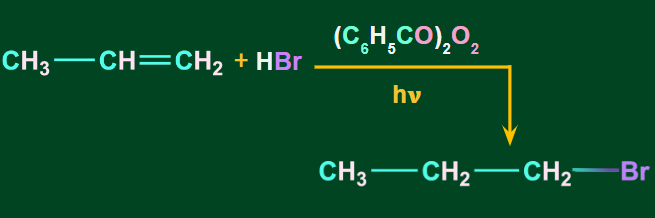
When alkenes are treated with HBr in the presence of peroxides, anti-markovnikov’s addition occurs such that the H-atom of HBr gets attached to the C-atom with the fewer H-atoms. It is also known as the peroxide or the Kharasch effect.
The addition of HBr to an alkene in the presence of peroxide takes place through a free radical mechanism.
Where is benzoyl peroxide
The mechanism is given as follows:
- Chain initiation
Step 1: The peroxide bond of RCO–O–O–CO–R breaks into radicals (i.e., RCOO) in the presence of sunlight, which is then followed by the removal of carbon dioxide and the formation of alkyl radical (i.e., R).
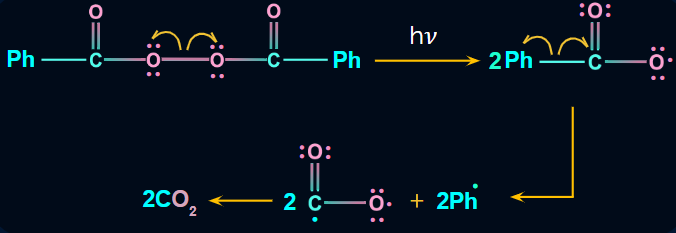
Step 2: Now, the alkyl free radical (R) (i.e., phenyl radical) attacks on HBr and results in the formation of R−H (i.e., benzene in benzoyl peroxide) and Br radicals.
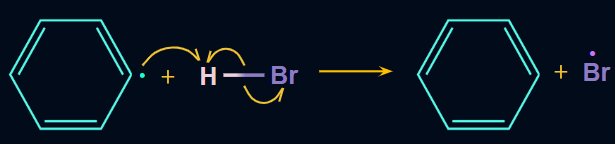
- Chain propagation
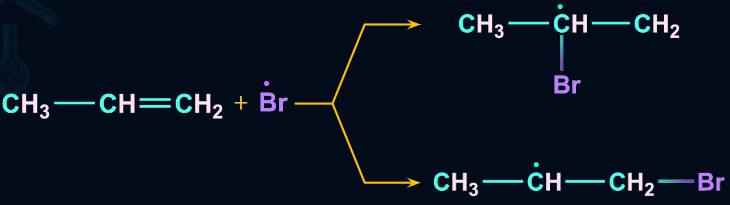
The order of stability of the free radicals is 3oradical>2oradical>1oradical. The two intermediates formed are given and we know that a 2o free radical is more stable than a 1o free radical.
- Chain Termination
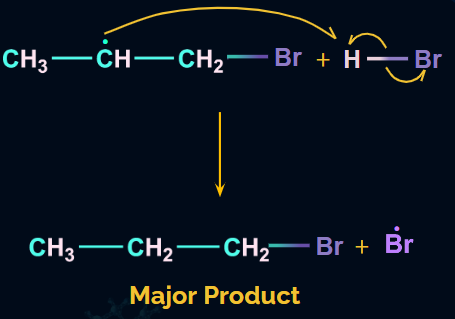
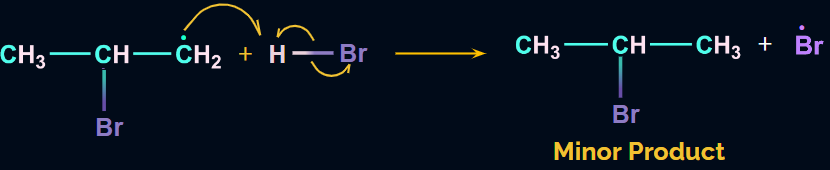
The hydrogen radical formed from HBr will attack the 2o free radical to give the major product. Hence, in the major product, Br will be attached to the carbon with more hydrogen atoms, which is in contradiction to Markovnikov’s rule. Hence, it is an anti-Markovnikov addition.
Oxidation of Alkenes-Hydroxylation of Alkene
During hydroxylation, the carbon-hydrogen bond in the alkene is converted to a carbon-hydroxyl bond. Hydroxylation of alkenes is an oxidation process. A reagent that increases the oxidation number is known as an oxidising agent.
Alkenes can be hydroxylated via two distinct stereochemical routes:
- Anti-dihydroxylation
- Syn-dihydroxylation
The anti-dihydroxylation mechanism is used to open epoxides, whereas potassium permanganate or osmium tetroxide produces syn-dihydroxylated products. The osmium tetroxide reaction can also occur in two steps: 1) OsO4 in pyridine, then 2) H2S or NaHSO3.
Anti-Dihydroxylation
The epoxides produced by m-CPBA can be cleaved by aqueous acid (meta-chloro peroxy benzoic acid). The epoxide's conjugate acid is formed by proton transfer from the acid catalyst, and it is attacked by nucleophiles like water. The anti-hydroxylation of the double bond is the outcome. The oxidation state of any of an epoxide's atoms or groups is unaffected by hydration. The reaction conditions dictate the mechanism of ring-opening in epoxides.
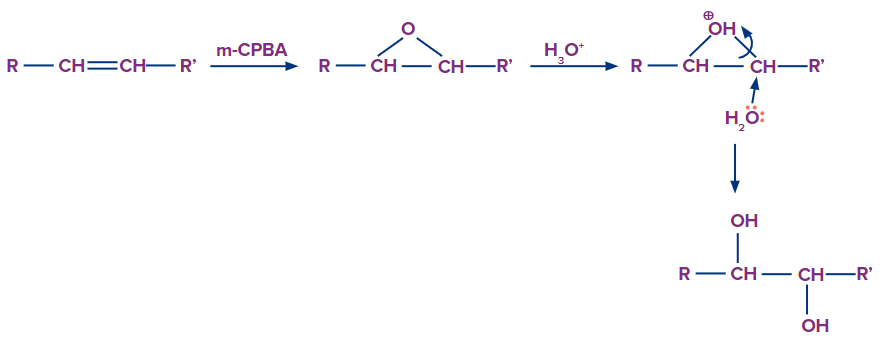
Syn-Hydroxylation
Dihydroxylated products are formed by reacting pyridine with aqueous potassium permanganate or osmium tetroxide. Both reactions appear to be the result of the same mechanism (shown below). We would expect syn-stereoselectivity in the bonding to oxygen based on the mechanism shown here.
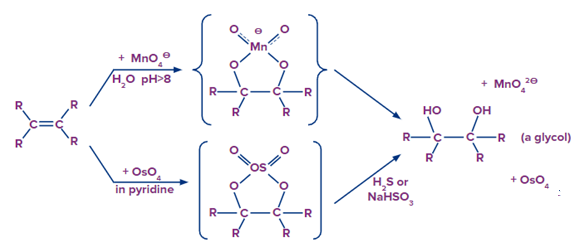
Baeyer’s Reagent (Potassium Permanganate)
Depending on the reaction environment, alkenes can be moderately or substantially oxidized. Alkenes, for example, create vicinal diols in a neutral permanganate solution.
The following summarizes the generic reactions for the various mild oxidative situations.
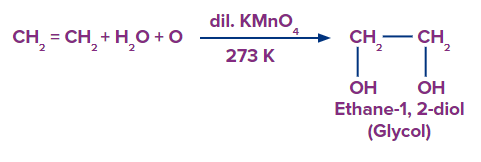 \
\
During severe oxidation with acidic potassium permanganate, the alkene is split into two components.
A ketone is formed when unsaturated carbon is disubstituted.
A carboxylic acid is formed when unsaturated carbon is monosubstituted.
If the terminal is unsaturated, the result is carbon dioxide and water.
The generic reactions for the various oxidative conditions - strong- are summarized below.
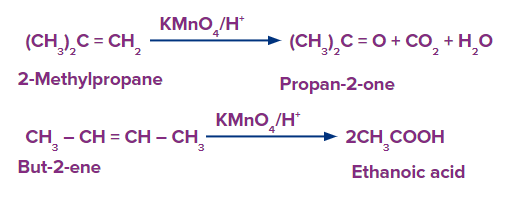
Practice Problems
Q1. In the given reaction, what will be the major product?
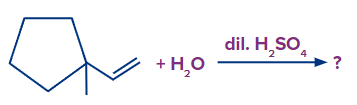
A. 2-(1-methyl cyclopentyl) Ethan-1-ol.
B. 1,1-Dimethylcyclohexan-2-ol.
C. 2,2-Dimethylcyclohexan-1-ol.
D. 1,2-Dimethylcyclohexan-1-ol.
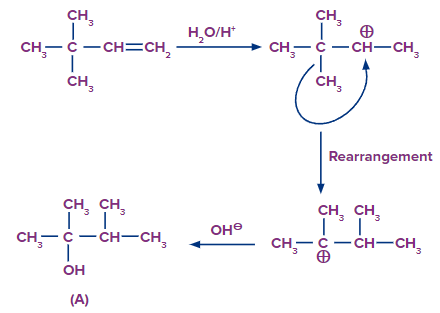
Solution: Step 1: The first step is the protonation of the alkene, which results in the formation of a secondary carbocation. However, in this case, the carbocation formed results in the ring expansion and forms a six-membered stable ring.
Step 2: The addition of OH- occurs and the product formed is shown as follows:
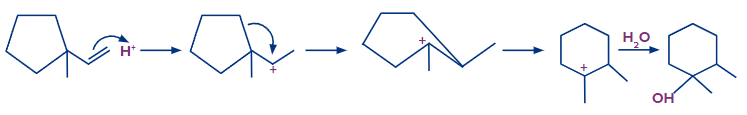
After rearrangement and addition of H2O, the final product is 1,2-dimethyl cyclohexan-1-ol.
Hence, a correct answer is an option (D).
Q2.
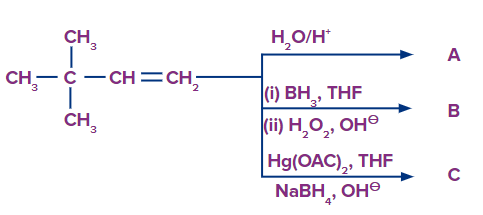
Predict the product A, B and C are
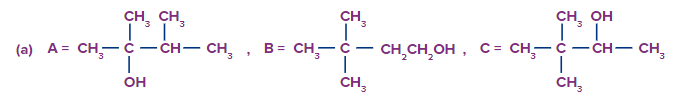
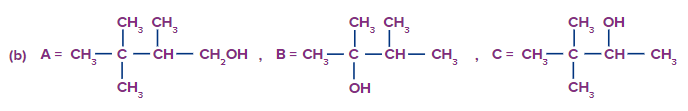
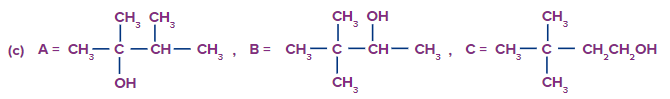
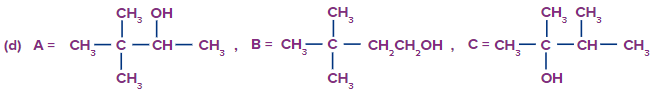
Solution: The oxymercuration-demarcation reaction can convert alkenes to alcohol. In a THF–water solution, an alkene is treated with mercuric acetate to produce a product, which can then be reduced with NaBH4 to produce alcohol. According to Markownikov's rule, addition occurs in this reaction.
The hydroboration oxidation reaction is an organic chemical reaction that converts alkenes to neutral alcohols or alkynes to ketones or aldehydes. This is accomplished via a two-step procedure that includes a hydroboration step and an oxidation step that employs an anti-Markovnikov Rule.
A Markovnikov addition reaction occurs when (acid catalyzed hydration) is added to alkenes.
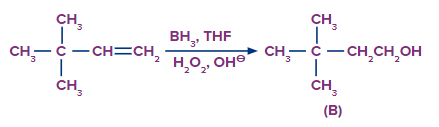
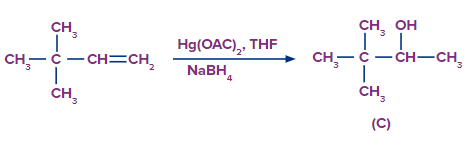
Hence, correct option is (A).
Q3. Hydrocarbon A adds hydrogen to produce n-hexane in the presence of a platinum catalyst. Only a single Bromo compound is generated when hydrogen bromide is introduced to A instead of hydrogen??
A.
B.
C. Both (a) and (b)
D.
Solution: As (hex-3-ene) is a symmetrical alkene, only one product will be obtained on the addition of HBr, i.e., 3-Bromohexane.
As (hex-2-ene) is an unsymmetrical alkene, more than one product is formed. Thus, 2-Bromohexane and 3-Bromo hexane will be formed as the products.
As (hex-1-ene) is an unsymmetrical alkene, 2-Bromohexane will be formed as the major product and 1-Bromohexane will be formed as a minor product.
Since only one bromo compound is formed as the product in the case of hex-3-ene. Hence option (A) is the correct answer.
Q4. Why don’t the hydrogen halides (HF, HCl, and HI) exhibit the peroxide effect?
Solution: In the propagation step of the chain mechanism, there are two steps, which are given as follows:
Step 1:
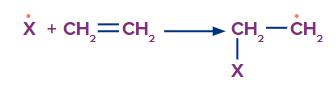
Here, the CX sigma bond is formed and the C=C pi-bond is broken. In the case of X = F, Cl, Br, the sigma bond is strong. Thus, the formation of a sigma bond with these halogens is an exothermic reaction. However, if X = I (iodine), the sigma bond between C and I is very weak. Thus, it is an endothermic reaction. Therefore, this step is unfavourable with iodine. Hence, HI cannot give this reaction.
Step 2:
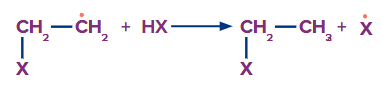
Here, the CH bond is formed and the HX bond is broken. As we move down the group, the bond length of HX increases and the bond strength decreases. Thus, in the case of HF and HCl, this step is endothermic. However, in HBr, the bond is weak. Thus, it is exothermic in nature.Thus, this step is unfavourable with HF and HCl. Hence, both HF and HCl cannot give this reaction.
Hence, HBr is the only hydrogen halide that can give both the steps of propagation and show peroxide effect on reaction with an alkene in the presence of peroxide.
Frequently Asked Questions
Q1. What are some typical responses that alkenes cause?
Answer: The addition reaction to a C=C double bond is the most frequent form of reaction for alkenes. In an addition reaction, a tiny molecule is added to several bonds, resulting in the conversion of one bond into two bonds (a decrease in the unsaturation degree).
Q2. What exactly is a stereospecific reaction in organic chemistry?
Answer: A reaction that is stereospecific is one in which the outcome is determined by the stereochemistry of the reactants. Generally speaking, one stereoisomer of one reactant produces one stereoisomer of that reactant's product, whereas a different stereoisomer of the same reactant produces a different stereoisomer of that product.
Q3. Why are Lindlar catalysts' lead salts and quinoline 'Poisoned'?
Answer: Typical palladium catalysts have high catalytic activities that can reduce even double bonds. Alkanes can be formed during the hydrogenation of alkynes using such catalysts (the alkene products undergo further hydrogenation under the influence of the catalyst).The poisoned Lindlar catalyst is incapable of reducing double bonds. As a result, using this catalyst in the hydrogenation of alkynes does not result in the formation of alkanes.
Q4. What exactly is a regiospecific reaction in organic chemistry?
Answer: When multiple structural isomers are theoretically feasible, a chemical reaction is said to be regiospecific if it only ever yields one of those isomers.


