-
Call Now
1800-102-2727
Raoult's Law - Statement, Graphical Representation, Limitations, Practice Problems and FAQ
Assume you have a class of 20 students. These 20 students are all bright, deserving, and well-mannered. A total of 10 students are admitted to this class from the outside. They are rather distracting, arrogant, and uninterested in studies. The introduction of 10 outside students has now degraded the overall performance of the class of 20 good students. This is because they caused a distraction among the 20 good students. As a result, the performance of each student and the entire class has changed.
The situation is quite similar to Raoult's law. The 20 students are analogous to pure solvents that exert a certain vapour pressure (performance) in their pure state, while the 10 students introduced from outside are analogous to non-volatile solutes that have their own vapour pressure (performance). The addition of a solute to a pure solvent alters the overall vapour pressure, just as the addition of those ten students from outside affected the overall performance of the class.
Let us now try to understand how the total vapour pressure gets affected, and what is the new vapour pressure of each component, with the help of Raoult's Law.
Raoult’s law was proposed by a French chemist Francois-Marie Raoult while conducting an experiment in 1877. He found out that when substances were mixed in a solution, the vapour pressure of the solution decreased simultaneously.
TABLE OF CONTENTS
- Raoult’s Law
- Graphical Representation
- Limitations
- Practice Problems
- Frequently Asked Questions - FAQ
Raoult’s Law
Raoult’s law can be stated as, for a solution of volatile liquids, the partial vapour pressure of each volatile component in the solution is directly proportional to its mole fraction present in the solution.
Mathematically, Raoult’s law can be expressed as
![]()

Consider a container containing a solution of two volatile liquids A and B. Since both the liquids are volatile, the vapour phase contains particles of both components A and B
Thus for component A,
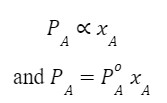
Where ![]() is the vapour pressure of a pure component of A at the same temperature.
is the vapour pressure of a pure component of A at the same temperature.
Similarly, for component B,
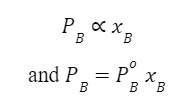
Where  is the vapour pressure of the pure component of B at the same temperature.
is the vapour pressure of the pure component of B at the same temperature.
According to Dalton’s law of partial pressures, the total pressure (Ptotal) over the solution phase in the container will be the sum of the partial pressures of the components of the solution and is given as
Ptotal= PA+PB
Substituting the values of PA and PB,
![]()
We know that xA+xB= 1 then,
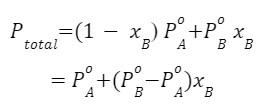
If yA and yB are the mole fractions of the components A and B respectively in vapour phase then, using Dalton’s law of partial pressures,
PA=yA Ptotal
PB=yB Ptotal
Graphical Representation
A plot of PA or PB versus the mole fractions xA and xB for a solution gives a linear plot as shown in the below figure. These lines pass through the points for which xA and xB are equal to unity. Similarly, the plot of Ptotal versus xB is also linear as shown in the below figure. The minimum value of Ptotal is ![]() and the maximum value is
and the maximum value is ![]() assuming that the component A is less volatile than the component B.
assuming that the component A is less volatile than the component B.
Limitations
- Raoult’s law is apt for describing the ideal solutions.
- Raoult’s law is applicable to only dilute solutions.
- Raoult’s law is not applicable to solutes that undergo dissociation or association in a particular solution.
Practice problems
Q 1. The vapour pressure of two liquids A and B are 80 torr and 60 torr respectively. Find the total vapour pressure of the solution obtained by mixing 3 mol of A and 2 mol of B.
Answer:
From Raoult’s law,

Q 2. The vapour pressure of the pure liquid A is 40 torr at 310 K. Given the vapour pressure of this liquid in the solution with solid B is 32 torr, calculate XA in solution.
Answer:
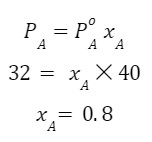
Q 3. 1 mol heptane (vapour pressure= 92 mm of Hg) is mixed with 4 mol octane (vapour pressure= 31 mm of Hg) form an ideal solution. Find the vapour pressure of the solution.
Answer:
Total moles =1+4=5

![]()
Q 4. Over the entire range of composition liquids A and B forms an ideal solution. At 350 K, the vapour pressure of pure A and pure B is 7✕103 Pa and 12✕103 Pa respectively. The composition of the vapour when it is in equilibrium with a solution containing 40 mol % of A at this temperature is:
Answer:

Frequently Asked Questions - FAQ
Q 1. Is the graph of pressure v/s mole fraction always a straight line?
Answer: If the solution is ideal, the 3 graphs (PA,PB and PT) will always be a straight line because
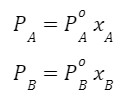
and
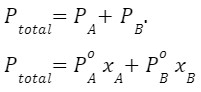
If the solution is non-ideal, the 3 graphs(PA,PB and PT) are not straight-line because
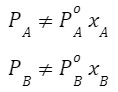
and
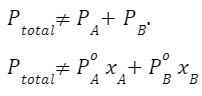
Q 2. What is the relationship of Raoult’s law with other laws?
Answer: Raoult’s law is quite similar to the ideal gas law. The only exception of Raoult's law is that it applies only to ideal solutions. We know that the ideal gas law assumes the ideal behaviour of gases in which the intermolecular forces that are present between dissimilar molecules is zero or do not exist. Raoult’s law on the other hand assumes that the intermolecular forces that exist between different molecules and similar molecules are equal.
Q 3. For what type of solutions Raoult’s law is applicable?
Answer: Raoult's law is valid only in the case of ideal solutions. For an ideal solution, solvent-solute interactions are the same as solvent-solvent or solute-solute interactions. This implies that both the solute and the solvent take the same amount of energy to escape to the vapour phase as when they are in their pure states.
Q 4. What is the main difference between Henry’s law and Raoult’s law?
Answer: Both these laws describe the relationship between a solution and its vapour that are in equilibrium. Henry’s law can be used to explain the dissolution of gas in a liquid solvent. Raoult’s law specifies the behaviour of solvents in a solution that is in equilibrium with its vapour pressure.
Related Topics
|
Types of Solutions |
Henry’s Law |
|
Gay-Lussac’s Law |
Avogadro's Law |
|
Dalton's Law of Partial Pressure |
Graham's Law of Diffusion |


