-
Call Now
1800-102-2727
Racemic Mixture – Enantiomers, Nomenclature, Characteristics, and Resolution of Racemic Mixture, Practice Problems and FAQ
Consider that you have two bags of toffees, one of which only contains red toffees and the other only contains blue toffees. There is the same number of toffees in both bags. Consider tearing open both toffee packages and pouring the contents into a bowl. At this point, you may claim that the toffees are split equally between the two colours.

Now imagine that the bowl contains two enantiomers of a compound in equal amounts rather than red and blue toffees. This mixture is now referred to as a racemic mixture.
Are you curious about what racemic mixtures and enantiomers are? Don't worry, in this article, we shall learn about them. We will learn more about racemic mixture, its properties, and how to isolate individual enantiomers from the mixture in this article.
TABLE OF CONTENTS
- Enantiomers
- Racemic Mixture
- Naming of Racemate Compounds
- Racemic Mixtures – Characteristics
- Racemic Mixtures – Resolution
- Practice Problems
- Frequently Asked Questions – FAQ
Enantiomers
To understand racemic mixtures and racemisation, we must first understand enantiomers. Enantiomers are molecules with the exact same chemical structure but different molecular arrangements. They frequently show a distinct set of chemical properties but a different set of physical properties.
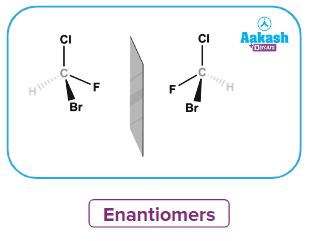
We must understand how these molecules impact the plane of polarised light in order to understand this form of isomers. The only way to distinguish two enantiomers apart is to run a polarised light beam across them and look at how they rotate the plane of the polarised light.
Racemic Mixture
Enantiomers are two organic isomeric compounds with the same molecular formula and the structural formula but are non-superimposable mirror images of each other. A mixture containing equal amounts of each of the enantiomers (50:50) is referred to as a racemic mixture. Though the individual isomer is optically active, the mixture is optically inactive since the optical rotation caused by the molecules of one isomer is exactly cancelled by the equal and opposite optical rotation caused by the same number of molecules of the other isomer.
- The mixture is said to be resolved when the enantiomers are separated.
- The resolution of a racemic mixture is a critical and essential technique widely practised because of the characteristic importance of one enantiomer over the other.
Naming of Racemate Compounds
When a racemic mixture exhibits no effect or change on polarised light passing through it, it is said to be racemate and the amounts of both compounds are equal.
There is a specific change that can be seen after these compounds have been separated and subjected to polarised light. Assuming that the ability of two racemic compounds to rotate polarised light is the only difference between them, this ability is used as the nomenclature to distinguish between the two compounds. The enantiomers rotate the plane of polarised light to either right or left, in other words clockwise or counterclockwise respectively. When the plane-polarised light is rotated in a clockwise direction, it is known as dextrorotation. Levorotation is the term used to describe the opposite direction of rotation.
In the nomenclature of the racemates, the chemical name of the compound is given a specific prefix indicating the direction of rotation. A "+" or "d" prefix is attached to a dextrorotatory compound in accordance with the racemic nomenclature and meaning. The meanings are all identical. Similar to this, a levorotatory compound has the prefix "-" or "l".
Example: (+)- fructose or d- fructose are all possible symbols for the dextrorotatory fructose compound. Similarly, (-)- fructose or l-fructose can be assigned to represent the levorotatory enantiomer.
Racemic Mixtures – Characteristics
- The enantiomers in the racemic mixture rotate light in opposite directions, hence the mixture will not have any optical properties. When both of these substances are present in the mixture in an equal quantity, it happens.
- Due to the similarity of all their physical characteristics, these compounds are difficult to identify and separate. These substances must be optically active to recognise them individually.
- The melting and boiling points are typically where the variations are seen. Additionally, they may exhibit various solubilities.
- Several pharmacological medications are available, either as pure enantiomers or as racemates. This is done to boost the bioavailability of medicinal substances due to the various chiral and racemic asymmetries in the biological system of the human body.
Racemic Mixture – Resolution
Achiral compounds or SN1 reactions naturally end with a racemic mixture containing a 50:50 composition of both enantiomers. Resolution refers to the process of separating racemates into their individual enantiomers. The physical characteristics of the two enantiomers are almost the same making the separation or resolution of them using solubility or crystallisation and other physical properties is almost impossible and very challenging.
Diastereomers, unlike enantiomers, differ in their physical characteristics and can be easily separated into pure components. When a racemate containing equal proportions of the enantiomers reacts with another pure chiral compound, two diastereomers are formed. These diastereomers can be separated in pure form and hydrolysed to pure enantiomers and the reagent enables the resolution of the enantiomers in pure form.
For example, a racemate of acids can be reacted with a chiral alcohol to produce a diastereomeric ester or with a base to give a diastereomeric salt. The same reactions can be used to separate an alcohol racemate or a racemate base. For example, a racemic mixture of alcohol reacts with a chiral carboxylic acid to form diastereomeric esters. If the acid used is pure (+) enantiomer, the products are a mixture of (+ +) and (+ -) diastereomeric esters. They can be easily separated and hydrolyzed to yield or "resolved" to their enantiomerically pure (+) and (-) alcohols. Chemical and physical reactions can form diastereomers, with different properties needed for easy separation.
This general concept is illustrated in the above Figure. Say we are having an equal number of right (R) and left (L) handed bolts of the same size mixed together. For separating them, take a left or a right handed nut. A left-handed bolt can be screwed to both left and right-handed nuts, but their direction of winding and unwinding are in opposite directions, thus differentiating the product. The products will be of R-R and R-L combinations that can be easily identified. R-R and R-L combinations can be easily separated. The unwinding of the nut from the separated combinations will give the R or L form of bolts.
Enantiomers also can be similarly made to react with a pure optically active substance to give (+ +) or (- -) stereoisomers that can be easily separated. And ultimately isolated in pure forms of + or -. Because enantiomers have identical physical properties, they are rarely separated by simple physical methods such as fractional distillation or crystallisation. Enantiomers only behave differently under the influence of another chiral substance, and almost all methods of enantiomer resolution are based on this fact.
Separation of Racemic Mixtures of Acids
Enantiomers are often separated by converting them into a mixture of diastereomers, which will have various physical properties, such as melting and boiling points and solubility.
For instance, if you take a racemic or (R)/(S) mixture of enantiomers of a carboxylic acid and convert it to a salt with a chiral amine base bearing the (R) configuration, the salt will be a mixture of two diastereomers, ((R)-acid(R)-base) and ((S)-acid(R)-base). Pure optically active enantiomeric acids can be separated from their racemic mixture by treating with an optically active base substance. The reaction is a simple acid-base reaction forming salt and water. But the salt here will be of two types and diastereomeric with distinguishing physical properties. The two salts can be separated utilising the difference in physical properties first, and hydrolysed to get back the pure enantiomeric substances.
For example, if you want to resolve a racemic mixture of acids, choose a chiral base of either d or l form and allow them to react to form diastereomeric salts. A chiral d-base forms two salts with a racemic mixture (d/l) acid namely, d-acid - d-base and l-acid - d-base. The salts being diastereomeric will have different physical properties enabling resolution, from which the pure form enantiomers can be liberated.
The process has to be sometimes repeated till the maximum optical rotation indicating the pure compound is achieved.
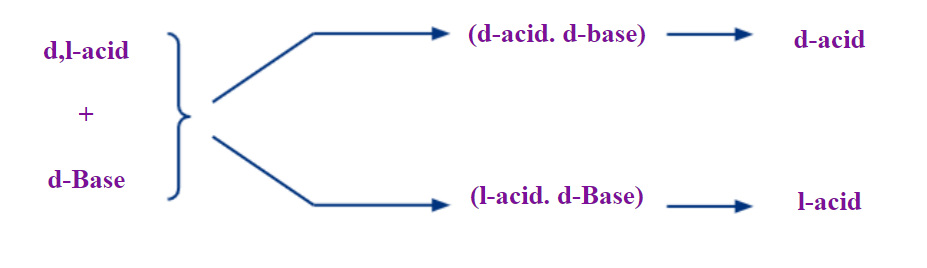
The optically active bases or acids can be natural or synthetic. Many naturally occurring chiral bases like brucine, strychnine, and quinine are available for this kind of resolution of racemates. Examples of synthetic optically active bases that are available are amphetamine, 1-phenylethanamine, and 2-amino-1-butanol.
Separation of Base Racemates
In the light of the discussion above, a mixture of enantiomers of a base can be separated by reacting the mixture with an optically pure acid which forms diastereomeric salts. These salts are separated and hydrolysed to obtain pure optically active base enantiomers. Naturally occurring chiral acids of (-)-malic acid, (+)-tartaric acid, and (-)-mandelic acid are widely used in the separation of base racemic mixture.
Resolution of Alcohol Racemates
A chiral acid, on reacting with the racemate alcohol, shall form diastereomeric esters. But the products mostly are liquid in nature and techniques like crystallisation cannot be applied. Other techniques like chromatography, solvent extraction are tried for the separation of pure alcohols from the racemate mixture.
Resolution Methods
Considering the importance and necessity of specific enantiomers, other methods to selectively isolate a particular enantiomer at the expense of the other are being developed. Chemicals that selectively react with one of the enantiomers can be used kinetically to destroy the unwanted enantiomer leaving behind the required enantiomers for further isolation. Chemicals with higher physical interaction may hold back one of the enantiomers and allow the movement of the other enantiomer, say in chromatographic methods enabling the separation of them. But all these ideas are developed on an individual case of separation rather than as a common technique.
Practice Problems
1. Which of the following statements is incorrect?
a. Racemic modification is an equal mixture of dextro- and levo rotatory enantiomers.
b. More than one chiral carbon centre can be found in meso compounds.
c. Meso compounds are optically inactive due to external compensation.
d. Racemic mixtures are designated as d/l pairs.
Answer: C
- A racemic modification is a combination of enantiomers in exactly equal amounts. Racemic modifications are optically inactive because, when enantiomers are combined, the rotation brought on by one isomer's molecule is precisely counterbalanced by the rotation brought on by a molecule of that isomer's enantiomer. Thus, the statement given in option A is correct.
- A meso compound is an achiral substance that has chiral centres. A meso compound has multiple chirality centres despite being achiral and optically inactive (i.e., it does not rotate the plane-polarised light). Thus, the statement given in option B is correct.
- The molecules in meso form have a plane of symmetry as a result of which the optical rotations of the top and lower parts are equal and in the opposite direction, balancing internally and making the compound optically inactive. Internal compensation is the name given to this property. Therefore, meso compounds are optically inactive due to total internal compensation. Thus, the statement given in option C is incorrect.
- Racemic mixtures contain equal amounts of two enantiomers which are optically active. Though the individual isomers are optically active, the mixture is optically inactive since the optical rotation caused by the molecules of one isomer is exactly cancelled by the equal and opposite optical rotation caused by the same number of molecules of the other isomer. Since one enantiomer is dextrorotatory (d) and the other is levorotatory (l), racemic mixtures are designated as d/l pairs. Thus, the statement given in option D is correct.
So, option C is the correct answer.
2. Among the following, which statement about any (S)-enantiomer is true?
a. It rotates the plane-polarised light to the left
b. It rotates the plane-polarised light to the right
c. It is a racemic form
d. It is the mirror image of the related (R)-enantiomer.
Answer: D
Solution: Carbons with four different substituents on them are referred to as stereocenters (also known as chiral centres) and are classified as having either R stereochemistry or S stereochemistry. If a molecule has one stereocenter with a R configuration, its mirror image would have a S configuration stereocenter, and vice versa. The R,S is related to the configuration of the penultimate hydroxyl group and has nothing to do with plane polarised light rotation.
So, option D is the correct answer.
3. How many optical isomers are possible in a compound with one chiral carbon?
a. 5
b. 4
c. 2
d. 3
Answer: C
Solution: Two optical isomers exist in a compound that only contains one chiral carbon. These are the enantiomers, a pair of non-superimposable mirror images. R and S will be the configuration of these isomers.
Also, the total number of optical isomers =2n, where n is the number of chiral centres.
21=2; therefore, a compound with one chiral carbon has two optical isomers
Therefore, a compound with one chiral carbon has two optical isomers (+) and (-).
So, option C is the correct answer.
4. The number of racemic forms of molecules having (n) different chiral carbons is:
a. 2n
b. 2n
c. 2n-1
d. 2n+1
Answer: C
Solution: The number of racemic forms of molecules having (n) different chiral carbons is 2n-1 whereas the number of configurational isomers of molecules having (n) different chiral carbons is 2n.
So, option C is the correct answer.
5. Racemic mixture is a
a. Mixture of enantiomers and diastereomers
b. 50:50 mixture of enantiomers
c. Mixture of enantiomers in any ratio
d. Mixture of diastereomers
Answer: B
Solution: Enantiomers are two organic isomeric compounds with the same molecular formula and the structural formula but are non-superimposable mirror images of each other. A mixture containing equal amounts of each of the enantiomers (50:50) is referred to as a racemic mixture. Though the individual isomer is optically active, the mixture is optically inactive since the optical rotation caused by the molecules of one isomer is exactly cancelled by the equal and opposite optical rotation caused by the same number of molecules of the other isomer.
So, option B is the correct answer.
Frequently Asked Questions – FAQ
1. What are some examples of racemic mixtures used in pharmaceuticals?
Answer: The enantiomers of some pharmacological compounds, which are chiral, have different biological activity. One enantiomer or a racemic mixture of them may be offered for use in such cases. Thalidomide, ibuprofen, cetirizine, and salbutamol are a few examples. Amphetamine is a well-known medication whose effects vary according to the ratio of its enantiomers. An unequal combination of both amphetamine enantiomers makes up Adderall. Amphetamine, when prescribed by a doctor and used to treat medical conditions including obesity, narcolepsy, or attention deficit hyperactivity disorder (ADHD), is legal .
2. How can the components of a racemate combination be distinguished?
Answer: When both of a substance's enantiomers are present in equal amounts in a solution, it is known as a racemic mixture, also known as a racemate. Racemic mixes are denoted by the prefix (d/l)- or (+/-)- before the name of the substance. Due to the equal and opposite specific rotations of enantiomers, a racemic mixture has no optical activity. As a result, polarimetry by itself is unable to distinguish between an achiral substance and a racemic mixture. Chirality is a characteristic of individual molecules. Solution properties include optical activity. Despite being a racemic mixture of chiral molecules, it has no net optical activity.
3. What other physical characteristics of enantiomers are different?
Answer: Since the physical characteristics of the two enantiomers are so similar, it is difficult to distinguish between them. Enantiomers may differ somewhat in boiling points, solubility, and melting points in addition to the polarisation effect. These characteristics are frequently used to distinguish these substances.
4. Where does the term "racemate" for enantiomers come from?
Answer: The Latin word "racemes," which signifies a bunch of grapes, is where the word "racemate" originates. Louis Pasteur discovered the first enantiomers, tartaric acid and racemic acid, in grapes, and these kinds of enantiomers are known as racemate chemicals. They use a distinctive nomenclature. The optical characteristics of these compounds are determined by prefixing them with symbols and letters. The two enantiomers of a compound will be the mirror images of each other.


