-
Call Now
1800-102-2727
Ethers: Introduction of Ethers, Classification of Ethers, Nomenclature of Ethers, Uses of Ethers, Practice Problems & Frequently Asked Questions
Ether usage has been spread from the health industry to the field of sanitization. Ether is much safer than other groups of chemicals since it acts swiftly and has no side effects in smaller doses.

Because of their intoxicating and anaesthetic effects, ethers of the ethyl group, such as diethyl ether, are used as recreational drugs. This means that because they are safe when taken at therapeutic amounts and have the intended effects for drug addicts, they can be used recreationally.
One part diethyl ether and three parts alcohol (ethanol) are combined to create the analgesic Spirit of Ether.
Ether-based drugs can be used to relieve stomach discomfort and general stomach pain. Headaches and general anxiety can both be treated with ether-based drugs. The drugs based on ether can also be used to relieve spasm-related pain.
They are also used in supplements and syrups to impart the flavour and aroma of various scents.
Come let us know about what ethers are and a bit more about them.
Table of Contents:
- Introductions of Ethers
- Classification of Ethers
- Nomenclature of Ethers
- Uses of Ethers
- Practice Problems
- Frequently Asked Questions(FAQs)
Introduction of Ethers:
Organic compounds known as ethers, which have an oxygen atom sandwiched between two alkyls or aryl or a mixture of alkyl and aryl groups. The two alkyl or aryl groups,attached to the oxygen can be the same or different.
So the ethers can be represented as R-O-R, R-O-Ar, or Ar-O-Ar where, R will be an alkyl group, and Ar will be an aryl group.
The ether components are tasteless, volatile liquids, whereas methoxy methane and methoxy ethane are gases. Lower homologues include liquids that are combustible, transparent, fragrant and have a unique ethereal odour.
Ethers are polar compounds. Higher electronegativity of oxygen compared to the attached carbon brings a partial polar nature by which oxygen gets a partial negative charge and carbon a partial positive charge.
Classification of Ethers:
This classification is based on the nature of the hydrocarbon groups on either side of the oxygen.
We can classify four types of ethers, as follows:
1. Symmetrical Ethers or Simple Ethers: These ethers have identical alkyl or aryl groups connected on each side of the functional group -O-.
Example: CH3-O-CH3 (dimethyl ether), C2H5-O-C2H5 (diethyl ether).
2. Unsymmetrical Ethers or Mixed Ethers: These ethers have different alkyl or aryl groups connected on either side of the functional group -O-.
Example: CH3-O-C6H5 (methyl phenyl ether), C3H7-O-CH3 (methyl propyl ether).
3. Cyclic Ethers: Cyclic ethers are heterocyclic compounds that contain an oxygen atom in a saturated ring. Due to their inert nature, many cyclic ethers have well-known names and are frequently used as solvents. These ring forms are common in biological molecules like sugars and DNA. The rings are set up so that place one is occupied by oxygen.
For example, Epoxide, commonly known as oxirane, is a type of cyclic ether.
4. Crown Ethers: Crown ethers are so called because of the appearance of the heterocyclic ring looking like a crown. It has four or more ether groups forming a ring structure. So it is a polyether. Each oxygen in the crown ether is separated by two or more carbon atoms.
Crown ethers are named with the total number of carbon and oxygen atoms forming the ring structure and the number of oxygen atoms in the structure. The two numbers are separated by the word ‘crown’.
In the given crown ether the number of carbon and oxygen atoms in the ring is 12 and 6 respectively. The total number of carbon and oxygen atoms present in the ring is 18 while that of oxygen is 6. So, the name of this crown ether will be 18-crown-6.
All crown ethers will have a central cavity within the ring. Cations like potassium that can be accommodated in the cavity can interact with the unpaired electron pairs of oxygen of the ring and stabilise the crown ethers.
Hence crown ethers can dissolve ionic substances like potassium permanganate. The ring being hydrophobic can mix with nonpolar solvents. Hence the dissolved ionic compounds will be dissolved in nonpolar solvents like toluene. Crown ethers can be used to dissolve ionic compounds in nonpolar solvents also.
Nomenclature of Ethers:
The common ether nomenclature adheres to the principle of designating various alkyl/aryl groups that are connected to the oxygen atom on either side in alphabetical sequence, followed by the word ether.
For example, the name CH3 O C6H5 is given by Methyl Phenyl Ether.
Greek number prefixes like "di" are used to denote the oxygen atoms of ethers that are connected to the same group on either side. The suffix "di" is placed before the aryl/alkyl groups that are linked to the oxygen atom to give various types of ethers their names.
For example, the name C6H5 O C6H5 is known by Diphenyl Ether.
There are several rules for IUPAC ether nomenclature. The parent hydrocarbon is represented by a substituent group with more carbon atoms in accordance with IUPAC nomenclature. While the name of the other substituent group, which is connected to the same oxygen atom, begins with the prefix "oxy."
For example CH3O C2H5 is named in IUPAC as 1-Methoxy Ethane.
Let us see some common names and IUPAC names of some ethers in the tabular form
|
Formula |
Common Name |
IUPAC Name |
|---|---|---|
|
CH3 O CH3 |
Dimethyl Ether |
Methoxy Methane |
|
C2H5 O C2H5 |
Diethyl Ether |
Ethoxy Ethane |
|
C2H5 O CH3 |
Ethyl Methyl Ether |
Methoxy Ethane |
|
C6H5 O C6H5 |
Diphenyl Ether |
Phenoxy benzene |
|
C6H5 O CH3 |
Anisole or Methyl Phenyl Ether |
Methoxy benzene |
|
CH3 O CH(CH3)2 |
Methyl isopropyl Ether |
2-Methoxy Propane |
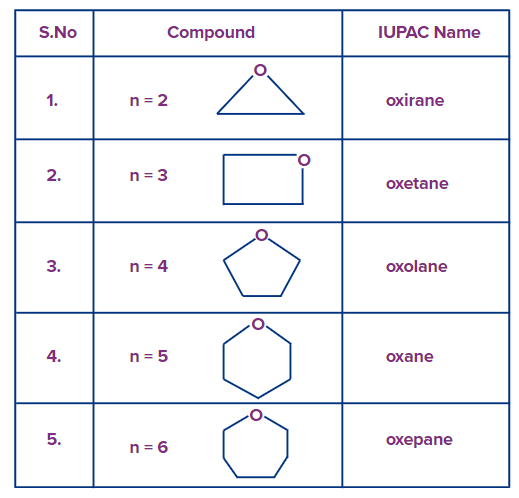
In the IUPAC nomenclature for cyclic ethers, the suffixes "irane," "etane," "olane," "ane" and "epane" are placed after the prefix "ox" to indicate the number of carbon atoms in the three-, four-, five, six- and seven-membered ring systems, respectively.
The IUPAC names and structures of the compounds are given below
Uses of Ethers:
- Ethoxyethane is the most crucial of the simple ethers due to its low price, solvent characteristics, and lack of chemical reactivity. It serves as a solvent in products like oils, gum resins, etc.
- As an inhaling anaesthetic, ethoxy ethane has also been utilised extensively.
- However, newer substances like ethrane and isoflurane have substituted ethoxy ethane as an anaesthetic due to its slow impact and unpleasant recovery period.
- Due to its high boiling point 531 K, phenyl ether, another simple ether, is utilised as a medium for transferring heat.
- Due to their practically inert nature and strong dissolution power, they are also used as a reaction medium in a variety of reactions, including the Wurtz reaction, the production of Grignard reagent, and their reactions. Additionally, it helps keep things cold.
- Tetrahydrofuran (THF), a cyclic ether, is a widely used organic chemical solvent. Due to their low boiling points, they are better suited for use as solvents since evaporation makes it simple to remove them from a molecule after a chemical process.
- Due to their pleasant aroma, a number of naturally occurring phenols and ethers, especially ring substituted anisoles, were utilised as flavouring components in perfumes. Some phenols and ethers used as fragrances and flavouring agents include anethole (a component of anise seed), eugenol (found in the oil of cloves), vanillin (present in the oil of vanilla bean), and thymol (present in thyme and mint).
Practice Problems:
Q1. As functional isomers, alcohols and ethers are a member of
(A) Distinct homologous series
(B) Identical homologous series
(C) Same functional group
(D) None of the above
Answer: (A)
Solution: Because they have a similar chemical formula but a different structure and come from distinct homologous series, alcohols and ethers are functional isomers.
Q2. What is the following compound's IUPAC name?
CH3-O-CH2-CH2-CH3
(A) Methoxy Butane
(B) Methoxy propane
(C) Ethoxy ethane
(D) Methoxy ethane
Answer: (B)
Solution: Here the parent chain is propane because a substituent group consist of three carbon atoms and another substituent group which is connected to oxygen atom has one carbon atom so it is Methoxy so the IUPAC name of the compound is Methoxy Propane or 1-Methoxy propane.
Q.3. Choose the simple ether from the options below
(A) CH3 O C2H5
(B) C6H5 O C6H5
(C) CH3 O C3H7
(D) CH3 O C6H5
Answer: (B)
Solution: It is a simple or symmetrical ether because C6H5 O C6H5 has the identical aromatic ring(phenyl ring) on both sides of the oxygen atom.
Q4. Choose the incorrect name for the following compound C6H5 O C2H5
(A) Phenetole
(B) Ethyl Phenyl Ether
(C) Ethoxy Benzene
(D) Anisole
Answer: (D)
Solution: Its common names are phenetole or ethyl phenyl ether and IUPAC name is ethoxy benzene. This substance is an aromatic ether with an ethoxy substituent. Anisole is the most common compound C6H5 O CH3 which has a methoxy substituent and its IUPAC name is methoxy benzene.
Frequently Asked Questions(FAQs):
Q1. Why do ethers react more quickly than alkanes but less quickly than alcohols and carbonyl compounds?
Answer: Ethers are considerably less reactive than alcohols and carbonyl compounds, but considerably greater reactive than alkanes.
Ethers are less reactive because they lack polarity or charge separation in the molecule. There is no transitory or partial positive charge, unlike carbonyl carbon. Therefore, there is no negative effect when nucleophiles interact with ethers (no reaction takes place). As a result, ethers are unaffected by bases, alkali metals, active methylene groups, etc.
Q2. Why do epoxides(cyclic ethers with three-membered ring) have higher chemical reactivity than simple ethers?
Answer: Epoxides are substantially greater reactive than regular ethers because of ring strain. The electrophilic carbon of the C-O bond is attacked by nucleophiles, causing the bond to break and a ring opening. Opening the ring releases the tension in it.
Q3. Why are ethers effective solvents for a wide range of organic reactions?
Answer: Alcohols are indeed H-bond donors and acceptors, but ethers can only accept H-bonds. Ethers make excellent solvents for a variety of organic compounds due to their capacity to take H-bonds and the London forces created by the alkyl groups attached to the oxygen. Ethers are a favoured solvent for many organic processes due to their low chemical reactivity. Ethers' high volatility also makes it possible for them to evaporate when separating reaction products.
Q4. Why is the dipole moment of ethers not equal to zero?
Answer: Two polar C-O bonds in the ether are angled at an angle of 111.7 degrees toward one another. As a result, the two dipoles do not cancel out, creating a net dipole moment. In other words, ether has a dipole moment of ≠0. Ethers have a higher polarity than alkenes, but a lower polarity than alcohols, esters, or amides.


