-
Call Now
1800-102-2727
Properties of Carbon: Introduction, Properties of Carbon, Practice Problems, Frequently Asked Questions
In your younger ages, you might have burned wooden items and seen them form finally a black-coloured carbon. You would have used or used a pencil even now.
Do you know, what is that central black part of the pencil that writes?
It is carbon, but a special type of carbon called graphite. Graphite has a layered structure that is held together by van der Waal forces. Because graphite slips easily between layers, it is exceedingly slippery and soft. As a result, it is utilised in pencils and as a lubricant in machinery operating at high-temperature.
Graphite is said to be the main allotropes of carbon.
In this article, we will discuss in detail the properties of carbon.
Table of Contents:
- Introduction of Carbon
- Properties of Carbon
- Practice Problems
- Frequently Asked Questions(FAQs)
Introduction of Carbon:
Carbon is a naturally occurring element found in large quantities in the Earth's crust. It has the atomic number 6 and is found in Periodic Table Group 14. Carbon is the fourth most abundant element in the universe and the fifteenth most abundant element in the Earth's crust. It is a non-metallic element. Carbon is derived from the Latin word carbo, which means "coal" or "charcoal."
Properties of Carbon:
Carbon has two electron shells, the first of which holds two electrons and the second of which holds four of the available eight spaces. When atoms bind, electrons in their outermost shell are shared. Carbon contains four vacant spaces in its outer shell, allowing it to form an octet configuration with four other atoms.
Charcoal is one of the most important carbon components and is produced when carbon is heated without the presence of air.
Apart from these properties carbon also exhibits versatile properties which we are going to discuss now
1. Catenation:
The ability of the element to bind with the same atoms of its kind to form two-dimensional (linear chain) or three-dimensional structures is known as catenation. When an element establishes bonds between its atoms to create substantial molecules, this is referred to as a property catenation. Catenation is often referred to as self-linking.
Only carbon and silicon of group14 in the periodic table show catenation.
One factor contributing to the availability of organic molecules or carbon compounds is the tendency of carbon atoms to connect with one another through covalent bonds to form long chains or rings of carbon atoms. Because it can combine with its atoms to produce the longest (linear or branched) chains, carbon has a unique property.
The longest chains, made up of millions of carbon atoms, can be found in molecules like some proteins.
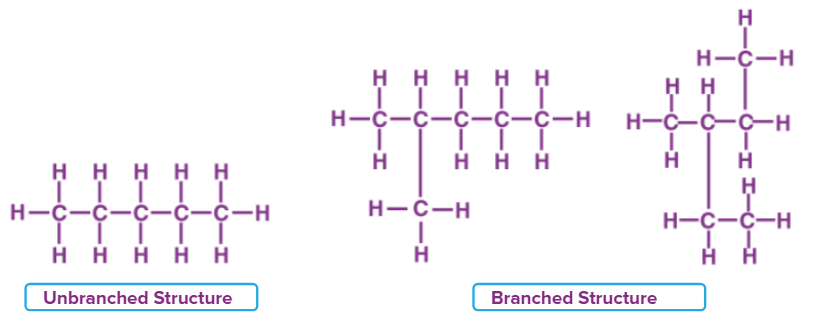
As a result of catenation, carbon forms many homologous series.
A sequence of carbon compounds with the same functional group but different numbers of carbon atoms and each subsequent member differing by ‘CH2’ in their molecular formula are referred to as a homologous series.
The alkane homologous series, which includes gases like propane, butane, ethane, and methane, is one example of it. In this series, the extra methylene bridge (-CH2) that is added to the chain causes the next members to differ in mass.
Examples: CH3CH3 Ethane
CH3CH2CH3 Propane
CH3CH2CH2CH3 Butane
CH3CH2CH2CH2CH3 Pentane
2. Allotropy:
The ability of some chemical elements to exist in two or more different forms of the same physical state is known as allotropes of the element. Allotropes are specific structural variants of an element in which the bonds between the atoms are varied.
In contrast to metals, which nearly invariably have a positive oxidation state, carbon can have an extremely wide range of oxidation states, from -4 (in CH4) to +4 (such as in CO2). Carbon is one of the few elements that has a significant number of allotropic forms. As a result, unique carbon allotropes are produced. Diamond is the most well-known and most common crystalline allotrope of carbon followed by graphite and fullerene.
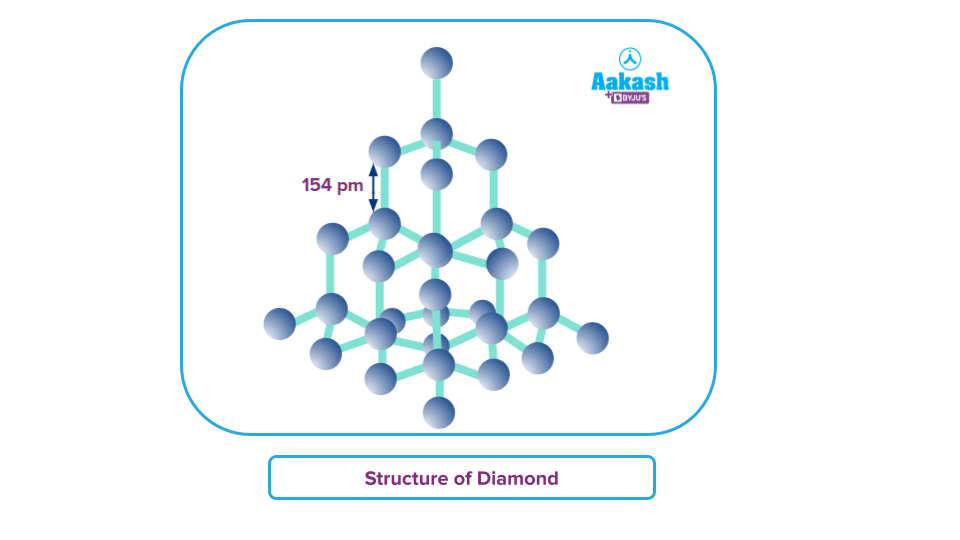
Diamond:
In its crystalline state, it is the purest form of carbon. It is composed of tetrahedrally linked carbon atoms. Each tetrahedral unit is made up of carbon bonded to four carbon atoms, which are linked to other carbons. This leads to the formation of an allotrope of carbon with a three-dimensional arrangement of C-atoms.
Each carbon atom is sp3 hybridised and forms covalent bonds with four other carbon atoms in the four corners of the tetrahedral structure.
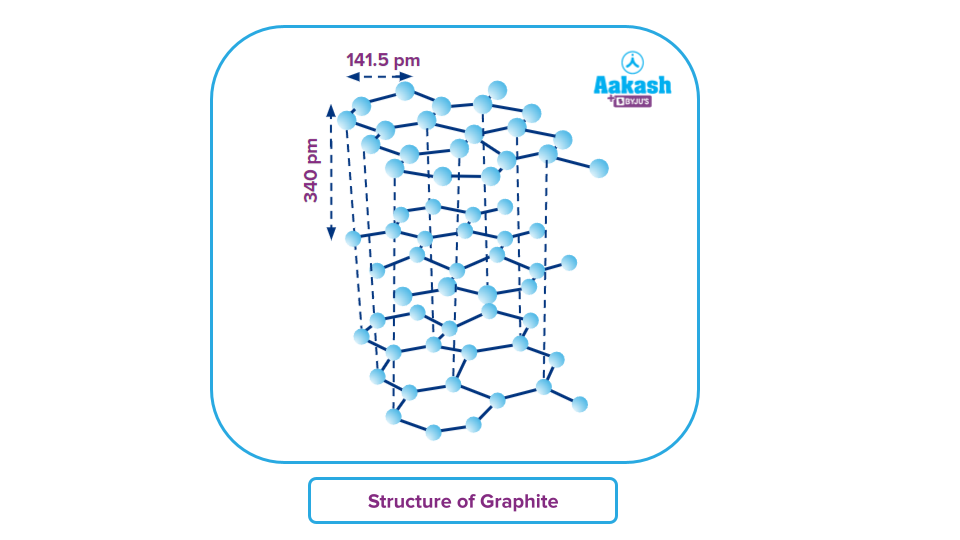
Graphite:
Two two-dimensional, flat sheets or layers of carbon atoms constitute graphite. Each layer or sheet is made up of flat hexagonal rings of carbon atoms, which can be thought of as a network of fused benzene-like rings. Each carbon atom in the layers is joined by a covalent bond to three other carbon atoms. Each hexagonal ring has a carbon-carbon bond length of 141.5 pm because each carbon atom has undergone sp2 hybridization. i.e., the formation of the sigma bond requires just three of each carbon atom's valence electrons (using sp2 hybrid orbitals). A pi bond is created by the fourth electron.
Graphite is a good conductor of heat and electricity because of the delocalized pi electrons throughout the entire sheet. A sheet (or layer) can conduct electricity, but not across parallel sheets.
The atoms of the different layers are stacked on top of one another so that they are vertically stacked underneath one another. The spacing between the layers, or the interlayer distance, is 340 pm. The interlayer gap, which is more than twice the covalent radius of carbon, is notably large. This suggests that weak van der Waal forces, rather than covalent bonds, are holding the layers together. As a result, the layers slide over one another, giving graphite its lubricating property. Because the layers are so far apart, atoms cannot pack tightly enough to properly fill the space. Consequently, graphite has a lower density than diamond.
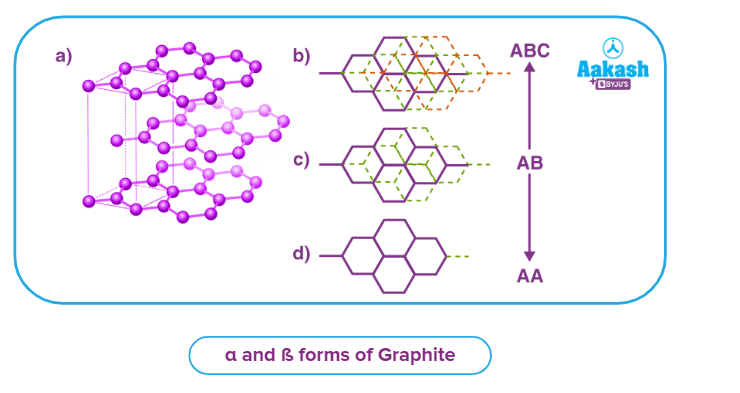
There are two forms of graphite: 𝛼 and 𝛽.
The layers are arranged in the form of a ABAB..... pattern in the 𝛼 form, with the third layer lying directly on top of the first.
The layers are arranged in the form of ABCABC...... pattern in the 𝛽 form.
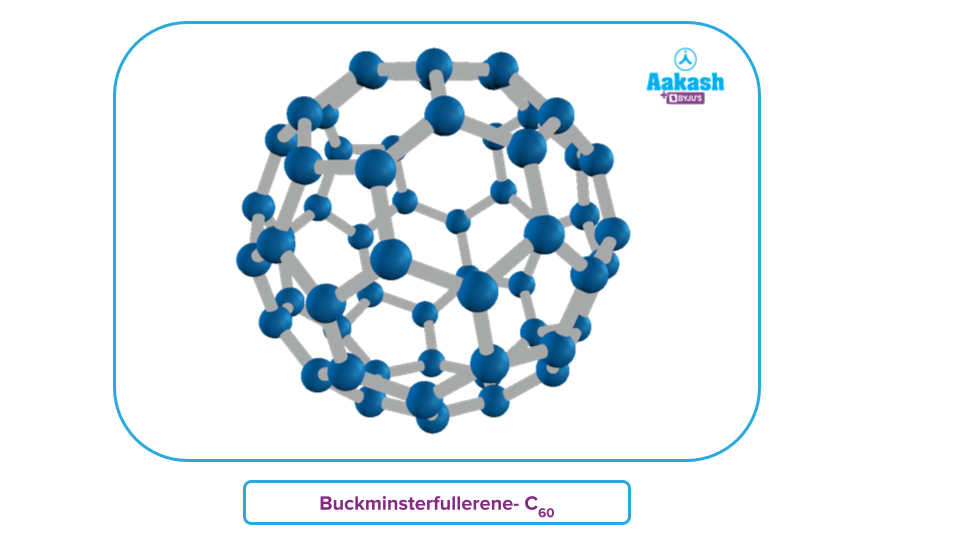
Fullerene:
Fullerenes are crystalline carbon allotropes with a smooth structure devoid of "dangling" bonds, making them the only source of pure carbon. With the chemical formula C2n, where n ≥ 30, the fullerene is a large, spherical molecule.
When graphite is heated by an electric arc in an inert atmosphere, such as He or Ar, fullerene is practically produced, but a sooty material is also produced.
The sooty substance that is produced is predominantly made of C60, with modest amounts of C70 and traces of other fullerenes. The C60 and C70 fullerenes can be entirely extracted from the fullerenes soot by extracting the fullerenes with benzene or toluene, followed by chromatography with alumina.
The Buckminsterfullerene C60 molecule resembles a soccer ball, and fullerenes are cage-like compounds. The 60 vertices of the saucer-shaped fullerene molecule contain one carbon atom each. It has 20 six-membered rings and 12 five-membered rings.
3. Tetravalency:
Because of its atomic number of 6, the carbon atom possesses a total of 6 electrons. Its electronic configuration can be expressed as 2,4. This indicates that the valence shell contains four electrons. Carbon needs four electrons to reach the inert gas electronic configuration, which is stable. In order to form four covalent bonds with other atoms, carbon adheres to the octet rule. As a result, carbon is tetravalent, which means it has a valency of four and has the ability to form four covalent bonds with both other atoms and carbon atoms. This is known as carbon tetravalency.
Because of the extraordinary ability of carbon to establish exceptionally strong covalent bonds, carbon molecules are remarkably stable. The fact that carbon has a valency of four, which is a relatively large number, is another factor contributing to the abundance of organic chemicals or carbon compounds. A carbon atom may form covalent bonds with a wide variety of other atoms, including hydrogen, oxygen, nitrogen, sulphur, and many more, due to its large valency of 4. The result is the formation of many compounds.
4. Formation of p-p bond:
Carbon has a great ability to make p-p bonds, either with other atoms of the same size or with itself. This is caused by carbon's small size and high electronegativity. When it comes to carbon, there is the greatest amount of overlap that can lead to the formation of numerous bonds. i.e. In C=C, the carbon 2p orbital overlaps with the carbon 2p orbital in a sideways manner.
Practice Problems:
Q1. Various compounds are formed by carbon as a result of
(A) Variable valency
(B) Catenation property
(C) Large chemical affinity
(D) Allotropy
Answer: (B)
Solution: Due to the unique ability of carbon, which has the property of catenation, to form bonds with other carbon atoms, a large number of molecules and compounds containing long carbon chains, branched carbon chains, or carbon atoms arranged in rings and connected by single, double, or triple bonds are created.
Q2. Among the following, which is a good conductor of electricity?
(A) Diamond
(B) Graphite
(C) Coal
(D) None of these
Answer: (B)
Solution: Each carbon atom in graphite has an unpaired electron, which makes it act as a good conductor of electricity.
Q3. Which of the given statements about carbon is false?
(A) Diamond is one of the carbon allotrope
(B) It has crystalline and amorphous allotropes
(C) It cannot form p-p bond with other carbon atom
(D) It cannot form p-d bonds due to the absence of vacant d-orbitals
Answer: (C)
Solution: Diamond is one of the carbon allotropes and carbon has both crystalline (diamond,graphite and fullerene) and amorphous allotropes(caol, charcoal and animal charcoal) and in the 14th group except carbon every element forms p-d bonds because carbon does not contain vacant d-orbitals. But carbon forms p-p with other carbon atoms.
Q4. What is carbon’s hybridization in fullerene?
(A) sp2
(B) sp
(C) sp3
(D) None of these
Answer: (A)
Solution: In fullerene, the hybridization of the carbon is sp2 and in graphite it is sp2 and diamond it is sp3.
Frequently Asked Questions:
Q1. Define radiocarbon dating.
Answer: Carbon dating is a technique for determining the age of any substance that contains carbon based on the amount of unstable carbon isotopes (mostly carbon-14) that are present in the substance. The stable isotopes are produced when the unstable isotopes decay over time, changing the total proportion of the isotopes in the sample being studied.
Q2. Why are the majority of carbon compounds not good electrical conductors?
Answer: The valence shell of carbon contains four electrons, making it a tetravalent compound. For the formation of covalent bonds, all four of these electrons are available.
All organic molecules, including those containing carbon, are typically poor electrical conductors. Since carbon has four valence electrons, it can create four covalent bonds. Therefore, carbon compounds do not have an additional electron that would conduct electricity. They lack free electrons. Carbon-compounds do not conduct electricity but graphite which is a allotrope of carbon electricity due to availability of one unpaired electron.
Q3. What role does CO2 play in the greenhouse effect?
Answer: The gas carbon dioxide is one of the elements that constitute air. Carbon dioxide behaves as a pollutant when it is present in excess in the atmosphere. Plants use atmospheric carbon dioxide for photosynthesis, which reduces the amount of carbon dioxide in the air. Deforestation results from more trees being cut down. Deforestation causes an increase in the amount of carbon dioxide in the atmosphere. Additionally, it enhances the greenhouse effect.
Q4. What are the different isotopes of carbon?
Answer: Carbon is made up of three isotopes, For instance, while612C and613C are stable isotopes of carbon,614C is a radioactive isotope of carbon. Both of these stable carbon isotopes contain varying proportions of carbon. While carbon-12 contains 98.93% pure carbon, carbon-13 has around 1.07% carbon.


