-
Call Now
1800-102-2727
Phenol: Structure, Preparations, Properties, Classification, Chemical Reactions, Uses, Practice Problems and Frequently Asked Questions
Approximately two-thirds of all phenol produced globally is used to make reagents for the plastic manufacturing industries.
We can't imagine living without plastic.
Most of the things we encounter are either made of plastics or contain plastic components. Bisphenol A is produced by the condensation reaction of phenol and acetone, and it is widely used in the polymer industry to synthesize various epoxide resins and polycarbonates.
Phenolic resins are commercially prepared through the polymerization of phenol with formaldehyde. The resulting resin is known as phenol-formaldehyde resin, and it is commercially known as bakelite.
Bakelite is widely used in electrical switches and automobiles due to its ability to withstand extreme heat and resist electricity and other chemicals. The intermediate formed during the polymerization reaction is known as novolac; it is a resin that is widely used as a binding agent or adhesive in a variety of industries. Novolac is also used as a protective coating.
Table of content
- Structure of phenol
- Preparations of phenol
- Properties of phenol
- Natural sources of phenol
- Classification of phenol
- Test for phenolic group
- Chemical Reactions of phenol
- Uses of phenol
- Practice problems
- Frequently asked questions
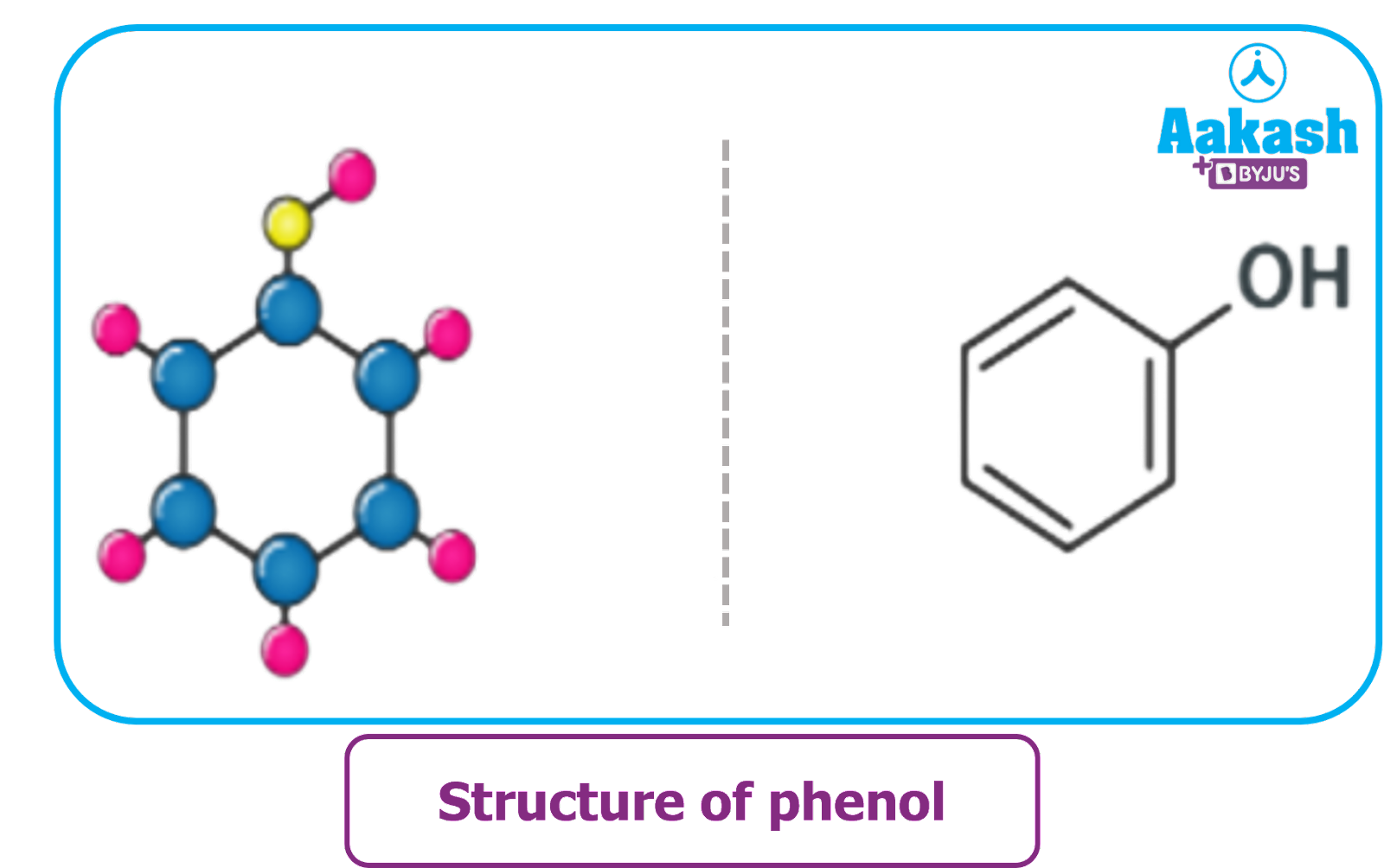
Structure of phenol:
Preparations of phenols
Phenol can be synthesized by the following methods.
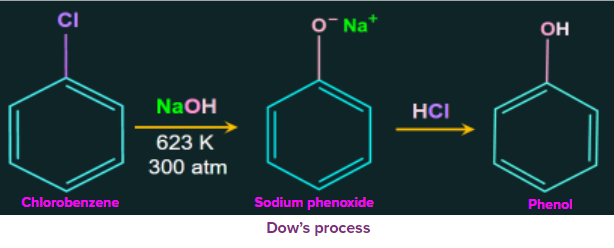
1. Dow’s process:
Dow’s process is a method to prepare phenol. The reactant chlorobenzene is heated with aqueous sodium hydroxide at temperatures of 623 K and 300 atm to get sodium phenoxide ion. Then in the next step sodium phenoxide ion is treated with dil. HCl which gives the final product as phenol.
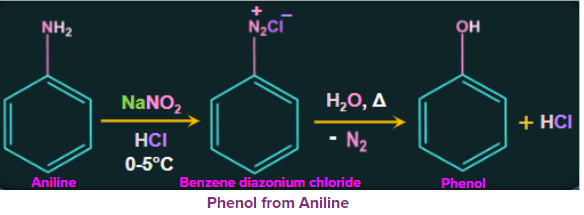
2. From Aromatic primary amines
Treatment of aromatic primary amines at 273-278 K with freshly prepared nitrous acid
(NaNO2+HCl) gives the diazonium salt. These diazonium salts are inherently very reactive. When heated with water, these diazonium salts eventually hydrolyze to phenol. Phenol can also be obtained from diazonium salts by treatment with dilute acids.
3. Phenol production from cumene
Cumene is an organic compound obtained by Friedel-Crafts alkylation of benzene with propylene. Cumene hydroperoxide is obtained from the oxidation of cumene (isopropylbenzene) in the presence of air. Further treatment of cumene hydroperoxide with dilute acid gives phenol. Acetone is also produced in large quantities as one of the by-products of this reaction. Therefore, the phenol produced by these processes needs to be purified.

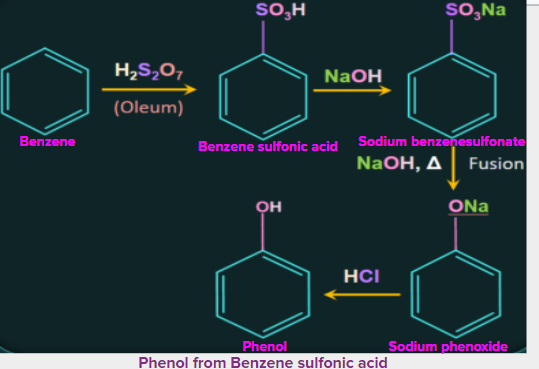
4. Production of phenol from benzenesulfonic acid
Benzenesulfonic acid can be obtained from benzene by reaction with oleum. This benzenesulfonic acid can be treated with molten sodium hydroxide at high temperatures to promote the formation of sodium phenoxide. Finally, sodium phenoxide produces phenol during acidification.
Properties of phenol
|
C6H6O |
Phenol |
|
Molecular Weight/ Molar Mass |
94.11 g mol-1 |
|
Density |
1.07 g cm-3 |
|
Melting Point |
40.5 oC |
|
Boiling Point |
181.7 oC |
1. Boiling point of phenol
Phenol generally has a higher boiling point compared to other hydrocarbons of the same molecular weight. This is because there are intermolecular hydrogen bonds between the hydroxyl groups of the phenol molecule.
2. Solubility of phenol
The solubility of phenol in water depends on the hydroxyl groups present. The hydroxyl groups of phenol are involved in the formation of intermolecular hydrogen bonds. Hydrogen bonds are formed between water and phenol molecules, making phenol water-soluble. However, the aryl group attached to the hydroxyl group is hydrophobic in nature. Therefore, as the size of the aryl group increases, the solubility of phenol decreases.
3. Acidity of phenol
Phenol reacts with active metals such as sodium and potassium to form the corresponding phenoxide. These reactions of phenol show its acidic nature. In phenol, the SP2 hybrid carbon of the benzene ring is directly attached to the hydroxyl group and functions as an electron-withdrawing group. Therefore, the electron density of oxygen decreases.
Phenoxide ions are more stable than alkoxide ions because the negative charge of the oxygen is delocalized over the benzene ring. As a result, phenol is more acidic than alcohol. In substituted phenols, the acidity decreases when an electron-donating group is attached to the ring, and increases when an electron-withdrawing group is attached.
Natural sources of phenol
Phenol is a component of coal tar and is produced when organic matter decomposes. Elevated phenol levels in the environment can be caused by forest fires. It has been detected in the volatile components of fertilizer.
Industrial sources of phenol and other related aromatic compounds are petroleum refineries, petrochemicals, basic organic chemicals manufacturing, coal refining, pharmaceuticals, tanning factories, pulp and paper mills.
Classification of phenols
Phenol is an organic compound containing at least one -OH group directly attached to the benzene ring. Phenol can be divided into monovalent, divalent and trivalent phenols depending on the number of hydroxyl groups attached to the benzene ring.
Monovalent Phenol:The simplest member of this series is hydroxybenzene, commonly known as phenol, while others are called substituted phenols. The three isomeric hydroxy toluenes are ortho cresol, meta cresol and para cresol.
Dihydric phenol: Dihydroxybenzenes (benzene diols) are organic chemical compounds in which two hydroxyl groups are substituted onto a benzene ring.
Dihydroxybenzene- 3 isomers of dihydroxybenzene, namely catechol, resorcinol, and quinol, are well known by their common names.
Trihydric phenol: Trihydroxybenzene (benzene triols) are organic chemical compounds in which three hydroxyl groups are substituted onto a benzene ring.
Trihydroxybenzene- 3 isomers of trihydroxybenzene are known by their common names pyrogallol, hydroxyquinol, and phloroglucinol.
Test for phenolic group
1. Litmus Test
Purpose: The purpose of the litmus test is to determine the presence of a phenol functional group in a given material.
Conjecture: Litmus paper changes color depending on whether the solution is acidic or basic in nature. Blue litmus paper changes to red in acidic solutions. Red litmus paper changes to blue in basic solutions. Because phenols are acidic in nature, they pass the litmus test.
Materials needed: blue Litmus paper, a dropper, a test tube, and so on.
Procedure: Place 1-2 drops of the given solution from the test tube and place it on the blue litmus paper.
Remark: The color of the litmus paper shifts from blue to red.
Outcome: The given solution may include phenol as an Outcome.
2. The Liebermann Test
This test is performed by phenols with a free para position.
Purpose: The goal of this test is to detect the presence of a phenol functional group in a given material.
Conjecture: When phenol is dissolved in conc. Sulfuric acid and sodium nitrite are added. The sodium nitrite then interacts with concentrated sulfuric acid to produce nitrous acid and sodium sulfate in this combination. Nitrous acid now interacts with phenol to form p-nitrosophenol. This p-nitrosophenol with an excess of phenol generates an indophenol complex, which is red in color when diluted. When the indophenol complex combines with a strong alkali (NaOH), a blue indophenol anion is formed. The reactions that are involved can be written as follows ;
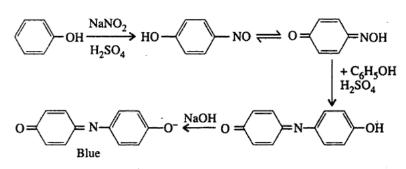
Materials needed: included a test tube, a test tube holder, the provided organic chemical, concentrated sulfuric acid, sodium hydroxide, sodium nitrite, distilled water, and a burner.
Procedure:
1. Begin with a clean, dry test tube.
2. Fill it with a little amount of sodium nitrite crystals.
3. Now add 100 mg of the provided phenolic compound and gently heat the test tube for 30-35 seconds.
4. Set aside the test tube to cool.
5. Add 1 mL of the concentrated sulfuric acid and gently shake the test tube to combine the contents.
6. Examine the color change and dilute the solution with water.
Remark : When we dilute the solution with water, it gets red, but when we add NaOH to it, it turns blue (deep blue).
Outcome: The presence of phenol in the provided sample is confirmed by the deep blue color.
Chemical reactions of phenol
1. Nitration of phenol
Phenol is nitrated when treated with dilute nitric acid at a low temperature (298 K) to give a mixture of ortho-nitrophenol and para-nitrophenol. Due to its volatility, the resulting mixture is further separated into ortho-nitrophenol and para-nitrophenol by steam distillation.
Due to the absence of intermolecular hydrogen bonds, ortho nitrophenols are less volatile than para nitrophenol, which contain only intermolecular hydrogen bonds, while ortho nitrophenols contain intramolecular hydrogen bonds.
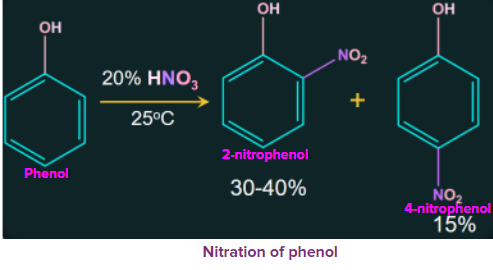
When phenol is treated with concentrated nitric acid, the nitration results in the formation of 2, 4, 6-trinitrophenol (commonly called picric acid) along with many oxidized products.

2. Halogenation of Phenols
Due to the strong activating effect of the hydroxyl groups in phenol, they are halogenated in the absence of Lewis acid. Treatment of phenol with bromine at low temperatures in the presence of low polar solvents such as CS2 or CHCl3 results in the formation of mono bromophenol.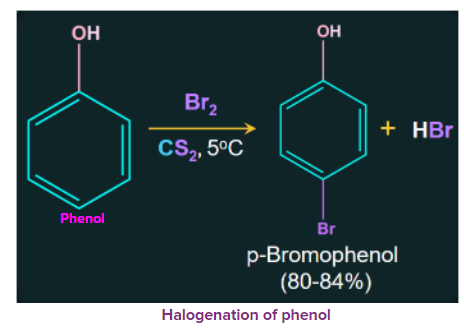
When phenol is treated with bromine water, a white precipitate of 2, 4, 6-tribromophenol is formed.

3. Kolbe's reaction
Treatment of phenol with sodium hydroxide produces phenoxide ions. The formed phenoxide ions are highly reactive to electrophilic substitution reactions and then treatment with a weak electrophile (carbon dioxide) causes an electrophilic substitution reaction to form ortho hydroxybenzoic acid followed by acidification. This reaction is commonly known as the Kolbe reaction.

Uses of phenol
- Phenol's capacity to establish strong hydrogen bonds increases its water solubility.
- Phenols are frequently utilized in home goods and as industrial synthesis intermediates.
- Phenol is used as a disinfectant in home cleaners and mouthwash at low concentrations.
- Phenol is a beginning ingredient in the manufacturing of plastics, explosives like picric acid, and pharmaceuticals like aspirin.
- The photographic developer contains phenol hydroquinone, which converts exposed silver bromide crystals to black metallic silver.
- In the dye business, several substituted phenols are utilized to create highly colored azo dyes.
- Phenol is present in vaccines as a preservative. It prevents germs from developing in the vaccination solutions and contaminating them.
- Phenol is included in several throat sprays, which can relieve symptoms of a sore throat or mouth irritation caused by tiny ulcers.
- Muscle spasticity is a problem that can be treated by phenol injections. This occurs when your brain's communication with nerves and spinal cord is disrupted. Your muscles will get tense as a result of it.
- To separate DNA, RNA, and proteins and isolate them in their purest form, liquid phenol is commonly mixed with chloroform.
- Carbolic soap is a kind of soap that includes compounds derived from phenol. It's been used as an antiseptic during surgery.
- Antioxidants are known to exist in plant-based substances containing phenol. This means they can block free radicals from reacting with other molecules in your body, reducing DNA damage and long-term health consequences.
Practice problems
Q.1. Out of these, which test is used to distinguish alcohol and phenol?
(A) Lucas Test (B) Ferric chloride test
(C) Tollen Test (D) None of the above
Answer : (B)
Solution: Ferric Chloride test: Alcohols are readily differentiated from phenols using this test. Addition of a drop or two of ferric chloride solution to a sample of phenol (3-4 drops) will produce a distinct violet/purple coloration. Alcohols do not produce such deep coloration when treated with ferric chloride solution.
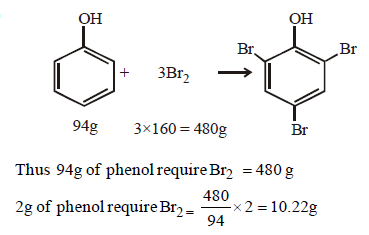
Q.2. What amount of bromine will be required to convert 2 g of phenol into 2, 4, 6-tribromophenol?
(A) 4.0 (B) 6.0
(C) 10.22 (D) 20.44
Answer : (C)
Solution:
Q.3. Which one of the following properties is exhibited by phenol ?
(A) It is soluble in aq. NaOH and evolves CO2 with
aq. NaHCO3.
(B) It is soluble in aq. NaOH and does not evolve CO2with
aq. NaHCO3.
(C) It is not soluble in aq. NaOH but evolves CO2 with
aq. NaHCO3.
(D) It is insoluble in aq. NaOH and does not evolve CO2 with
aq. NaHCO3.
Answer : (B)
Solution: 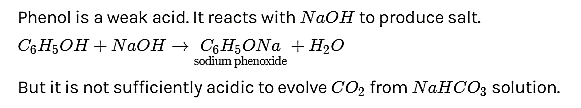
Q.4. Which one of the following phenols has the highest PKa value?
(A) O-Nitrophenol (B) P -Cresol
(C) m-Nitrophenol (D) Picric acid
Answer : (B)
Solution: Phenol is acidic because of the presence of the electron-withdrawing benzene ring as it stabilizes the phenoxide ion formed after the removal of proton. On the other hand, electron- donating group decrease the acidic strength of phenol. More acidic strength, less PKa value. Less acidic strength, more PKa value.Cresol (C7H8O), any of the three methyl phenols with the same molecular formula but having different structures: ortho- (o-) cresol, meta- (m-) cresol, and para- (p-) cresol have the more PKa value than others because of the presence of electron-donating group methyl, hence P-Cresol will have the highest PKa value.
Frequently asked questions
1. Which is more reactive, alcohol or phenol?
Answer: Phenol is more reactive because it rapidly loses H+ and produces resonance-stabilized phenoxide ions. Alcohol, on the other hand, produces the alkoxide ion RO- which is unstable due to the + I effect of the R group and increases the electron density on the oxygen atom.
2. Why does phenol produce tribromophenol when brominated with bromine water?
Answer: Bromonium ions can easily be formed in the polar solvent and also stabilized by polar solvent. Thus, the formation of strong ortho and para-directed groups and the stability of Br+ enhance the formation of Tribromophenol.
3. Which groups increase the acidity of phenol?
Answer: -NO2,-CN,-X(halogen) group increases the acidity of phenol. These are electron-withdrawing groups that stabilize the negative charge of phenoxide ions. This facilitates the removal of protons.
4. Is it safe to use phenols?
Answer: Phenol exposure can irritate the skin, eyes, nose, throat, and neurological system. Weight loss, weakness, fatigue, muscular pains, and discomfort are all signs of phenol exposure. Severe exposure can result in liver and kidney damage, as well as skin burns, tremors, convulsions, and twitching.


