-
Call Now
1800-102-2727
Pericyclic Reaction
Alchemists believed that by transformation of some basic material into a higher form, it was possible to turn lead into gold, cure ailments, and extend human life. But later, scientists concluded that pure science is involved for such chemical reactions to occur, but no magic. Still there is a class of reactions which occurs just in presence of heat or light and requires no other solvent or reagent for reaction to proceed. This class of reactions are known as pericyclic reactions.

Pericyclic reactions are involved in major physiological processes like non-enzymatic, photochemical electrocyclic ring opening and a (1,7) sigmatropic hydride shift in vitamin D synthesis.
Cycloaddition reactions applicable in anticancer and antiviral compounds.
1,3-dipolar cycloaddition reactions of selected 1,3-dipoles, such as azides, nitrones, and nitrile oxides, are reviewed in the light of their application in the preparation of key intermediates for antiviral synthesis. A few examples of [2+2] cycloaddition reactions are also presented. The products obtained from these pericyclic reaction approaches were all tested for their activities in terms of blocking the virus replication, and the relevant biological data are highlighted.
Table of content
- Introduction
- Characteristics of Pericyclic reactions
- Types of Pericyclic reactions
- Electrocyclic reaction
- Cycloaddition reaction
- Sigmatropic rearrangement
- Group transfer reaction
- Practice Problems
- Frequently Asked Question-FAQs
Introduction
Pericyclic reactions are described as reactions in which electrons undergo a concerted cyclic shift. A pericyclic reaction is defined by two important features as defined by the above definition.
The first point is that there is a concerted reaction which means reactant bonds are broken and product bonds are formed simultaneously. The final product is made without the formation of intermediates. In pericyclic processes, the second crucial point involves a cyclic electron shift. Pericyclic is a word that means "around the circle." The term is derived from the cyclic shift of electrons. As a result, pericyclic reactions are characterized by the and - bonds, involved in a cyclic transition state.
Characteristics of Pericyclic reactions
1. Pericyclic reactions don’t require solvents.
2. They occur in absence of any reagent.
3. They occur in the presence of heat or light only.
4. No intermediate is formed.
5. Involves a cyclic transition state.
6. These are stereospecific in nature.
Types of Pericyclic reaction
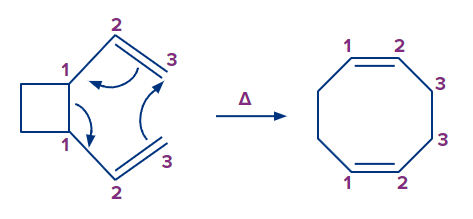
Let's have a look at four different forms of pericyclic reactions. The first sort of reaction is the electrocyclic reaction, which involves the closure or opening of a ring at the expense of a conjugated double or triple bond.
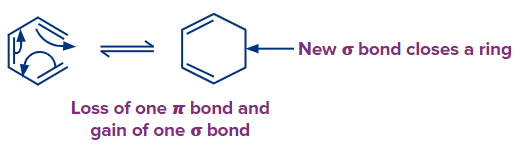
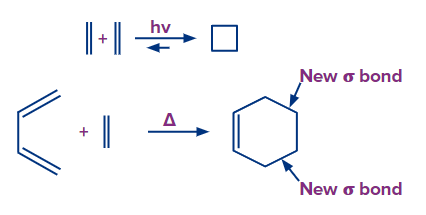
The cycloaddition reaction is the second type of reaction. It occurs when two or more electron systems react to form a ring at the expense of one bond in each of the reacting partners. This reaction results in the production of two new bonds, which closes a ring. In total, two bonds are lost in reactants and two bonds are gained in the product.
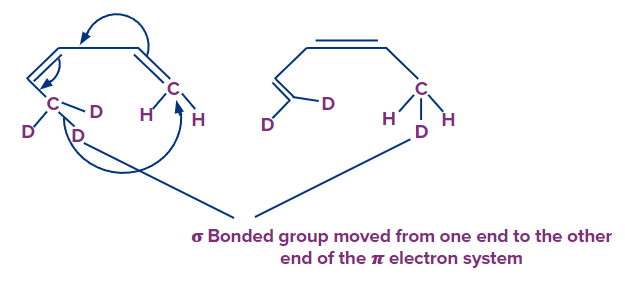
The sigmatropic rearrangement (or reaction) is the third type of reaction: A process in which a bond formally migrates from one end of the electron system to the other while the net number of bonds remains unchanged.
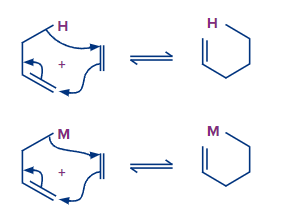
The group transfer reaction is the fourth type of reaction: One or more groups or atoms transfer from one molecule to another in this reaction. Both molecules are bonded together in this reaction by a () bond.
Let us see each type of pericyclic reaction in detail:
1. Electrocyclic reaction
The concerted interconversion of a conjugated polyene and a cycloalkene is known as an electrocyclic reaction. Thermally or photochemically driven electrocyclic reactions are possible.
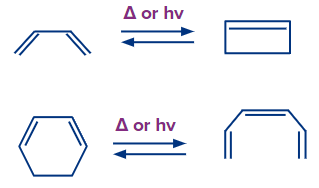
Reversible reactions characterize all electrocyclic reactions. The reaction's open-chain partner is invariably a conjugated system, but the other cyclic partner may or may not have a conjugated system.
In electrocyclic processes, a ring is produced when a new bond is established and a bond is lost (i.e., gain of one bond and loss of one bond) or a ring is broken when one bond is lost and another is gained.
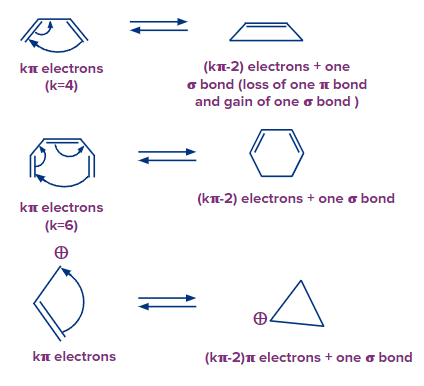
As a result, electrocyclic processes can be divided into two categories:
1. Electrocyclic ring opening,
2. Electrocyclic conjugated system closure.
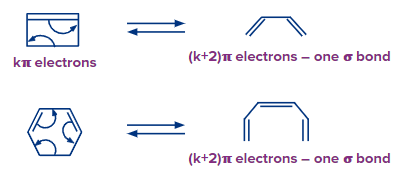
If the open-chain partner's system includes k electrons, the equivalent cyclic partner contains (k – 2) electrons and one additional bond in electrocyclic ring closure (or ring-closing electrocyclic reaction). If the ring partner has k electrons, the open-chain partner will have (k + 2) electrons with the loss of one bond in a ring-opening electrocyclic process.
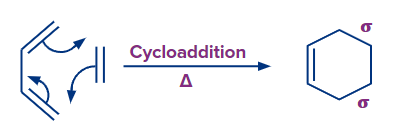
2. Cycloaddition Reaction
A cycloaddition is a process that involves the addition of two unsaturated molecules to produce a cyclic product. The formation of a cyclic product requires the loss of one (pi) bond from each reactant and the gain of two sigma bonds respectively.
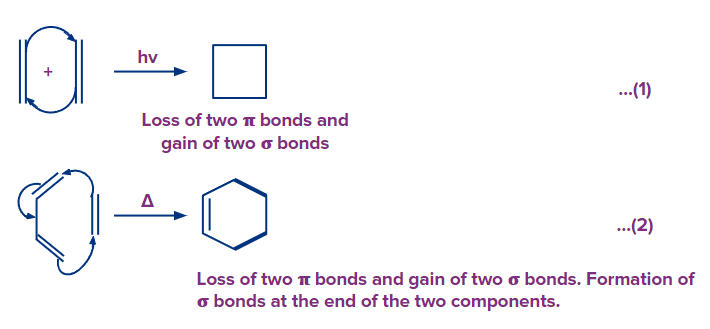
Because the reaction in equation (1) involves two electrons from one reactive component and two electrons from the other, it is called a [2 + 2] cycloaddition reaction.
Similarly, A [4 + 2] cycloaddition is the reaction in equation (2).
Photo-Induced [2 + 2] Cycloaddition Reactions:
When UV radiation is used to irradiate ethylene, one electron is promoted from the 1 to the 2* orbital in some, but not all, of the molecules. The end result is a mixture of ethylene molecules in the ground and excited states.
As a result, photo-induced cycloaddition occurs between the photochemical HOMO of one molecule and the ground state LUMO of another. Because the cycloaddition reaction is a concerted reaction, stereochemical integrity is preserved.
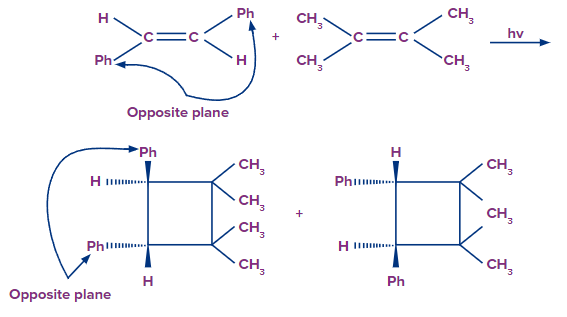
Example-
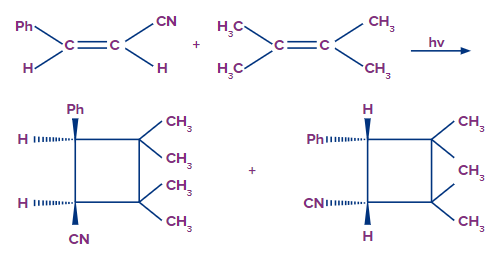
4+2 Cycloaddition:
The most well-known [4 + 2] cycloaddition reaction is the Diels-Alder reaction. This is a thermally permitted reaction. Photochemically, the Diels-Alder process is prohibited. Let's start with the general description of the Diels-Alder reaction, which is the most prevalent [4 + 2] cycloaddition reaction. Diels-Alder reactions take place when a conjugated diene reacts with an alkene (or alkyne), which is referred to as the dienophile.
Dienes
The Diels-Alder reaction involves a reaction between an electron-rich diene and an electron-poor dienophile. There have been some Diels-Alder reactions with electron-poor dienes and electron-rich dienophiles, but these are uncommon. Simple dienes, such as 1,3-butadiene, have enough electrons to be useful in the Diels-Alder reaction. The presence of electron-releasing groups like alkyl, phenyl, or alkoxy groups may increase the reactivity of dienes even further.
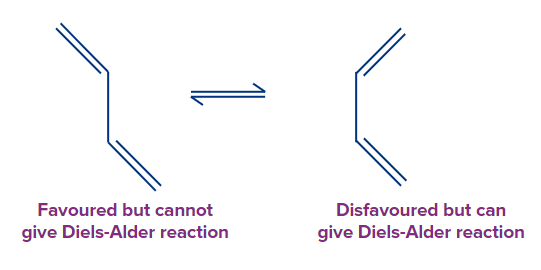
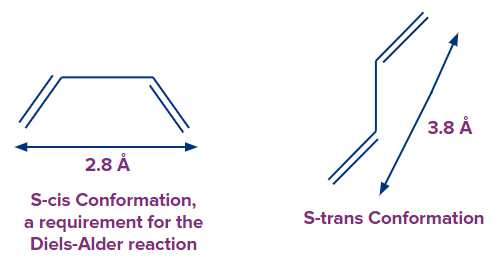
The Diels-Alder reaction's diene component can be open-chain or cyclic, but it must be in s-cis conformation. For steric reasons, butadiene usually favors the s-trans conformation, with the two double bonds as far apart as feasible. Rotation around the central bond has a low energy barrier (approximately 30 kJ/mol at 25°C), and rotation to the less favourable yet reactive s-cis conformation is quick.
For cyclic dienes that are always in the s-cis configuration, the Diels-Alder reaction is ideal.
The Dienophile
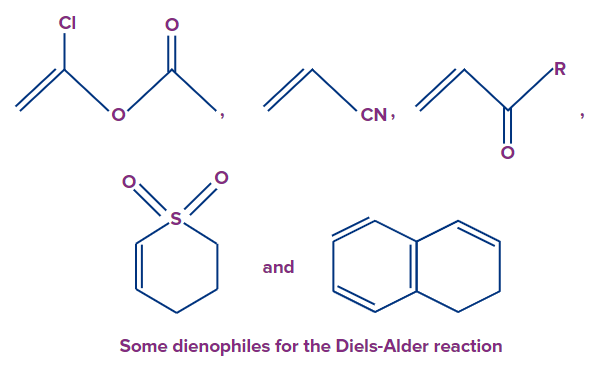
The most common dienophiles are electron-poor alkenes and alkynes. Dienophiles (lovers of dienes) are electron-deficient alkenes and alkynes that react readily with a diene. Dienophiles are simple alkenes and alkynes like ethylene and acetylene, for example. In a good dienophile, one or more electron-withdrawing groups move electron density away from the link. Dienophiles that undergo the Diels-Alder reaction include conjugated carbonyl compounds, nitro compounds, nitriles, sulphones, arylalkenes, arylalkynes, vinyl ethers, vinyl esters, haloalkenes, and dienes.
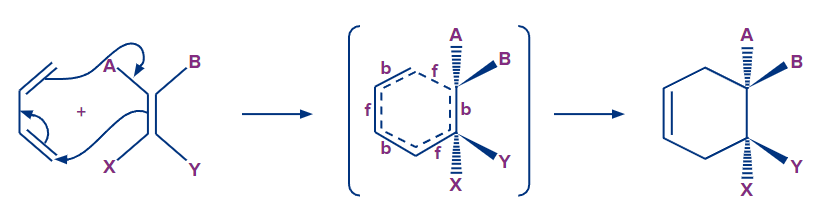
Mechanism
The Diels-Alder reaction is a cyclic movement of six electrons in which four are in the diene and two are in the dienophile. The mechanism depicted in the diagram below is fairly realistic. This is referred to as a coordinated reaction since all of the bonding takes place at the same time. At the same time, the bond breakdown happens. However, for these three pairs of electrons to move at the same time, the transition state must have a geometry that allows the diene's two end p-orbitals to overlap with the dienophiles.
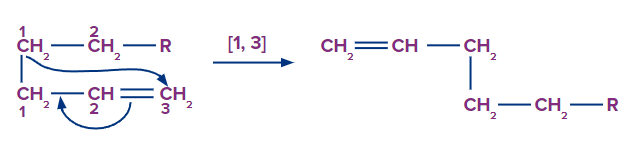
3. Sigmatropic rearrangement Another type of coordinated pericyclic reaction guided by orbital symmetry is sigmatropic rearrangements. A group linked by a (sigma) bond migrates to the terminus of a neighboring -electron system during this rearrangement, which requires a coordinated remodeling of electrons. Because a (sigma) bond appears to migrate from one place to another during the reaction, it is called sigmatropic rearrangement. The (pi) electrons are all shifting at the same time. The number of (pi) and (sigma) bonds are both unchanged.
The following examples best show the method used to classify sigmatropic rearrangement:
The substrate in sigmatropic rearrangement is separated into two parts: the alkenyl (or polyalkenyl) chain and the migratory group. For sigmatropic rearrangement, all substrates have at least one allylic carbon in the alkenyl chain.
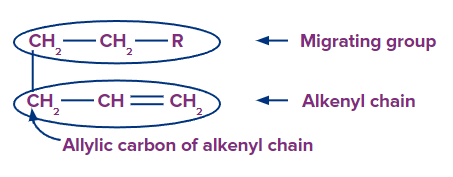
Alkenyl Chain Numbering
The numbering of alkenyl chains always begins with the allylic carbon, which is numbered-1.
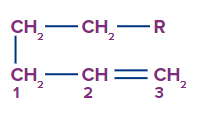
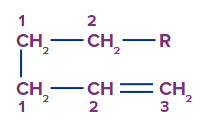
Migrating Group Numbering
Number-1 is usually assigned to an atom (H, C, or heteroatom) of a migration group that is connected to allylic carbon by a (sigma) bond.
In the first case, the migratory group's atom-1 migrates to the alkenyl chain's atom-3. As a result, this rearrangement is characterised as a sigmatropic [1, 3] rearrangement.
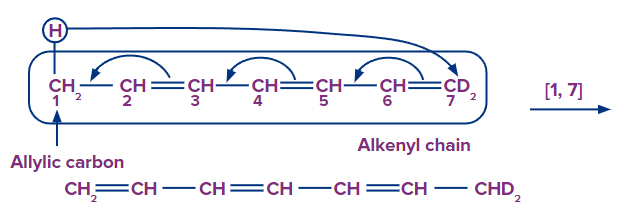
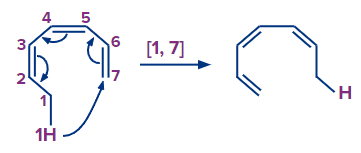
The reaction shown above is an example of [1, 7] sigmatropic rearrangement.
In the rearrangement, the initial atom of the migratory group does not always become connected to the alkenyl chain.
Consider the following illustration:
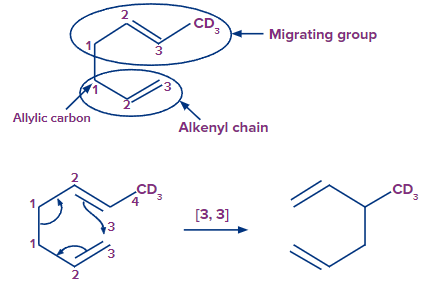
The migratory group's atom-3 migrates to the alkenyl chain's atom-3 in this rearrangement. As a result, this rearrangement is a [3, 3] sigmatropic rearrangement.
Types of sigmatropic rearrangement:
1. [1,3] sigmatropic rearrangement
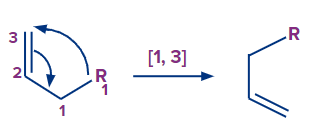
2. [1,5] sigmatropic rearrangement:
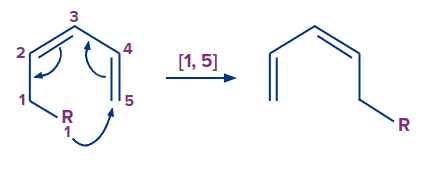
3. sigmatropic rearrangement.
4. Group transfer reaction
Group transfer reaction is a pericyclic process that involves the transfer of one or more groups or atoms from one molecule to another. In this class, there are only a few reactions. One of the most prevalent group transfer reactions is the ene reaction. The reduction of alkenes and alkynes by diimide is another well-known group transfer process.
The thermal reaction of an alkene (named ene) with an allylic hydrogen with a molecule with multiple bonds (X Y, X Y), called enophile, is known as the ene reaction.
Transfer of allylic hydrogen (1, 5 migration of hydrogen), shift of allylic double bond, and bonding between two unsaturated termini (one terminus of ene and the other terminus of enophile) all occur together throughout the reaction, resulting in a 1: 1 adduct.
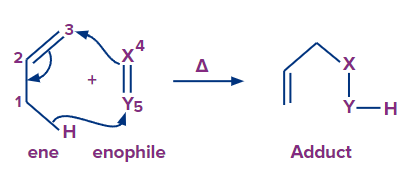
Here, X= Y is C= C, C =O, C =S, C =N, N= O , —N =N— , O= O and X Y is C C, C N.
Practice Problems
1. The given reaction is which type of reaction?
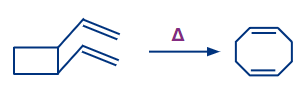
A). [3, 3] sigmatropic rearrangemen
B). [1, 3] sigmatropic rearrangement
C). [1, 5] sigmatropic rearrangement
D). None of the above
Answer: A
Solution: The given reaction is an example of [3, 3] sigmatropic rearrangement.
2. In cycloaddition reaction, the difference in the number of sigma bonds of reactant and product is:
A. 1
B. 2
C. 3
D. 4
Answer: B
Solution: Cycloaddition reaction can be characterized by Δσ = 2, (where Δσ is the difference of the number of sigma bonds formed in the product and the number of sigma bonds that were broken in the reactant).
3. Which of the following is true about the electrocyclic reactions?
A) These are reversible reactions
B) They may occur in presence of heat or light
C) A ring is produced from the conjugated acyclic system at a expense of one - bond
D) All of the above
Answer: D
Solution: Reversible reactions characterize all electrocyclic reactions. In electrocyclic processes, a ring is produced when a new bond is established and a bond is lost (i.e., gain of one bond and loss of one bond) or a ring is broken when one bond is lost and another is gained. Thermally or photochemically driven electrocyclic reactions are possible.
4. Which of the following are dienophiles?
A)
B)
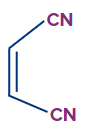
C)
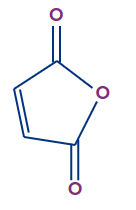
D) All of the above
Answer: D
Solution: Dienophiles (lovers of dienes) are electron-deficient alkenes and alkynes. Dienophiles that undergo the Diels-Alder reaction include conjugated carbonyl compounds, nitro compounds, nitriles, sulphones, arylalkenes, arylalkynes, vinyl ethers, vinyl esters, haloalkenes, and dienes.
Frequently Asked Questions-FAQs
1. What are stereospecific reactions?
A stereospecific reactions are those reactions in which reactant determine the stereochemistry of the reaction giving a single major product.
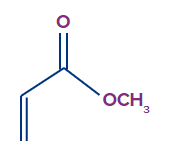
2. How does an electron rich species attached to dienes increase its electron density?
An electron- rich species attached to dienophiles increases its electron density via +M (mesomeric effect) and + I (inductive effect).
Example-
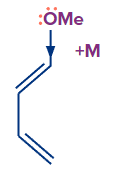
In the above example, lone pair of electrons on -OMe get delocalised via +M (mesomeric effect), thus increasing the electron density on the dienophiles.
3. How electron deficient species attached to dienophiles decrease their electron density?
Electron deficient species attached to dienophiles decrease their electron density via -R (resonance) and -I (inductive effect).
Example-
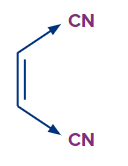
In the above example, -CN withdraw the electron density via -I effect.
4. Will the Diels-Alder reaction occur in cyclic dienes that are permanently in the s-trans conformation?
The Diels-Alder reaction will not occur in cyclic dienes that are permanently in the s-trans conformation and unable to adopt the s-cis conformation. If the diene was in the s-trans conformation, the dienophile couldn't reach both ends at the same time.


