-
Call Now
1800-102-2727
Percentage yield - Percentage purity-Stoichiometry Problems Based on Percentage Yield and Percentage Purity: Definitions, Examples, Practice Problems & FAQs
Have you ever purchased gold jewellery?
If not, then let me tell you that it comes in different grades like 24-carat, 22-carat, 18-carat etc.
24-carat gold means almost 100% pure gold, 22-carat gold implies almost 91% pure gold, 18- carat means almost 75 % pure gold and in all these cases, the rest percentage is intentionally and generally filled by other metals such as copper, zinc etc.
24-carat gold is very soft to resist wear and tear and, as a result, becomes tough to use as ornaments. So, the presence of intentionally added metals in 22-carat gold helps to make the jewellery more durable.

Table of contents
- Percentage purity
- Percentage yield
- Practice problems
- Frequently asked questions-FAQs
Percentage purity
It is defined as the percentage of a specified compound or element in an impure sample.
% purity = 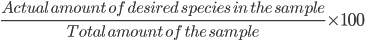
For example, 100 g of 80% pure NaCl implies that the sample contains 80 g of NaCl and 20 g of
unwanted substances, also known as impurities.
One may ask what the need for purity is. Imagine if we have a factory that makes medical drugs. The yield will still be important, but the purity of the product maybe even more important. This is because the impurities may harm the people using the drugs.
Percentage yield
Percent yield is the percent ratio of the actual (or experimental) yield to the theoretical yield, where actual yield is the yield obtained by doing an experiment.
% yield = 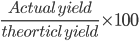
- Usually, the percent yield is lower than 100% because the actual yield is often less than the theoretical value.
- Reasons for this can include incomplete or competing reactions and loss of samples during recovery.
- It's not possible for percent yield to be over 100%, which means more samples were recovered from a reaction than predicted.
- This can happen when other reactions are occurring that also form the product. It can also be a source of error if the excess is due to incomplete removal of water or other impurities from the sample.
- Percent yield is always a positive value.
- Usually, one has to calculate the theoretical yield based on the simplest stoichiometrically balanced chemical equation.
Practice problems
Q1. Calculate the mass of CO2 produced by heating 40 g of 20% pure calcium carbonate(CaCO3)
- 4.87 g
- 3.52 g
- 2.98 g
- 4.78 g
Answer: (B)
Solution: CaCO3(s) --> CaO(s) + CO2(g)
We can say, 1 mole of CaCO3 (100 g/mol) on decomposition produces 1 mole of CO2 gas (44 g mol-1)
So, 40 g of CaCO3 will produce  g of CO2 gas.
g of CO2 gas.
= 17.6 g CO2 gas (Assuming that the compound is 100 % pure)
So, 20% pure compound amount of CO2 gas produced = 17.6 g  = 3.52 g
= 3.52 g
Q2. A 10 g sample of KClO3 on complete decomposition gave 2.27 L of oxygen at STP. What is the percentage purity of the sample of potassium chlorate?
- 67.8 %
- 87.5 %
- 82.1 %
- 90.67 %
Answer: (C)
Solution: 2KClO3(s) --> 2KCl(s) + 3O2(g)
Moles of 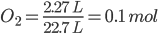
We know, 3 mol of O2 produced by 2 mol of KClO3
So, 0.1 mol of O2 will produce ( ) mol of KClO3
) mol of KClO3
= =0.067 mol of KClO3
Molar mass of KClO3 = 122.5 g mol-1
Mass of 0.67 mol of KClO3 = 0.067 × 122.5 = 8.21 g
For a 100% pure KClO3 sample, 8.21 g KClO3 will produce 2.27 L of oxygen gas at STP.
The mass of the sample is equal to 10 g
% purity = 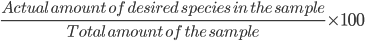
% purity =  = 82.1 %
= 82.1 %
Q3. A 20 g sample of magnesium carbonate decomposes on heating to give carbon dioxide and 8.0 g of magnesium oxide. What will be the percentage purity of magnesium carbonate in the sample? (Molar mass of Mg = 24 g mol-1)
- 84 %
- 89%
- 76 %
- 76%
Answer: (A)
Solution: MgCO3(s) --> MgO(s) + CO2(g)
Mass of MgCO3 sample = 20 g
Mass of MgO produced = 8 g
Moles of MgO produced = 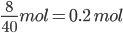
1 mole of MgO is produced by the decomposition of 1 mole of MgCO3
So, 40 g of MgO is produced by 84 g MgCO3
8 g MgO is produced by  g MgCO3
g MgCO3
For 100 % pure MgCO3, we need only 16.8 g MgCO3 to produce 8 g MgO
But the mass of the given sample of MgCO3 = 20 g
% purity = 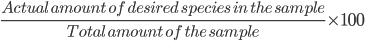
% purity =  = 84 %
= 84 %
Q4. The percent yield for the following reaction carried out in carbon tetrachloride (CCl4) solution is 70%.
Br2(g) + Cl2(g) ⟶ 2BrCl (g)
What amount of BrCl would be formed from the reaction of 0.05 mol Br2?
- 7 mol
- 0.007 mol
- 0.7 mol
- 0.07 mol
Answer: (D)
Solution: Br2(g) + Cl2(g) ⟶ 2BrCl (g)
From the equation, it can be written that 1 mole of Br2 will give rise to 2 moles of BrCl
This implies 0.05 moles of Br2 will give rise to 2 * 0.05 moles of BrCl (Assuming 100% yield).
0.05 moles of Br2 will give rise to 0.1 moles of BrCl (Assuming 100% yield).
But according to the question, the reaction yield is only 70%.
So 0.05 moles of Br2 will give rise to 0.1 * 70100 moles of BrCl
0.05 moles of Br2 will give rise to 0.07 moles of BrCl
Q5. Consider a series of reactions used in the preparation of nitric acid (HNO3) as shown below.
- 4NH3 + 5O2 --> 4NO + 6H2O [Yield = 70%]
- 2NO + O2 --> 2NO2 [Yield = 50%]
- 4NO2 + 2H2O + O2 --> 4HNO3 [Yield = 64%]
Starting with 3 moles of NH3 and excess of O2 and H2O, find the moles of HNO3 formed.
- 0.456 mol
- 0.672 mol
- 0.876 mol
- 0.54 mol
Answer: (B)
Solution: 4NH3 + 5O2 --> 4NO + 6H2O, yield = 70%
NH3 is limiting reagent. So,
Moles of NO formed = ![]() 3 mol
3 mol
With 70% yield; Moles of NO formed =  2.1 mol
2.1 mol
2NO + O2 --> 2NO2, yield = 50%
2 mol of NO produce 2 mol of NO2
2.1 mol of NO produce 2.1 mol of NO2
With 50 % yield; Mol of NO2 produced = 2.1 
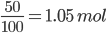
4NO2 + 2H2O + O2 --> 4HNO3, yield = 64%
4 mol of NO2 produce 4 mol of HNO3
1.05 mol of NO2 produce 1.05 mol of HNO3
With 64 % yield; Mol of HNO3 produced = 1.05 
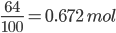
Frequently asked questions (FAQs)
Question 1. What is limiting reagent?
Answer: Limiting Reagent: The reactant that is consumed first and limits the amount of the product formed in the reaction is known as the limiting reagent, assuming a complete reaction.
- Limiting reagent is present in the least stoichiometric amount.
- Limiting reagents controls the amount of the product formed.
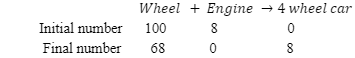
In this case, the engine is limiting reagents. The number of engines will decide the number of cars produced.
Question 2. How can decide the percentage of yield for any reaction?
Answer: Practically, not any reaction produces a product with 100 % efficiency but in the laboratory in a controlled condition we can achieve the highest yield % for any chemical production route (for lab production method, cost-effectiveness is not a priority).
Question 3. Is % yield for any particular chemical route (reaction) is same for all industries?
Answer: No, it depends on running condition and proper maintenance of temperature, pressure etc.
Question 4. Is this mandatory to balance a chemical reaction to calculate its % yield?
Answer: No, a balance equation is required to find the mole of the product formed by mole-mole analysis. We can calculate the number of moles produced or moles of the consumed reactant by another method also like the equivalence method, no need to balance reaction for solving by equivalence method.
Related Topics:
|
Empirical formula |
Mole |
|
Molarity |
Normality |
|
Atomic mass |
Density |


