-
Call Now
1800-102-2727
Pauli's Exclusion Principle - Statement, Examples, Practice Problems & FAQs
Did you ever see a box of shoes where two shoes of the same foot can fit?
I guess, not! It's a common observation that a pair of shoes (both of right foot and left foot) fits in a box, not of the same foot.

Similarly, two electrons with the same spin can not fit into an orbital. The spin has to be different and this principle is known as Pauli’s Exclusion Principle.
Table of Contents
- Statement of Pauli’s Exclusion Principle
- Importance of Pauli's Exclusion Principle
- Table Showing the Quantum Number Values for Different Orbits
- Practice Problems
- Frequently Asked Questions - FAQs
Statement of Pauli’s Exclusion Principle
Atomic electrons cannot share the same quantum state. The quantum numbers n, l, ml and ms must be unique for each electron in an atom. The principle is empirical because Pauli arrived at his result after studying atomic spectra. Electronic configuration of multi-electron species given by this rule.
According to Pauli’s Exclusion Principle, “No two electrons in an atom can have the same set of all four quantum numbers”. Only two electrons are allowed to share an orbital, and they must have the opposite spin.
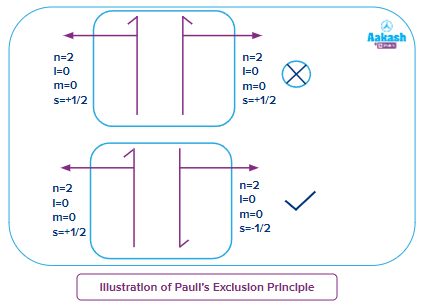
As an illustration of Pauli's Exclusion Principle, we use a neutral helium atom. In this, the atom is bound to two electrons, which have oppositely polarised outer shell positions.
The 2s orbital of the helium atom has two electrons and both the electrons have the same value of principal quantum number (n=1), azimuthal quantum number (l=0) and magnetic quantum number (ml=0), but these two will have different spin quantum numbers, if one electron is having , then the other electron will have .
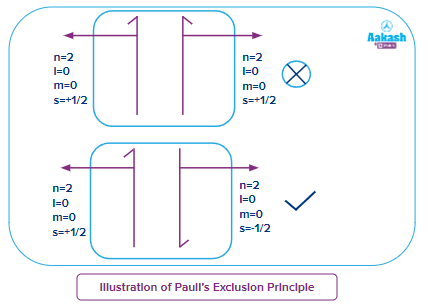
Importance of Pauli's Exclusion Principle
- This idea contributes to the explanation of a wide range of physical phenomena.
- It demonstrates how the elements work together to create chemical connections.
- With the use of this rule, periodic tables are also described.
Table Showing the Quantum Number Values for Different Orbits
|
Orbit |
n |
l |
ms |
ms |
Name of the subshell |
Name of the orbital |
Total number of electrons |
|
K |
1 |
0 |
0 |
1s |
1s |
2 |
|
|
L |
2 |
0 |
0 |
2s |
2s |
2 |
|
|
1 |
-1 |
2p |
2px |
2 |
|||
|
0 |
2py |
2 |
|||||
|
+1 |
2pz |
2 |
|||||
|
M |
3 |
0 |
0 |
3s |
3s |
2 |
|
|
1 |
-1 |
3p |
3px |
2 |
|||
|
0 |
3py |
2 |
|||||
|
+1 |
3pz |
2 |
|||||
|
2 |
-2 |
3d |
3dxy |
2 |
|||
|
-1 |
3dyz |
2 |
|||||
|
0 |
3dzx |
2 |
|||||
|
+1 |
3dx2-y2 |
2 |
|||||
|
+2 |
3dz2 |
2 |
Practice Problems
Q.1. Which of the following arrangements violates the Pauli's exclusion rule?
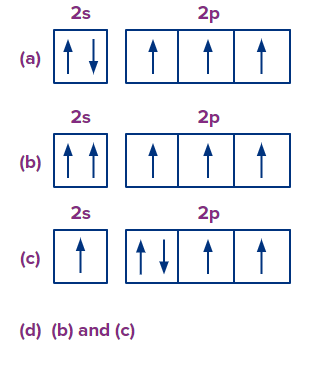
Answer: (B)
Solution: According to Pauli’s exclusion principle, no two electrons in an atom can have all four quantum numbers the same.
In option (B), in the 2s orbital, two electrons have the same spin or same spin quantum number which is a violation of Pauli’s exclusion principle.
So, option (B) is the correct answer.
Q 2. Let’s assume 1 electron has 3 spin quantum number values, then how many maximum electrons can be accommodated in the 2nd shell?
A. 8
B. 2|
C. 12
D. 18
Answer: (C)
Solution: According to our existing theory, one electron can have two spin values, so one orbital can have a maximum of two electrons. If a hypothetical experimental theory suggests that one electron can have three spin values then one orbital can accommodate a maximum of three electrons.
For n=2,
2s contains one orbital. So, no of electrons = 3 (1s3)
2s contains three orbitals. So, no of electrons = 9 (2p9)
So, in total there would be 12 electrons in the 2nd shell. Hence, the correct answer is an option (C).
Q.3. Which of the following represents the correct sets of the four quantum numbers n, l, m & srespectively for an electron in 5d orbital?
A. 5, 4, 2,
B. 5, 2, 3,
C. 5, 3, – 2,
D. 5, 2, 1,
Answer: (D)
Solution: For 5d orbital, n=5, l = 2, the possible values of m=-2,-1,0,+1,+2 and
So, the option (D) is matching these possible quantum numbers and hence option (D) is the correct answer.
Q.4. Let’s assume one electron has three spin quantum number values, then how many maximum elements can be accommodated in the 3rd period of the periodic table?
A. 8
B. 27
C. 12
D. 32
Answer: (C)
Solution: According to our existing theory, one electron can have two spin values, so one orbital can have a maximum of two electrons. If a hypothetical experimental theory suggests that one electron can have three spin values then one orbital can accommodate a maximum of three electrons.
Elements of the 3rd period in a periodic table contain electrons in 3s and 3p outer shell orbitals.
|
Subshells |
Number of orbitals |
Number of electrons (according to the new rule) |
|
3s |
1 |
3 |
|
3p |
3 |
9 |
Total number of elements which can be accommodated in the third period of the periodic table = 3+ 9 = 12
So, the correct answer is option (C).
Frequently Asked Questions-FAQs
Q1. Which equation produced the quantum numbers?
Answer: The Schrodinger wave equation yields three quantum numbers, the principal, azimuthal, and magnetic quantum numbers.
Q2. Which quantum number is not derived from the Schrodinger wave equation?
Answer: The Schrodinger wave equation cannot be used to calculate the spin quantum number because of its spin, an electron possesses a magnetic field, which is the foundation for determining electron spin. There is no net magnetic field when oppositely spinning electrons are combined because the positive and negative spins cancel one another out. Otto Stern and Walter Gerlach made the initial discovery of it in 1925.
Q3. Define the quantum numbers.
Answer: Quantum numbers are the set of four integers needed to completely characterise an electron in an atom. We can get an exact address of a particular electron. We can get an idea about a particular electron like it in which shell, which subshell, which orbital and what is the spin of that electron.
Q4. Which rules are required for writing electronic configuration of elements?
Answer: Mainly rules required for writing electronic configuration of elements are:
- Aufbau Principle
- Hund's Rule of Maximum Multiplicity
- Pauli Exclusion Principle


