-
Call Now
1800-102-2727
Pauling Scale of Electronegativity - Electronegativity, Variation of Electronegativity in Periodic Table, Application of Electronegativity, Practice Problems & FAQs
On 29th August in Indi, a national sports day is usually celebrated across the country. Many sports events are arranged in schools and colleges on this day. Mostly we all are familiar with a famous game known as “Tug-of-War” which is a sport that pits two teams against each other in a test of strength. Teams pull on the ends of a rope, with the goal being to bring the rope a certain distance in one direction against the force of the opposing team's pull. And the team that pulls the rope to their area wins the game. So, the strong team which has more strength able to pull the weak team towards itself.

Similarly, electronegativity is like an atomic “Tug-of-War” where the more electronegative atom usually attracts the less electronegative atom towards itself.
Let’s discuss more about electronegativity and how it can be measured.
Table of Content
- What is Electronegativity?
- Types of Bond
- Variation of Electronegativity in Periodic Table
- Pauling Scale of Electronegativity
- Practice Problems
- Frequently Asked Questions - FAQs
What is Electronegativity?
Electronegativity is the measure of the tendency of an atom to attract the shared electrons towards itself in a covalently bonded molecule.
A covalent bond is formed by sharing of electrons between two atoms. There are two possibilities:
Both atoms are the same: If both atoms of a covalent bond are the same as in the case of H2 whose nuclei can pull electrons to the same extent (pull of nuclei is attributed to the electronegativity of atoms, the stronger the pull of a nucleus, the greater will be the electronegativity of an atom).
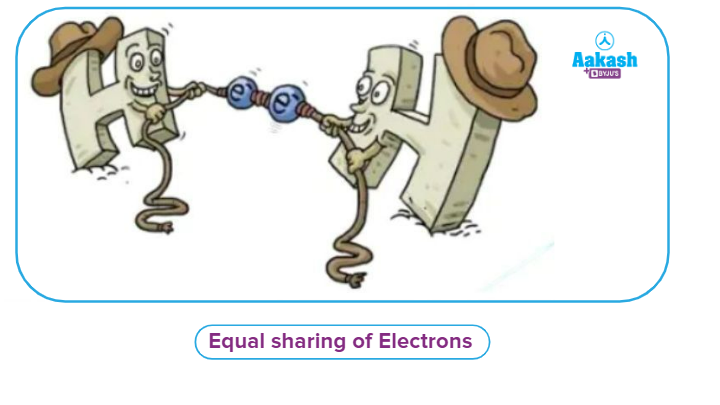
Here in the above image, we can see that at both ends hydrogen atom is present (whose nuclei can pull the electrons towards itself), so none of them is able to pull the electron cloud towards itself as there is no difference in electronegativity between these two atoms and hence equal sharing of electrons takes place between two atoms.
Both atoms are different: If both the atoms of a covalent bond are different, then due to the difference in electronegativity, unequal sharing of electrons takes place between the two atoms.

![]()
Here in the above image, we can see that a covalent bond is formed between two different atoms as chlorine and hydrogen. Chlorine will pull the electron cloud towards itself as it is more electronegative and hence unequal sharing of electrons takes place between two atoms.
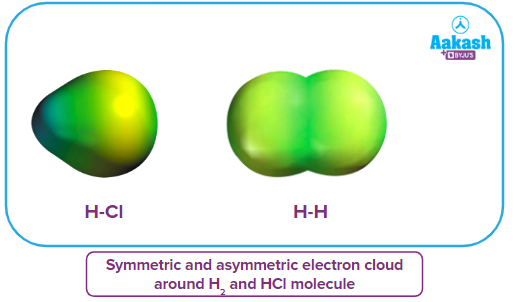
From the given image below, we can see that the electron cloud is distributed symmetrically in the case ofH2, while there is an asymmetrical distribution of the electron cloud in the case of HCl.
Types of Bonds
Non-polar covalent bond: If a covalent bond is formed between the same type of atoms then equal sharing of electrons takes place between two atoms as there will be no difference in electronegativity and there will be the symmetric distribution of electrons in the chemical bond.
So, a covalent bond in which the bonding electrons are shared equally between the two atoms is known as non-polar covalent bond.
In H2, we have the same atoms, meaning the same electronegativity value of both the atoms of hydrogen, leading to equal sharing of electrons. So, bond pair electrons are equally attracted by each nucleus.
Polar covalent bond: If a covalent bond is formed between two different types of atoms as H and Cl(Cl is more electronegative than H), then due to the difference in electronegativity, more electronegative atom will pull the electron cloud towards itself and due to this, there will be some partial negative charge on the chlorine atom and some partial positive charge on the hydrogen atom. Partial charges are created due to the asymmetric distribution of electrons in chemical bonds.
A polar covalent bond is a bond between the atoms attracting the shared electrons unequally.
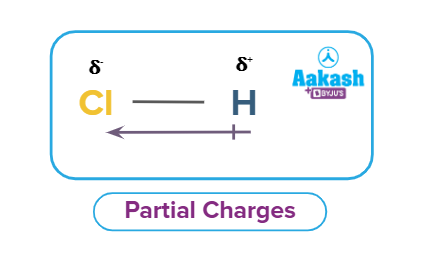
In the case of a polar covalent bond, i.e., the bond between two different atoms, the bonded pair of electrons is not shared equally by the two atoms. The atom that has a greater tendency to attract the shared pair of electrons towards itself acquires a partial negative charge, while the other atom acquires a partial positive charge. This relative tendency of an atom to attract the shared pair of electrons towards itself in a covalently bonded molecule was termed as the electronegativity of the atom by Linus Pauling.
Variation of Electronegativity in Periodic Table
Electronegativity across a period: In general, the atomic size decreases and the electronegativity increases as we move through a period from left to right.

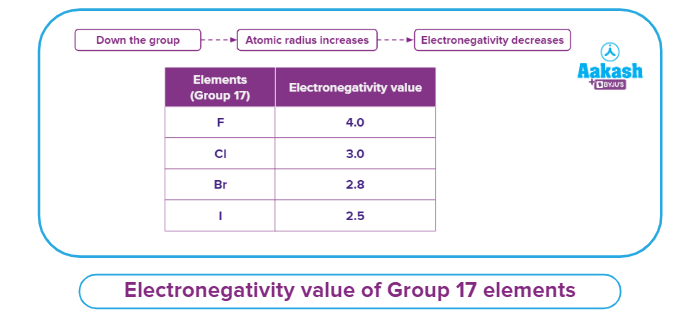
- Electronegativity down the group: In general, the atomic size increases and the electronegativity decreases as we proceed down the group in a periodic table.
- Exceptions/Irregularities in trends of electronegativity: In particular, group 13 and group 14 elements exhibit exceptions. From aluminium to thallium, electronegativity in group 13 increases down the group. As electrons transition from Al to Tl, they begin to fill the d and f-orbitals. The intermediate electrons in Ga exhibit a stronger force of attraction than those in Al due to the intervening electrons' inability to screen the nuclear charge as a result of their dispersed form, which reduces atomic radii and increases electronegativity.
Also for group 14 elements, it mostly remains the same from silicon to lead for similar reasons.
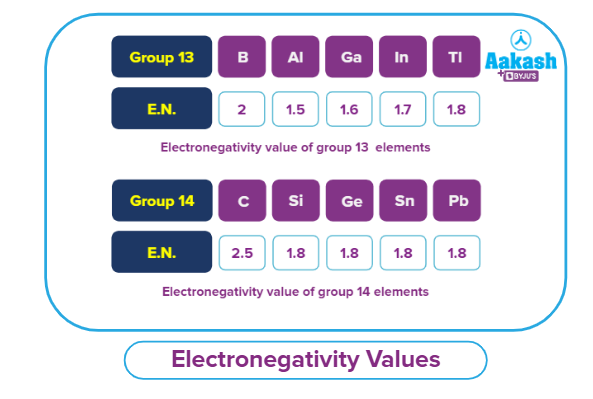
These electronegativity values will not vary much due to the poor shielding of the inner d and f-orbital
electrons.
Pauling Scale of Electronegativity
Electronegativity is a property of an atom in a molecule and it is not a fundamental property of an atom. It is not a measurable quantity unlike ionization energy and electron gain enthalpy. However, we assign the electronegativity of elements on the numerical scale. There are various scales on which electronegativity of elements are measured like the Pauling scale, Allred-Rochow scale and Mulliken scale. The Pauling scale is the most widely used among them.
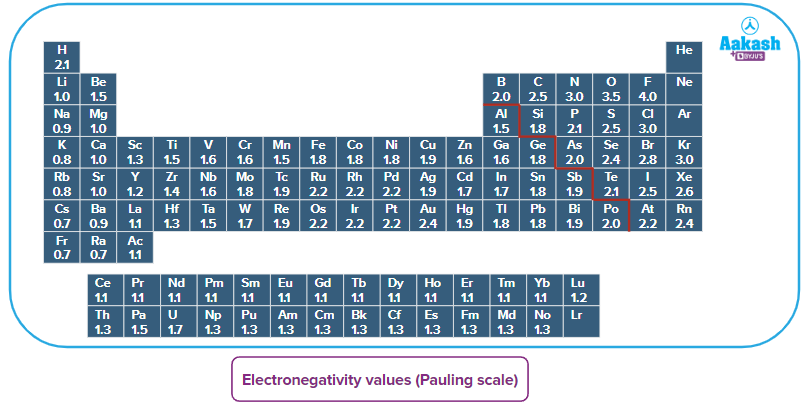
The scale for electronegativity that is most frequently used is the Pauling scale. Fluorine, the element regarded as having the highest electronegativity, was arbitrarily given a value of 4.0 by American scientist Linus Pauling in 1922.
The two atoms A and B must have stronger bonds than the average of the single bond energies of A-A and B-B molecules, he concluded. He asserts that two atoms A and B differ in their electronegativity in the following ways:
=
Where, Δ = (Actual bond energy - 100% covalent bond energy)
It also stands for the bond's polarity. According to Pauling, the geometric mean of the covalent bond energies between A-A and B-B molecules can be used to calculate 100% covalent bond energy, i.e.,
i.e.,
Substituting equation (ii) in equation (i),
Where, EA-B is the bond energy of A-B bond in kcal mol-1
EA-A is the bond energy of A-A bond in kcal mol-1
EB-B is the bond energy of B-B bond in kcal mol-1
𝜒A is the electronegativity of A and 𝜒B is the electronegativity of B
Recommended Link: https://www.youtube.com/watch?v=ZWqhTTduS7Y
Time Stamp: 1:39:18 to 1:47:35
Practice Problems
Q1. What is the correct order of electronegativity among the following options?
A) Li<Na<K<Rb<Cs
B) Li<K<Na<Rb<Cs
C) Li>Na>Rb>Cs>K
D) Li>Na>K=Rb>Cs
Answer: (D)
Solution: Electronegativity is the measure of the ability of an atom to attract shared pair of electrons towards itself when the atom is bonded covalently with the other atom in a chemical compound.
Down the group, the electronegativity values decreases, as atomic size increases.
The values of electronegativities of Li, Na, K, Rb and Cs are 1, 0.9, 0.8, 0.8 and 0.7 respectively.
So, option (D) is the correct answer.
Q2. With respect to fluorine, hydrogen will be:
A. Electropositive
B. Electronegative
C. Neutral
D. None of the above
Answer: (A)
Solution: In contrast to fluorine, which has the highest electronegativity (4.0), hydrogen has a lower electronegativity value (2.1). Their relative placements on the periodic table can also serve as justification for this. Fluorine has a bigger effective nuclear charge and is hence more electronegative since it is on the far right of its period. So, with respect to fluorine, hydrogen is electropositive. As a result, option (A) is the right response.
Q3. On Pauling's scale, which of the following elements has an electronegativity value less than 3?
A. F
B. Cl
C. O
D. C
Answer: (D)
Solution: On Pauling’s scale the electronegativity values of F , Cl , O and C are 4.0 , 3.16 , 3.44 and 2.55 respectively. So, carbon has an electronegativity value less than 3.
Q4. What will be the difference in electronegativities of F and H if bond energy of H-F bond , F-F bond and H-H bond are 134.6 kcal mol-1 , 37 kcal mol-1 and 105 kcal mol-1 respectively?
A. 1.8
B. 4.8
C. 3.8
D. 2.8
Answer: (A)
Solution:
We know that,
Where, EA-B is the bond energy of A-B bond (kcal mol-1)
EA-A is the bond energy of A-A bond (kcal mol-1)
EB-B is the bond energy of B-B bond (kcal mol-1)
𝜒A is the electronegativity of A and 𝜒B is the electronegativity of B
Now, substituting the given values;
Frequently Asked Questions - FAQs
Q1. Which is the least electronegative element of the periodic table?
Answer: Electronegativity decreases down the groups, and decreases from right to left across periods. Thus, francium is the least electronegative element.
Q2. What does the term "electropositive" mean?
Answer: Low ionisation energy and an easy tendency to lose electrons to create cations are characteristics of electropositive elements. They are essentially metallic elements, such as sodium, potassium, etc. Non-metallic elements, which are located more to the right side of the periodic table, have higher electronegativity as they attract the shared pair of electrons towards themselves.
Q3. Which elements have the lowest electronegativity values in the periodic table?
Answer: Electronegativity rises from left to right along the period and falls down the groups in a periodic table. One of the least electronegative elements is francium (0.7) according to Pauling's scale of electronegativity. Elements with low electronegativity values are Li(1.0), Na(0.9), K(0.8) , Rb(0.8) etc.
Q3. Which metal has the highest electronegativity value?
Answer: Among all metals, gold has the highest electronegativity value. According to Pauling's scale, it has an electronegativity value of 2.54.


