-
Call Now
1800-102-2727
PPM (Parts per million) - Definitions, Formula, Practice Problems and Frequently Asked Questions (FAQs)
On the evening of December 2, 1984, India witnessed one of its worst industrial incidents which led to the death of countless people in Bhopal. Thousands of people have died subsequently due to pollutants reaching their bodies through the air we breathe, the water we drink or the food we eat.
Each human being has a tolerance for such harmful chemicals but only to a certain limit. Beyond this, it can prove to be fatal. How can one ascertain those limits?
To overcome this problem, we need a concentration term which would help in throwing some light on such small but important values.
Parts per million (PPM) is the concentration term used for measuring such levels. Like each of the different concentration terms, PPM has a very specific use of providing information in minute quantities.
Table of contents
- PPM (Parts per million)
- Hardness of water
- Degree of hardness
- Example
- International standard for drinking water
- Practice problems
- Frequently Asked questions-FAQs
PPM (Parts per million)
- Generally, ppm concentration term is used when the amount of solute present in any solution is very small but significant due to its potential activity.
- Parts per million is defined as the number of parts of the solute present in every 106 parts of the solution.
E.g- 10 ppm solution means that 10 g of solute is present in every 106 g of solution.
ppm (parts per million) = ![]()
- Hardness of water generally expressed in ppm.
Hardness of water
We know, that bicarbonates  of Ca & Mgcauses temporary hardness and chlorides (Cl-) and sulphates
of Ca & Mgcauses temporary hardness and chlorides (Cl-) and sulphates  of Ca & Mgcause permanent hardness.
of Ca & Mgcause permanent hardness.
Degree of hardness
The number of parts of CaCO3 or equivalent to various calcium and magnesium salts present in a million parts of water. ( ppm in terms of CaCO3)
E.g- 160 ppm in terms of CaCl2; means 160 g CaCl2 present in 106g of solution.
Example:
Express hardness of the water in terms of CaCO3, water contains 160 ppm CaCl2
Answer: Equivalent weight of CaCl2 = ![]() =
= ![]()
Equivalent weight of CaCO3 = ![]() =
= ![]()

International standard for drinking water
|
Ions |
Maximum permissible range |
|
Flouride ion (F-) |
1 ppm |
|
Sulphate |
< 500 ppm |
|
Nitrate |
50 ppm |
|
Metals |
Maximum permissible range |
|
Fe |
0.2 ppm |
|
Mn |
0.05 ppm |
|
Al |
0.2 ppm |
|
Cu |
3.0 ppm |
|
Zn |
5.0 ppm |
|
Cd |
0.005 ppm |
Practice problems:
Q1. A water sample contains 3 mg of dissolved oxygen per 1000 g of sample. Calculate concentration of oxygen in parts per million?
- 3 ppm
- 30 ppm
- 300 ppm
- 3000ppm
Answer:(A)
Solution: 1000 g of sample water contains 3 mg or 3 10 -3g oxygen.
Concentration of oxygen (ppm) = 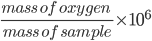
= ![]() = 3 ppm
= 3 ppm
Q2. A 400 g of body lotion sample has 0.1 g of fluoride. Find the concentration of fluoride ion in ppm
- 100 ppm
- 200 ppm
- 250 ppm
- 350 ppm
Answer:(C)
Solution: mass of solute (fluoride ion) = 0.1 g
Mass of solution = 400 g
Concentration of oxygen (ppm) = ![]()
= ![]() 250PP
250PP
Q3. A water sample contains 0.2 ppm carbon tetrachloride. How many molecules of carbon tetrachloride will be present in 0.5 g of the water sample.

- 3.1
- 3.8
- 4.6
- 2.4
Answer: (D)
Solution: 80 ppm of CaSO4 means 80 g of CaSO4 present in 106 g of solution.
80 g of Ca2+ ion present in 106 g of solution
Question 2. Is ppm concentration term also used for chemicals in pollutants?
Answer: Air quality indexes used to set a standard for air constitutes clean air. Ppm or ppb is used to measure air pollutants.
Question 3. What is the permissible range of CO2 in the atmosphere?
Answer: As explained by the Indian Meteorological Department (IMD) of India, the permissible values are
|
CO2[ppm] |
Air quality |
|
2100 |
Bad Heavily contaminated indoor, air ventilation required |
|
2000 |
|
|
1900 |
|
|
1800 |
|
|
1700 |
|
|
1600 |
|
|
1500 |
Mediocre Contaminated indoor, air ventilation recommended |
|
1400 |
|
|
1300 |
|
|
1200 |
|
|
1100 |
|
|
1000 |
Fair |
|
900 |
|
|
800 |
Good |
|
700 |
|
|
600 |
Excellent |
|
500 |
|
|
400 |
Question 4. What is PM2.5?
Answer: Particles with diameters less than 2.5 micrometres. PM2.5 is used to reflect the air quality index.
Related Topics:
|
Volume strength of H2O2 |
Mole |
|
Strength of oleum |
Normality |
|
Strength of solution |
Density |



 )
) )
)