-
Call Now
1800-102-2727
p-Block Elements– Definition, Electronic Configuration, General Features, Reactivity Towards Oxygen & Uses
Nature of Oxides and Hydrides of p-Block Elements - Introduction, Definition, Nature and Chemical Properties
We all know about the Taj Mahal, a magnificent monument in the city of Agra, made up by using white marble. But in recent times there is a lot of talk about the change in colour as the white marble turned yellowish. But do you know why this transformation of colour is taking place? Let me tell you, this is related to prevailing problems of the environment.
There are many industries nearby this beautiful architecture releases many gases like nitrogen and sulphur oxides. These gases when reacting with the moisture, they get converted into acids like nitric acid and sulphuric acid. When it rains, water droplets also bring these acids with them known as acid rain. Taj Mahal is made up of marble that is calcium carbonate and when it rains with acid along with it, these acids react with the marble and corrode it which is the reason why the marble used in Taj mahal gets this yellowish tone. Let’s delve in a bit deeper and we will try to learn more about the oxides and hydrides of p-block elements.

Table of contents
- Introduction of p-block elements
- Oxides of p-Block Elements
- Hydrides of p-Block Elements
- Practice problems
- Frequently asked questions-FAQ
Introduction of p-Block Elements
The modern periodic table is divided into 4 different blocks s-block, p-block, d-block and f-block. Elements belonging to group-13 to group-18 are classified as p-block elements because the last electron for all the elements enters into p-orbitals. For example,
Group-13
Boron has atomic number 5 belonging to the 13th group and is a p-block element as the last electron enters into the p-subshell.
Group-14
Carbon has atomic number 6 belonging to the 14th group and is a p-block element as the last electron enters into the p-subshell.
Group-15
Carbon has atomic number 7 belonging to the 15th group and is a p-block element as the last electron enters the p-subshell.
Group-16
Carbon has atomic number 8 belonging to the 16th group and is a p-block element as the last electron enters the p-subshell.
Group-17
Carbon has atomic number 9 belonging to the 17th group and is a p-block element as the last electron enters the p-subshell.
Group-18
Similarly fluorine which has an atomic number 10 belonging to the 18th group is also a p block element as the last electron is entering into the p-subshell.
Oxides of p-Block Elements
Definition of oxides
- Oxides are chemical compounds which are formed when elements react with oxygen. The compounds formed contain one or more oxygen atoms in the molecule. For example-
General reaction for the formation of oxide is:
oxide compound
Here “M” represents the element of the periodic table
- Elements from group-13 to group-17 react with oxygen and result in the formation of oxides like
- Group-18 elements generally do not react with oxygen because of its stable electronic configuration except xenon which can form oxides like xenon trioxide and xenon tetraoxide.
- More than one oxide can also be formed by the same element. For example, nitrogen which belongs to group-15 forms 5 stable oxides
Classification of oxides
Generally oxides are classified broadly into two types:
Metallic oxides:
Metallic oxides are a type of chemical compounds that are formed when metal and oxygen react. For example,
When sodium metal reacts with oxygen molecules it results in the formation of sodium oxide which is a metallic oxide.
Generally, s-block elements combine with oxygen to form metallic oxides because elements present in this block are metal.
Non-metallic oxides:
Non-metallic oxides are a type of chemical compound formed when non-metal reacts with oxygen. For example,
When carbon is allowed to burn in the presence of air it forms an oxide of carbon known as carbon dioxide.
Sulphur burns in the air to produce sulphur dioxide gas.
Generally, p-block elements combine with oxygen to form non-metallic oxides because most of the elements present in this block are non-metal.
Structures of some important oxides of p-block elements
|
Compounds |
Hybridisation |
Geometry |
Shape |
Structure |
|
CO2 |
sp |
Linear |
Linear |
|
|
CO |
sp |
Linear |
Linear |
|
|
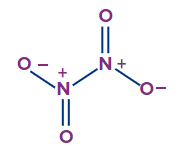
N2O3 |
Both nitrogens are sp2 hybridised |
Trigonal planar |
Planar |
|
|
N2O5 |
Both nitrogens are sp2 hybridised |
Trigonal planar |
Hexagonal |
|
|
N2O4 |
Both the nitrogens are sp2 hybridised |
Trigonal planar |
planar |
|
|
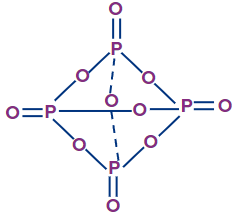
P4O10 |
All four phosphorus atoms are sp3 hybridised |
Tetrahedral |
Cage-like structure |
|
|
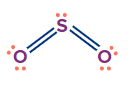
SO2 |
sp2 |
Trigonal planar |
Angular/ V-shape |
|
|
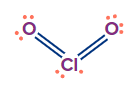
ClO2 |
sp3 |
Tetrahedral |
Angular/ V-shape |
|
|
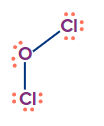
Cl2O |
sp3 |
Tetrahedral |
Angular/ V-shape |
|
|
Cl2O7 |
Both the chlorine atoms are sp3 hybridised |
Tetrahedral |
Tetrahedral |
|
|
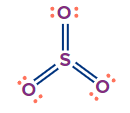
SO3 |
sp2 |
Trigonal planar |
Trigonal planar |
|
Nature of oxides of p-block elements
On the basis of the nature of oxides, oxides are classified into four different types:
- Acidic oxides: These compounds form an acidic solution when dissolved in water. The pH of the resultant oxide solution is less than 7 at, 25 ℃. For example,
Carbon dioxide reacts with water to form carbonic acid.
Sulphur reacts with water to form sulphuric acid.
- Basic oxides: These compounds form a basic solution when dissolved in water. The pH of the resultant oxide solution is more than 7 at 25 ℃. For example,
- Amphoteric oxides: These are the type of oxides which can react both with the acidic solution as well as with the basic solution to form salts. Generally, when it reacts with the acidic solution it behaves as a basic oxide and produces salt and vice versa. For example- etc.
- Neutral oxides: These are the type of oxides which are neither acidic nor basic in nature and are named as Neutral oxides. The pH of neutral oxides corresponds to 7 at25 ℃. For example- etc.
The p- block elements form all the four types of oxide- acidic oxide, basic oxide, amphoteric oxide, and neutral oxide.
Few examples of acidic oxides: etc.
Few examples of basic oxides:
Examples of neutral oxides:
Examples of amphoteric oxides: etc.
- On moving across the period the acidic character of the p-block oxides increases due to increases in the electronegativity of the element. For example,
In the 2nd period, the order of acidic strength of oxides is .
In the 3rd period, the order of acidic strength of oxides is
Similarly, on moving down the group the acidic character of the oxides of p-block elements generally decreases because when we move down the group, the metallic character of the element increases and electronegativity value decreases. Therefore, the acidic nature of the oxide decreases down the group.
Nature of oxides for group-13 elements
|
Formula of oxide |
Nature |
|
B2O3 |
Acidic |
|
Al2O3 |
Amphoteric |
|
Ga2O3 |
Amphoteric |
|
In2O3 |
Basic |
|
Tl2O |
Basic |
Nature of oxides for group-14 elements
|
Formula of oxide |
Nature |
|
CO2 |
Acidic |
|
SiO2 |
Acidic |
|
GeO2 |
Amphoteric |
|
SnO, SnO2 |
Amphoteric |
|
PbO, PbO2 |
Amphoteric |
Nature of oxides for group-15 elements
|
Formula of oxide |
Nature |
|
N2O5 |
Acidic |
|
P4O10 |
Acidic |
|
As4O10 |
Amphoteric |
|
Sb4O10 |
Amphoteric |
|
Bi2O3 |
Basic |
- For the element forming more than one oxide, the acidic strength of the oxide is determined by the oxidation state value. The higher the oxidation state of the central atom more will be the acidic nature of the oxide.
For example, SO3 is more acidic than SO2 a molecule because in the case of SO3 oxidation state of sulphur is +6 but in the case of SO2 the molecule, the oxidation state is +4.
General chemical properties of oxides of p-block elements
|
Reacting elements |
Non-metallic oxides |
Metallic oxides |
Amphoteric oxides |
|
Reaction with water |
It reacts with water to form acid. |
It reacts with water to form the base. |
It does not react with water. |
|
Reaction with basic or acidic oxide |
It generally reacts with a basic oxide to form salt and does not react with an acidic oxide. |
It generally reacts with an acidic oxide to form salt |
Amphoteric oxide reacts with acidic and basic oxide to form salt and water. |
|
Reaction with acid or base |
Acidic oxide reacts with a to form salt and water . |
Basic oxide reacts with an acid to form salt and water. |
It is the type of oxide that can react with both acid and base to form the salt.
|
|
Examples of oxides |
etc |
etc |
Hydrides of p-Block Elements
Definition of hydrides
Hydrides is defined as the compounds formed when elements react with hydrogen to form the compounds. For example - etc
In general it can be represented as
hydride compound
Note: Some hydrides may not be produced directly from the reaction of hydrogen with the element but can be produced by some other indirect methods.
For example,
Diborane is prepared from the reaction of sodium borohydride and iodine in diglyme.
Classification of hydrides
On the basis of the type of the compound formed generally hydride of an element is broadly classified into three types:
- Ionic hydrides: When a hydrogen molecule combines with highly electropositive s-block elements like (Na, Mg, Ca etc), they produce ionic hydrides that are non-conducting, crystalline, and non-volatile in their solid-state. For example,
- Metallic hydrides: These types of hydrides are formed from the elements of d-block or f-block and exhibit properties like high melting and boiling point. They sometimes also form non-stoichiometric hydrides. For example: .
- Covalent hydrides: Covalent hydrides are the type of compounds that are formed when hydrogen reacts with the non-metal present in the p-block. For example- etc.
Nitrogen molecules react with hydrogen at a high temperature to form an ammonia molecule.
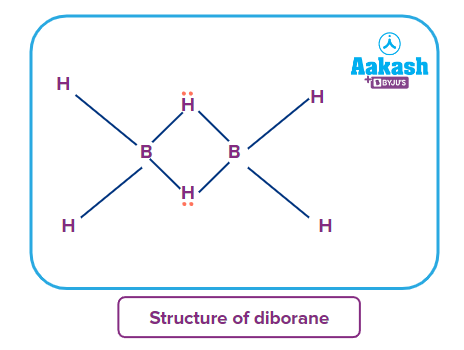
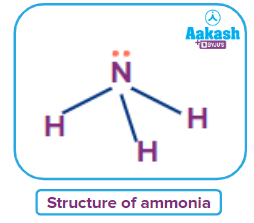
Structures of some important hydrides of p-block elements
|
Compounds |
Hybridisation |
Geometry |
Shape |
Structure |
|
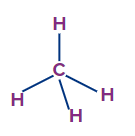
CH4 |
sp3 |
Tetrahedral |
Tetrahedral |
|
|
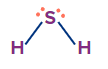
H2S |
sp3 |
Tetrahedral |
Angular/ V-shape |
|
|
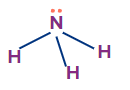
NH3 |
sp3 |
Trigonal planar |
Pyramidal |
|
|
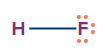
HF |
sp3 |
Tetrahedral |
Linear |
|
|
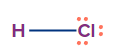
HCl |
sp3 |
Tetrahedral |
Linear |
|
|
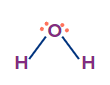
H2O |
sp3 |
Tetrahedral |
Angular/ V-shape |
|
|
HBr |
sp3 |
Tetrahedral |
Linear |
|
|
HI |
sp3 |
Tetrahedral |
Linear |
|
Some important chemical reactions of hydrides of p-block elements
- Diborane react with water to form boric acid and releases hydrogen gas.
- Diborane react with chlorine to form boron trichloride and hydrogen chloride is formed.
- Excess ammonia reacts with diborane to form boron nitride
- Group 14 hydride react with oxygen to form oxide compounds. For example,
- Group-15 hydride reacts with water to form weak base
- Ammonia reacts with oxygen to release nitrogen gas.
- Hydrogen sulphide reacts with excess oxygen with blue flame and releases sulphur
dioxide gas but in the presence of a limited supply of oxygen, sulphur is formed.
Practice problems
Q1. Select the correct option which represents the neutral oxide from the given list of oxides.
(
Answer: (B)
Solution: Neutral oxides are the type oxides which are neither acidic nor basic in nature and are named as neutral oxides. The pH of neutral oxides corresponds to 7 at 25 ℃. Out of the given oxides, H2O and N2Oare neutral oxides, Al2O3 is the example of amphoteric oxide, are acidic oxides.
Q2. Which of the following oxides is an amphoteric oxide?
- Al2O3
- CaO
- NO
- SO2
Answer: (A)
Solution: Amphoteric oxides are the type of oxides which can react with both acids and bases to form the salt. An oxide behaves as an acidic oxide when it reacts with a base to form a salt and when it reacts with acid then it acts as a basic oxide. An amphoteric oxide can react with both acid and base, whereas a neutral oxide won’t react with anyone. So, CaO is basic in nature, SO2 is acidic in nature, NO is a neutral oxide and Al2O3 is an amphoteric oxide as Al2O3 can react with both acid and base.
Q3. Select the correct option to identify the compound formed when P2O5 is allowed to react with water.
- It will result in the formation of acidic solution
- It will result in the formation of basic solution
- It will form a salt
- No reaction will occur
Answer: (A)
Solution: When the acidic oxide is allowed to react with water it results in the formation of an acidic solution and P2O5 is an acidic oxide.
Q4. Considering the oxides of the p-block elements, which of the following options is correct?
- Acidic character of oxides generally increases as we move from left to right in a period.
- On moving down the group the basic nature of oxides generally increases.
- SO3 is a stronger acid than SO2
- All of these are correct
Answer: (D)
Solution: On moving across the period (left top right), the acidic character of the oxides increases because the electronegativity of elements increases from left to right in a period. For example,
The order of acidic character of oxide of 2nd period elements will be:
Similarly, on moving down the group the acidic character of the oxides decreases because when we move down the group, metallic character increases and electronegativity value decreases. Therefore, the acidic nature of oxides decreases down the group. For example;
|
Formula of oxide |
Nature of oxide |
|
B2O3 |
Weakely Acidic |
|
Al2O3 |
Amphoteric |
|
Ga2O3 |
Amphoteric |
|
In2O3 |
Weakly Basic |
|
Tl2O |
Strongly Basic |
For the elements forming more than one oxide, the strength of the acidic oxide is determined by the oxidation state value. The higher the oxidation state of the central atom more will be the acidic nature of the oxide because an increase in the oxidation state of the central atom electronegativity increases which increases the electron-withdrawing tendency from surrounding oxygen atoms and result in an increase in the acidic nature of the oxide.
For example, SO3 is more acidic than SO2 because in the case of SO3 the oxidation state of sulphur is +6 but in the case of SO2, the oxidation state is +4 respectively.
Q5. Which among the given options represents the covalent hydride?
- NaH
- NH3
- HF
- Both B and C
Answer: (D)
Solution: Covalent hydride is the type of hydride which is formed by nonmetals when it reacts with hydrogen and is bonded together by covalent bonds like in the case of NH3 and HF molecule but hydrides formed by the metals which are held together by the electrostatic force of attraction are known as ionic hydrides like NaH.
Frequently asked questions
Q1. What is the difference between peroxide and superoxide?
Answer: Peroxide is the type of oxide in which an oxygen atom is bonded with another oxygen atom with a single bond such that the oxidation state of both the oxygen atoms is -1. For example -H2O2. Whereas the superoxide is a type of oxide which is formed when two oxygen atoms are covalently bonded together but the oxidation state of one oxygen atom is -1 and the other oxygen atom is 0 due to which the average oxidation state of oxygen become), for example-KO2.
Q2. Why fluorine compound with oxygen is not considered as an oxide?
Answer: Oxide is the compound which forms when oxygen is linked with less electronegative element and therefore carries (-2) an oxidation state. But in the case of fluorine which is the most electronegative element of the periodic table, oxygen becomes a less electronegative element and we generally do not call it an oxide and resulting in the formation of the compound with a chemical formula OF2 which is known as oxygen fluoride.
Q3. What are non-stoichiometric hydrides?
Answer: In the case of some of the compounds of d and f-block elements which are formed in where the atomic ratio of elements is not a simple whole-number ratio (i.e., does not follow a law of constant proportion) and are known as a non-stoichiometric compound.
For example- etc.
Q4. What are electron-rich and electron-deficient hydrides?
Answer: Electron-rich hydrides are covalent hydrides which are formed when a central atom contains eight electrons but some electrons are present in the form of lone pairs. For example- HF, NH3. Whereas electron-deficient hydrides like Diborane (B2H6) are the covalent hydrides which are formed where the central atom contains less than eight electrons in the valence shell or outermost shell.