-
Call Now
1800-102-2727
Oxoacids of Sulphur – Oxoacids of Sulphur, Preparation, Physical Properties, Chemical Properties, and Uses of Sulphuric Acid
Which substance is referred to as the "King of Chemicals"?
That's right; that is sulphuric acid. Sulphuric acid is referred to as the "King of Chemicals" since sulphuric acid consumption in any given nation determines its level of prosperity. Due to its widespread application in almost every industry, sulphuric acid is a chemical of significant commercial significance.
Sulphuric acid is presently made by the contact process, however, it was once made through the distillation of ferrous sulphate (FeSO4.7H2O), also known as green vitriol. Therefore, ‘oil of vitriol’ is another name for sulphuric acid.
We shall learn more about the structure and properties of sulphuric and other oxoacids of sulphur in this article.
TABLE OF CONTENTS
- Oxoacids of Sulphur
- Sulphurous Acid Series
- Sulphuric Acid Series
- Thionic Acid Series
- Peroxy Acid Series
- Sulphuric Acid – Manufacturing by Contact Process
- Sulphuric Acid – Physical Properties
- Sulphuric Acid – Chemical Properties
- Sulphuric Acid – Uses
- Practice Problems
- Frequently Asked Questions – FAQ
Oxoacids of Sulphur
In the case of sulphur, a sizable number of oxoacids are known, either in the free state or in the form of salts. Thioacids are oxoacids with S-S linkages, sulphuric acids are oxoacids with sulphur in the higher oxidation states, sulphurous acids are oxoacids with sulphur in the lower oxidation states, and peroxy acids are oxoacids with O-O (peroxide) linkages.
Sulphurous Acid Series
- Sulphurous Acid (H2SO3)
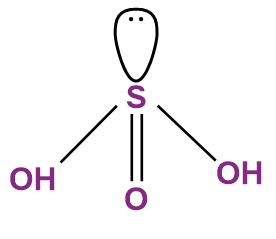
- The oxidation state of sulphur in sulphurous acid is +4.
- It acts as an oxidising and a reducing agent.
- Thiosulphurous Acid (H2S2O2)
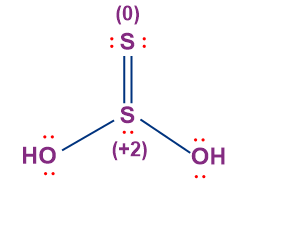
- The oxidation state of sulphur in thiosulphurous acid is +1.
- Hyposulphurous Acid (H2S2O4)
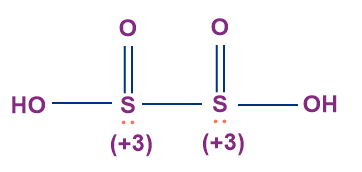
- It is also known as hydrosulphurous acid or dithionous acid.
- The oxidation state of sulphur in hyposulphurous acid is +3.
- Pyrosulphurous Acid (H2S2O5)
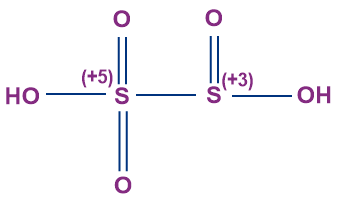
- It is not known to exist in the free state.
- It is also known as disulphurous acid.
- The oxidation state of sulphur in pyrosulphurous acid is +4.
Sulphuric Acid Series
- Sulphuric Acid (H2SO4)
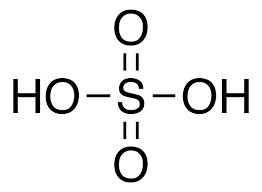
- The oxidation state of sulphur in sulphuric acid is +6.
- It acts as dehydrating, oxidising and pickling agent.
- Thiosulphuric Acid (H2S2O3)
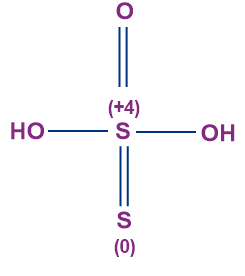
- The oxidation state of sulphur in thiosulphuric acid is +2.
- Pyrosulphuric Acid (H2S2O7)
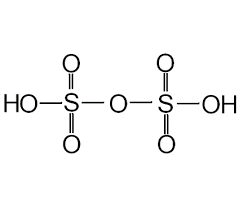
- The oxidation state of sulphur in pyrosulphuric acid is +6.
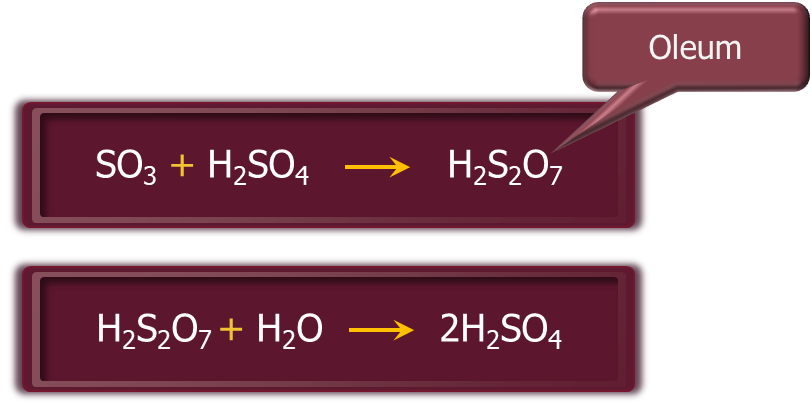
- It is also known as fuming sulphuric acid or anhydride of sulphuric acid or oleum.
Thionic Acid Series
- Dithionic Acid (H2S2O6)
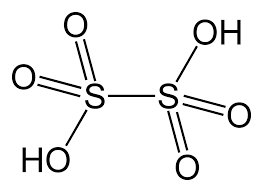
- The oxidation state of sulphur in dithionic acid is +5.
- Polythionic Acid (H2SxO6)
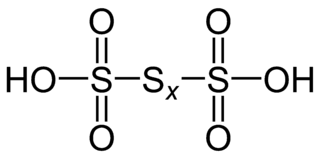
Where x=1-6
- The oxidation state of sulphur in polythionic acid is +5.
Peroxy Acid Series
- Peroxymonosulphuric Acid (H2SO5)
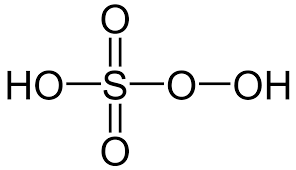
- It is also known as Caro’s acid.
- The oxidation state of sulphur in peroxymonosulphuric acid is +6.
- Peroxydisulphuric Acid (H2S2O8)
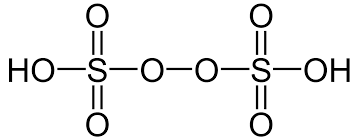
- It is also known as Marshall’s acid.
- The oxidation state of sulphur in peroxydisulphuric acid is +6.
The most important oxoacid of sulphur is sulphuric acid. Let us get to know the preparation, physical properties, chemical properties and uses of sulphuric acid.
Sulphuric Acid – Manufacturing by Contact Process
Sulphuric acid is manufactured by the contact process. Sulphuric acid obtained by the contact process is 96–98% pure.
Step – 1: Sulphur or sulphide ores are burnt in air to generate SO2.
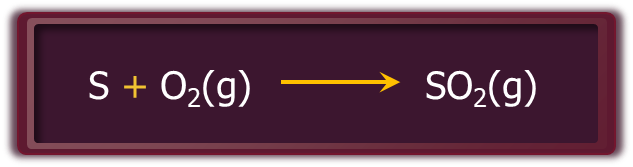
By eliminating dust and other contaminants, such as compounds containing arsenic, the SO2 thus created is refined.
Step – 2: SO2 produced in step – 1 is converted into SO3 by the reaction with oxygen in the presence of a catalyst (V2O5). This is the key step. This reaction is exothermic and reversible. Low temperature (not too low) and high pressure are favourable conditions for maximum yield.

![]()
Step – 3: The SO3 produced in the second step is absorbed in 98% H2SO4 to give oleum (H2S2O7). Dilution of oleum with water gives H2SO4 of the desired concentration. In the industry, two steps are carried out simultaneously to make the process a continuous one and to reduce the cost.




Sulphuric Acid – Physical Properties
- It is a thick, greasy liquid that is colourless.
- It dissolves in water while producing a significant quantity of heat.
- At 298 K, it has a specific gravity of 1.84.
- Its boiling point is 590 K
- Its freezing point is 283 K
- It has a highly corrosive nature. Therefore, it produces burns on the skin due to dehydration of the skin.
Sulphuric Acid – Chemical Properties
- Sulphuric acid ionises in two steps as given below.
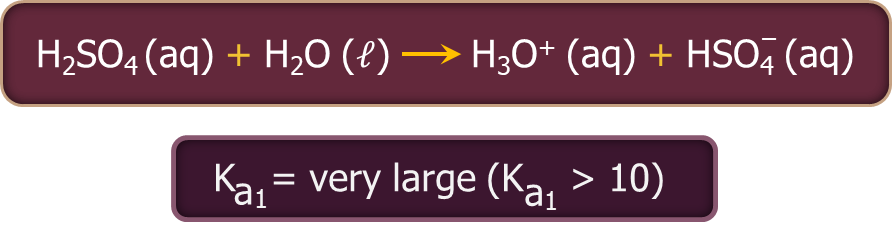
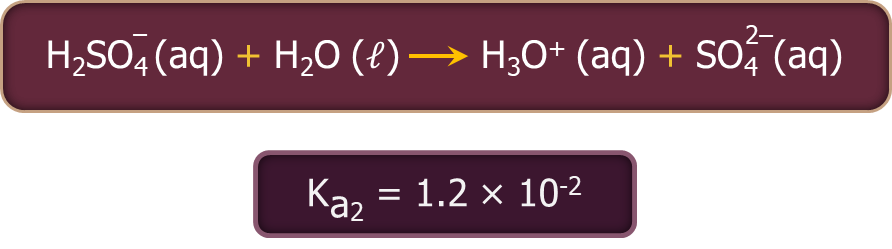
The larger value of Ka1 (Ka1>10) implies that H2SO4 is largely dissociated into H+ and HSO4- ions. The greater the dissociation constant, the stronger is the acid.
- Concentrated sulphuric acid is a strong dehydrating agent. It removes water from organic compounds.
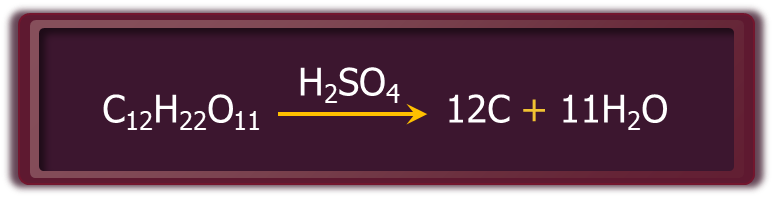
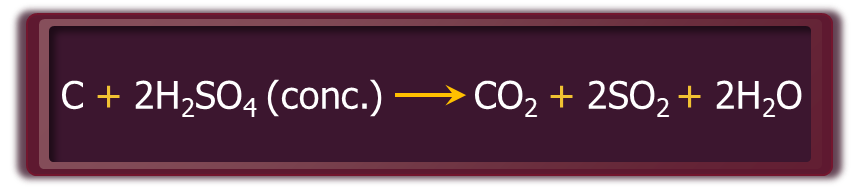
- Hot and concentrated sulphuric acid is a moderately strong oxidising agent. Its strength as an oxidising agent is intermediate between phosphoric and nitric acids. Both metals and non-metals are oxidised by sulphuric acid. In this process, it gets reduced to SO2.


Sulphuric Acid – Uses
- It is used in the manufacture of various chemicals such as hydrochloric acid, phosphoric acid, nitric acid, etc,.
- It is directly or indirectly used in the manufacture of dyes, drugs, paints, pigments and detergents.
- It is used in the manufacture of fertilisers such as ammonium sulphate and super phosphate of lime.
- A mixture of sulphuric acid and nitric acid, called the nitrating mixture, is used in the nitration of organic compounds. This nitration process is used in the manufacture of a large number of explosives including TNT (2,4,6-trinitrotoluene), picric acid (2,4,6-trinitrophenol), etc,.
- To remove unwanted sulphur and other tarry compounds, crude petrol is treated with sulphuric acid.
- Metals like copper and silver are extracted from their ores using sulphuric acid.
- It is used in the leather industry for tanning.
- It is used as an electrolyte in lead storage batteries.
- It is used as a drying and dehydrating agent in laboratories.
- It is used for cleaning the surface of metals (pickling) before electroplating, galvanising and enamelling.
Practice Problems
1. Which of the following oxoacids of sulphur has a lone pair of electrons on sulphur?
a. Pyrosulphuric acid
b. Peroxodisulphuric acid
c. Sulphuric acid
d. Sulphurous acid
Answer: D
Solution: Sulphur has six valence electrons.
- Pyrosulphuric Acid (H2S2O7)
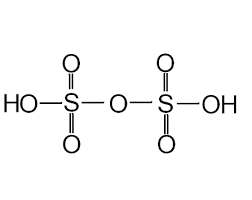
- Peroxydisulphuric Acid (H2S2O8)
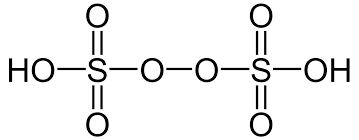
- Sulphuric Acid (H2SO4)
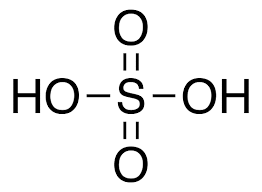
- Sulphurous Acid (H2SO3)
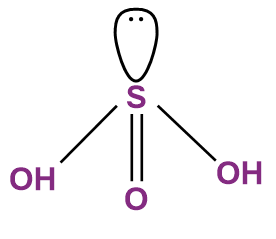
The oxoacid of sulphur that has a lone pair of electrons on the central sulphur atom is sulphurous acid (H2SO3). In sulphurous acid, 4 of the valence electrons of sulphur participate in bonds, leaving the other 2 electrons on sulphur as a lone pair. In the other oxoacids of sulphur, all the six valence electrons of sulphur participate in bonding, and therefore there are no lone pairs of electrons on the sulphur atoms.
So, option D is the correct answer.
2. More than one oxidation state of sulphur is seen in which of the following oxoacids of sulphur?
a. H2S2O8
b. H2S2O6
c. H2S2O4
d. H2S2O2
Answer: D
- Peroxydisulphuric Acid (H2S2O8)
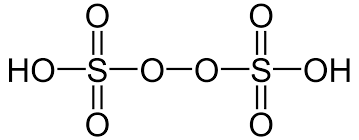
- The oxidation state of both the sulphur atoms in peroxydisulphuric acid is +6.
- Dithionic Acid (H2S2O6)
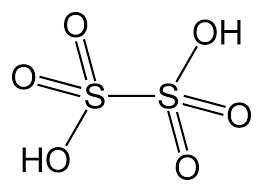
- The oxidation state of both the sulphur atoms in thionic acid is +5.
- Hyposulphurous Acid (H2S2O4)
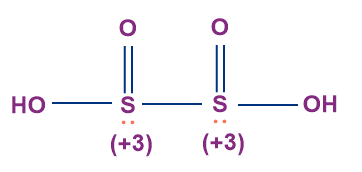
- The oxidation state of both the sulphur atoms in hyposulphurous acid is +3.
- Thiosulphurous Acid (H2S2O2)
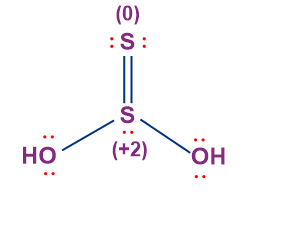
- The oxidation state of the central sulphur atom in thiosulphurous acid is +2, whilst the oxidation state of the terminal sulphur atom doubly-bonded to the central sulphur atom is 0. Thus, both the two sulphur atoms in thiosulphurous acid have different oxidation states.
So, option D is the correct answer.
3. S-S is present in which of the following oxoacids of sulphur?
a. Thiosulphuric acid
b. Dithionic acid
c. Marshall’s acid
d. Oleum
Answer: B
- Thiosulphuric Acid (H2S2O3)
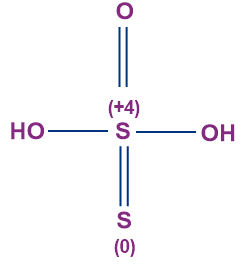
- The oxidation state of sulphur in thiosulphuric acid is +2.
- No S-S bond is present in thiosulphuric acid.
- Dithionic Acid (H2S2O6)
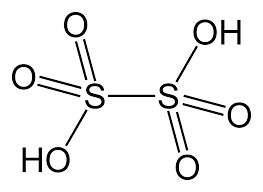
- The oxidation state of sulphur in dithionic acid is +5.
- S-S bond is present in dithionic acid.
- Peroxydisulphuric Acid (H2S2O8)
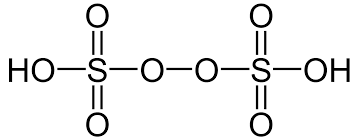
- It is also known as Marshall’s acid.
- The oxidation state of sulphur in peroxydisulphuric acid is +6.
- No S-S bond is present in Marshall’s acid.
- Pyrosulphuric Acid (H2S2O7)
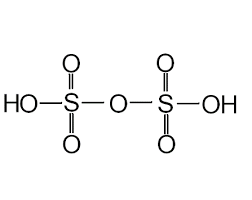
- The oxidation state of sulphur in pyrosulphuric acid is +6.
- It is also known as fuming sulphuric acid or anhydride of sulphuric acid or oleum.
- No S-S bond is present in Marshall’s acid.
So, option B is the correct answer.
4. The oxidation state of sulphur is the least in which of the following oxoacids of sulphur?
a. Thiosulphurous acid
b. Hyposulphurous acid
c. Sulphurous acid
d. Sulphuric acid
Answer: A
- Thiosulphurous Acid (H2S2O2)
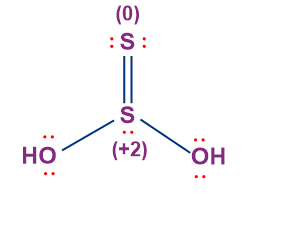
- The oxidation state of sulphur in thiosulphurous acid is +1.
- Hyposulphurous Acid (H2S2O4)
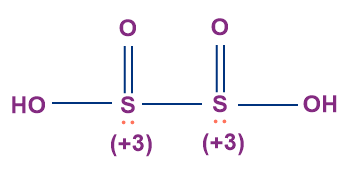
- The oxidation state of both the sulphur atoms in hyposulphurous acid is +3.
- Sulphurous Acid (H2SO3)
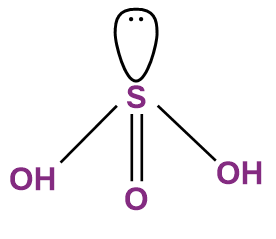
- The oxidation state of sulphur in sulphurous acid is +4.
- Sulphuric Acid (H2SO4)
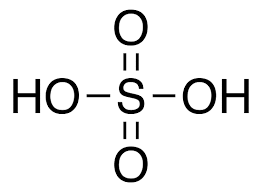
- The oxidation state of sulphur in sulphuric acid is +6.
So, option A is the correct answer.
Frequently Asked Questions – FAQ
1. What happens when water reacts with sulphuric acid?
Answer: Water and sulphuric acid react extremely vigorously and exothermically. If concentrated sulfuric acid is mixed with water, it may boil and sputter, giving you a painful acid burn.
2. What are the health hazards of sulphuric acid?
Answer: Sulphuric acid is a corrosive chemical that harms the skin, eyes, teeth, and lungs. Extreme exposure can be fatal.
3. Is sulphuric acid inflammable?
Answer: Sulphuric acid is a strong oxidising agent that improves the burning of other compounds even if it is not combustible itself. Sulphuric acid does not ignite on its own.
4. Which metal is resistant to the action of sulphuric acid?
Answer: The most corrosion-resistant metal currently in use is tantalum. The explanation for tantalum's high corrosion resistance in aggressive conditions is the existence of a naturally occurring oxide coating on its surface. It has unparalleled resistance to corrosion in both sulphuric and hydrochloric acids.


