-
Call Now
1800-102-2727
Oxidation of Alkynes-Ozonolysis, Baeyer’s Reagent, Baeyer’s test, Hydroboration Oxidation, Catalytic Oxidation, Practice Problems ad FAQs
Alkynes have at least one triple bond and so are highly unsaturated. The two pi-bonds are very weak and undergo chemical reactions. The triple bond is shorter than double and single bonds and needs more energy to break.

They can undergo partial or complete oxidation to give a variety of products like vicinal dialdehyde, vicinal diketone, carboxylic acid, or carbon dioxide. Can you tell me what happens when an alkyne is treated with oxidizing agents such as ozone or potassium permanganate?
When alkyne is oxidized, it is difficult to answer unless you know the alkyne, conditions and reagents. So, let us discuss and comprehend how we can select the product of the alkyne oxidation reaction.
Table of Contents
- Ozonolysis of alkynes
- Mechanism of ozonolysis reaction of alkyne
- Oxidation by Baeyer’s Reagent (Potassium Permanganate)
- Baeyer’s Test
- Hydroboration-Oxidation Reaction of Alkynes
- Catalytic Oxidation of Alkynes into 1,2-Diketone Derivatives
- Practice Problems
- Frequently Asked Questions
Oxidation of Alkynes
Alkynes can be oxidized primarily through ozonolysis, potassium permanganate in natural and alkaline forms, water oxidation, and hydroboration oxidation. Let us go over them one by one.
Ozonolysis of alkynes
Alkynes contain 2 pi bonds. The triple bond of alkynes, in the presence of an ozone molecule, undergoes oxidative cleavage. Alkynes undergo oxidation resulting in the formation of end products such as dialdehyde, diketones and acid anhydrides. In the presence of water, the acid anhydride gives rise to two carboxylic acids with the help of hydrolysis.
Let us understand the reaction mechanism of ozonolysis taking place in an alkyne.
Mechanism of ozonolysis reaction of alkyne
The reaction mechanism of ozonolysis involves three steps.
Step 1: Attack of the ozone molecule
The ozone molecule consists of 3 oxygen atoms. One of the oxygen atoms acquires a partial negative charge and the other one acquires a partial positive charge. The negatively charged oxygen ion attacks the triple-bonded carbon atom. The ozone molecule gets attached to the reactant. Rearrangement occurs within the structure, resulting in the formation of an intermediate structure called the molozonide.
Step 2: Formation of ozonide intermediate
The molozonide intermediate further rearranges itself, resulting in the formation of a stable ozonide intermediate. In ozonide, we can see that the triple bond is reduced to a single bond. Later on, the single bond between two carbon atoms also breaks, yielding different carbonyl compounds.
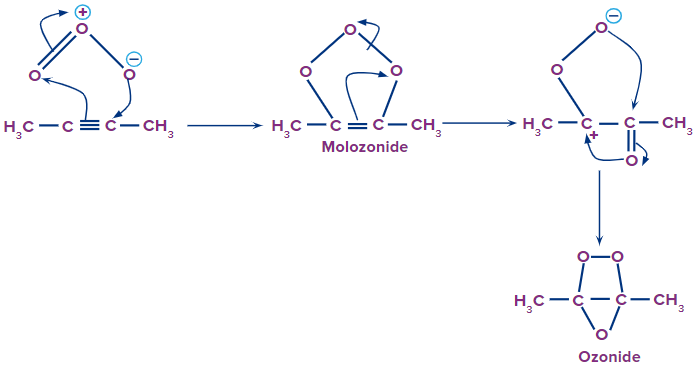
Formation of Molozonide and Ozonide
Step 3: Formation of carbonyl compounds
It can be performed by two types of mechanisms, which are given as follows:
- Reductive ozonolysis
- Oxidative ozonolysis
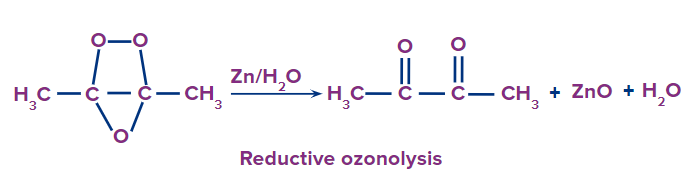
Reductive ozonolysis
This type of ozonolysis of alkyne involves the addition of an ozone molecule to an alkyne to form an ozonide. Ozonide is unstable and decomposes easily by reduction into , -diketones. The reagent that is used in reductive ozonolysis is given as follows:
(i) Zn and H2O
(ii) (CH3)2S and H2O
(iii) CH3COOH
The formation of , -diketones can be given as:
Oxidative Ozonolysis
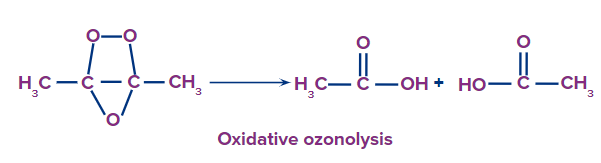
This type of ozonolysis of alkynes involves the addition of an ozone molecule to an alkyne to
form an ozonide, followed by the oxidation of the ozonide to smaller oxidized molecules like acid. The reagent which is used in oxidative ozonolysis is given as follows:
(i) H2O
(ii) H2O2
The formation of carboxylic acids can be given as:
In oxidative ozonolysis, if formic acid will form, it will further decompose into carbon dioxide and water
HCOOHCO2+H2O
Oxidative Ozonolysis of Acetylene:
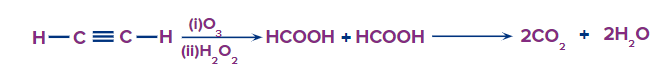
Oxidation by Baeyer’s Reagent (Potassium Permanganate)
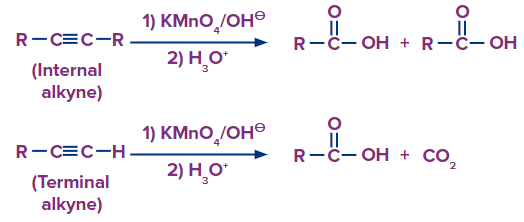
Alkynes, like alkenes, can be gently or strongly oxidized depending on the reaction environment. Because alkynes are less stable than alkenes, the reaction conditions can be more forgiving. In neutral permanganate solution, for example, alkynes form vicinal dicarbonyls.
The generic reactions for the various gentle oxidative conditions are summarized below.

Mechanism:
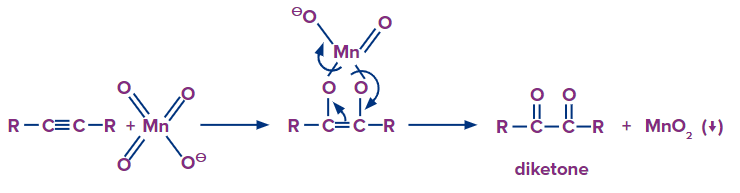
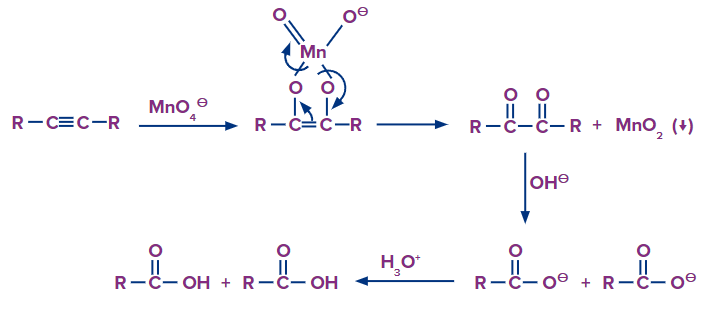
The alkyne is cleaved into two products during strong oxidation with basic potassium permanganate. Because at least one of the reaction products is a carboxylic acid, the acid-base chemistry of the product in the reaction solution must be considered. Carboxylic acids are deprotonated to carboxylates in basic solutions. To protonate the carboxylate to the neutral form of the carboxylic acid, a second reaction step is required.
The generic reactions for the various oxidative conditions - strong- are summarized below.
Mechanism:
Baeyer’s Test
In qualitative organic analysis KMnO4 is used to test for the presence of unsaturation. It is also known as Baeyer's reagent after the German organic chemist Adolf von Baeyer.
The reagent is an alkaline potassium permanganate solution.
When alkaline potassium permanganate is added to an unsaturated hydrocarbon, the pink colour of potassium permanganate disappears. The colour fades from purplish-pink to brown when it reacts with double or triple bonds (-C=C- or -C≡C-).
Bayer’s reagent will give brown precipitates with both terminal and Non- terminal alkynes. Hence, this test can be used for the detection of alkynes but does not specify whether they are terminal alkynes or non-terminal alkynes.
R-CC-R + KMnO4+KOH RCOO-K+ + MnO2
Baeyer’s Reagent Brown precipitates

Baeyer’s Test
Hydroboration-Oxidation Reaction of Alkynes
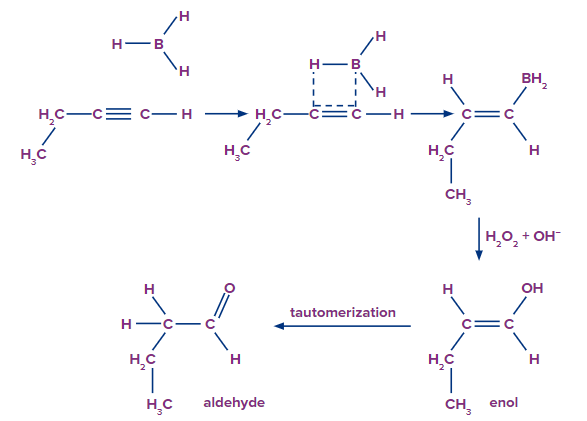
In an anti-Markovnikov way, the terminal alkynes can undergo hydroboration. The less substituted carbon, which is also the least hindered, becomes a priority target for the boron atom's attack. A bulky reagent of borane must be used to stop the reaction at the alkenyl group attached borane stage. If borane is used alone, it will result in the hydroboration of both the alkyne's pi bonds.
The oxidation step of this process begins with the oxidation of alkyl borane into vinyl alcohol, which contains both an alkene and an OH group. This alcohol is now tautomerized to produce a stable aldehyde form. The hydroboration reaction of a terminal alkyne is illustrated below
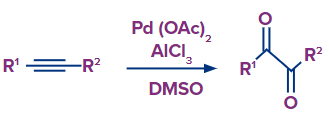
Catalytic Oxidation of Alkynes into 1,2-Diketone Derivatives
Using a Pd(OAc)2/Lewis acid AlCl3 catalytic system with DMSO as the oxidant and solvent, a new synthetic technique for effective oxidation of alkynes into 1,2-diketones has been devised. This method tolerated a wide range of functional groups and produced 1,2-diketone derivatives in 62–99 percent yields under our optimal reaction conditions.
Practice Problems
1. Which test can be used to distinguish between Cycloheptyne and Cycloheptane?
a. Tollen’s Test
b. Ammoniacal cuprous chloride
c. Baeyer’s Test
d. All of these
Solutions: Cyclohexane being non-terminal alkyne and cyclohexane being alkane can be distinguished with bromine water tests as in option (A) Tolen’s test and option (B) Ammoniacal cuprous chloride give precipitates with terminal alkynes only.
Hence, when an alkaline potassium permanganate (Baeyer’s reagent) is added to an unsaturated hydrocarbon, the pink colour of potassium permanganate disappears. The colour fades from purplish-pink to brown when it reacts with double or triple bonds
Hence, the correct option is (C).
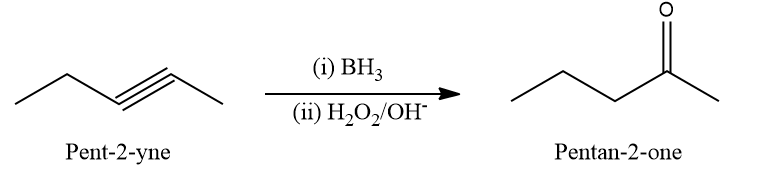
2. When Pent-2-yne undergoes a hydroboration-oxidation reaction, the expected product would be
a. Pentan-2-one
b. Pentan-3-one
c. Pentan-4-one
d. Pentanal
Solution: The hydroboration oxidation reaction converts alkynes to aldehydes and ketones by an organic chemical reaction. A two-stage technique, which involves a hydroboration step and an oxidation step, is used to produce this. Using an anti-Markovnikov Rule, a net addition of water is achieved.
When Pent-2-yne is reacted with BH3 followed by H2O2/OH-, the expected product according to the Anti Markovnikov rule is Pentan-2-one. Hence, the correct answer is an option (A).
3. What should be the expected product when cyclohexyne is treated with ozone in the presence of hydrogen peroxide and (dimethyl sulphide and water) separately
a. Cyclohexane-1,2-dione, Cyclohexane-1,6-dioic acid
b. Cyclohexane-1,6-dioic acid, Cyclohexane-1,2-dione
c. Cyclohexane-1,2-dione, Cyclohexane-1,2-dione
d. Cyclohexane-1,6-dioic acid, Cyclohexane-1,6-dioic acid
Solution: When cyclohexyne is treated with ozone in the presence of Hydrogen peroxide (O3 and H2O2), this is oxidative ozonolysis which involves the addition of an ozone molecule to an alkyne to form an ozonide, followed by the oxidation to form a carboxylic acid. So, the product should be Cyclohexane-1,6-dioic acid.
When cyclohexyne is treated with ozone in the presence of dimethyl sulphide and water ((CH3)2S and H2O), this is reductive ozonolysis which involves the addition of an ozone molecule to alkyne to form an ozonide, decomposes easily by reduction into , -diketones. So, the product should be Cyclohexane-1,2-dione.
So, the correct answer should be (B).
4. Predict the product of reaction when Acetylene is treated with ozone molecule in the presence of hydrogen peroxide?
a. Acetic Acid
b. Formic Acid
c. Carbon dioxide
d. Buta-1,3-diene
Solution: This type of alkyne ozonolysis involves the addition of an ozone molecule to an alkyne to form an ozonide, which is then oxidized to smaller oxidized molecules such as acid. Hence, when acetylene is treated with O3 and H2O2, the product should be formic acid.
But formic being unstable further decomposed to form carbon dioxide and water. Hence, the correct answer is an option (C).
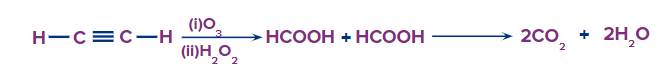
Frequently Asked Questions
1. What is the end result of the alkyne oxidation reaction?
Answer: Oxidations. When alkynes react with oxidizing agents such as potassium permanganate and ozone, the triple bond is usually cleaved, yielding carboxylic acid products. I can also form vicinal diketone in mild conditions with permanganate.
2. Why do alkanes go through substitution reactions but alkenes and alkynes do not?
Answer: Alkanes are already strongly bonded, whereas alkenes and alkynes have weak -bonds. They want to go through addition reactions in order to convert -bonds to stronger's and become more stable.
3. Is alkyne basic or acidic?
Answer: Because alkynes can release hydrogen atoms to form alkyne ions, they are acidic. As a result, the alkyne is used as Brönsted-Lowry acid. As previously stated, alkynes contain a triple-bonded carbon atom known as sp-hybrid.
4. Why do anti-Markovnikov products result from alkyne hydroboration?
Answer: Because the hydrogen (from BH3) attaches to the more substituted carbon and the boron attaches to the least substituted carbon in the alkyne double bond that is substituted by alcohol, the reaction proceeds in an anti-Markovnikov manner.


