-
Call Now
1800-102-2727
Oxidation of Alkenes- Ozonolysis of Alkenes, Ozonolysis Mechanism, Oxidation of Alkenes - Hydroxylation of Alkene, Baeyer’s Reagent (Potassium Permanganate), Practice Problems FAQs
Isn't it true that everyone enjoys vanilla ice cream? Vanilla essence is often used to enhance the aroma of baked goods such as cakes and muffins. Vanilla scent can also be utilised as a room freshener. But do you know what's behind Vanilla Fragrence's chemistry???
Vanillin is the main taste component of vanilla, as its name suggests. This tiny aromatic molecule has three oxygen atoms in separate functional groups: alcohol, aldehyde, and ether.
We'll go through how to make an aldehyde group from an alkene, as well as a variety of other oxidative alkene products.

Table of Contents
- Ozonolysis of alkenes
- Mechanism of ozonolysis reaction of alkene
- Oxidation of Alkenes-Hydroxylation of Alkene
- Baeyer’s Reagent (Potassium Permanganate)
- Practice Problems
- Frequently Asked Questions
Ozonolysis of alkenes
Alkenes have a single pi bond. In the presence of an ozone molecule, the double bond of alkenes undergoes oxidative cleavage. When alkenes are oxidised, end products such as ketones, aldehydes, alcohols, and carboxylic acids are formed.
Consider the following example to better understand the ozonolysis reaction mechanism in an alkene.
Mechanism of ozonolysis reaction of alkene
There are three steps in the ozonolysis reaction mechanism.
Step 1: Attack of the ozone molecule
The ozone molecule consists of three oxygen atoms. One oxygen atom acquires a partial negative charge, while the other gains a partial positive charge.
The negatively charged oxygen ion attacks the double-bonded carbon atom. The reactant is linked to the ozone molecule. Within the structure, rearrangement occurs, resulting in the development of an intermediate structure known as the molozonide.
Step 2: Formation of ozonide intermediate
The molozonide intermediate further rearranges itself resulting in the formation of a stable ozonide intermediate. We can observe that the triple bond is reduced to a single bond in ozonide. Later on, the single bond between two carbon atoms also breaks, yielding different carbonyl compounds.
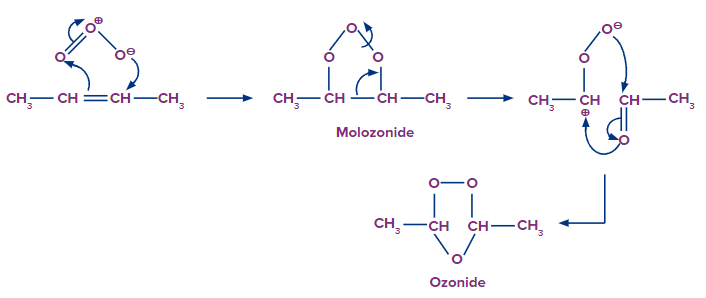
Formation of Molozonide and Ozonide
Step 3: Formation of carbonyl compounds
It can be performed by two types of mechanisms, which are given as follows:
- Reductive ozonolysis
- Oxidative ozonolysis
Reductive ozonolysis
This type of ozonolysis of alkyne involves the addition of an ozone molecule to an alkyne to form an ozonide. Ozonide is unstable and decomposes easily by reduction into , -diketones. The reagent that is used in reductive ozonolysis is given as follows:
(i)
(ii)
(iii)
The formation of , can be given as:
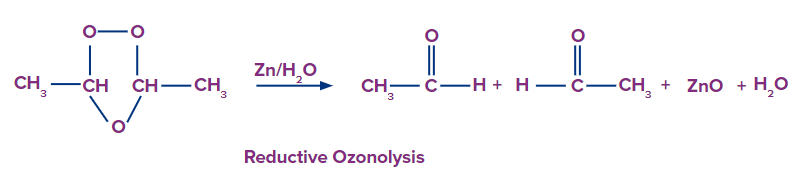
Oxidative Ozonolysis
This type of ozonolysis of alkynes involves the addition of an ozone molecule to an alkyne to
form an ozonide, followed by the oxidation of the ozonide to smaller oxidized molecules like acid. The reagent which is used in oxidative ozonolysis is given as follows:
(i) H2O
(ii) H2O2
The formation of carboxylic acids can be given as:
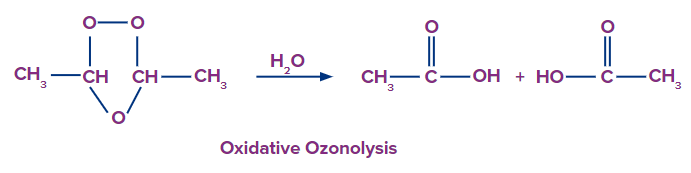
In oxidative ozonolysis, if formic acid is formed, it will further decompose into carbon dioxide and water
Oxidation of Alkenes-Hydroxylation of Alkene
The carbon-hydrogen bond in the alkene oxidises to a carbon-hydroxyl bond during hydroxylation. Alkene hydroxylation is an oxidation reaction. An oxidising agent is a reagent that increases the oxidation number.
Alkenes can be hydroxylated via two distinct stereochemical routes:
- Anti-dihydroxylation
- Syn-dihydroxylation
The anti-dihydroxylation mechanism is used to open epoxides, whereas potassium permanganate or osmium tetroxide produces syn-dihydroxylated products. The osmium tetroxide reaction can also occur in two steps: 1) OsO4 in pyridine, then 2) H2S or NaHSO3.
Anti-Dihydroxylation
Aqueous acid can cleave epoxides which can be formed by using m-CPBA (meta-chloro peroxy benzoic acid). Proton transfer from the acid catalyst produces the epoxide's conjugate acid, which is attacked by nucleophiles such as water. The result is the anti-hydroxylation of the double bond. The hydration of an epoxide has no effect on the oxidation state of any of its atoms or groups. The mechanism of ring-opening in epoxides is determined by the reaction conditions.
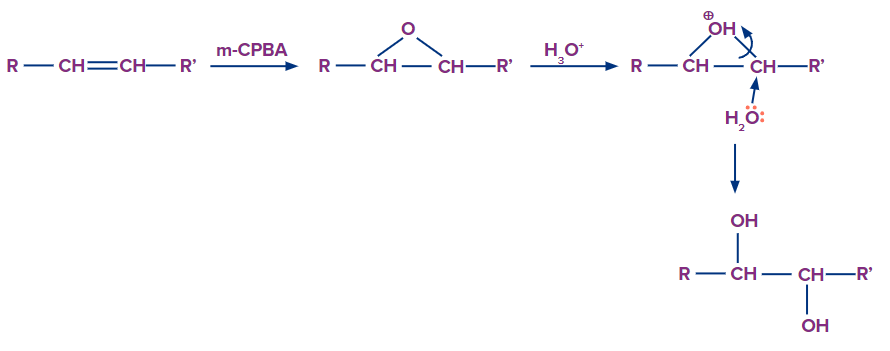
Syn-Hydroxylation
Dihydroxylated products are formed by reacting pyridine with aqueous potassium permanganate or osmium tetroxide. Both reactions appear to be the result of the same mechanism (shown below). We would expect syn-stereoselectivity in the bonding to oxygen based on the mechanism shown here.
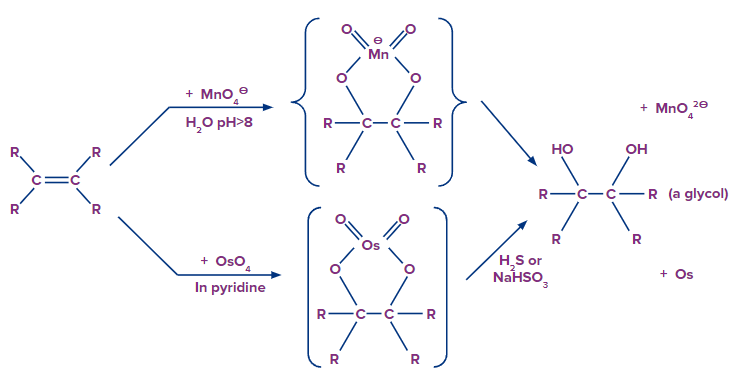
Syn-Hydroxylation
Baeyer’s Reagent (Potassium Permanganate)
Alkenes can be mildly or strongly oxidised depending on the reaction environment. In neutral permanganate solution, for example, alkenes form vicinal diols.
The generic reactions for the various mild oxidative conditions are summarised below.
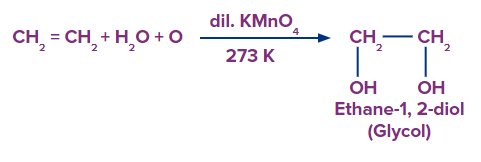
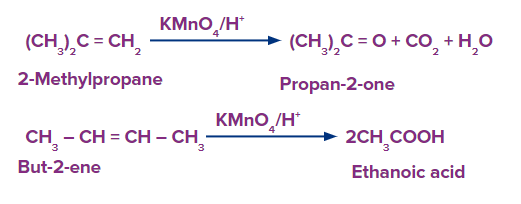
The alkene is cleaved into two products during strong oxidation with acidic potassium permanganate.
If unsaturated carbon is disubstituted, the product is a ketone.
If unsaturated carbon is monosubstituted, the product is a carboxylic acid.
If unsaturation is at the terminal, the product is carbon dioxide and water.
The generic reactions for the various oxidative conditions - strong- are summarised below.
Baeyer’s Test
In qualitative organic analysis KMnO4 is used to test for the presence of unsaturation. It is also known as Baeyer's reagent after the German organic chemist Adolf von Baeyer.
The reagent used in Baeyer’s test is an alkaline potassium permanganate solution.
When potassium permanganate is added to an unsaturated hydrocarbon like alkenes and alkynes, the pink colour of potassium permanganate disappears. The colour fades from purplish-pink to brown when it reacts with double or triple bonds (-C=C- or -C≡C-).
Baeyer’s reagent will give brown precipitates with both terminal and non- terminal alkenes. Hence, this test can be used for the detection of alkenes but does not specify whether they are terminal or non-terminal.
Baeyer’s Reagent Brown precipitates

![]()
Practice Problems
Q1. The metal atom manganese in permanganate occupies the centre of a tetrahedral grouping of negatively charged oxygen atoms. So, how would such a species interact with a double bond's nucleophilic pi-electrons?
Solution: One possible explanation is that an electrophilic metal atom's empty d-orbital extends far beyond the surrounding oxygen atoms and initiates electron transfer from the double bond to the metal, similar to how platinum does. This interaction is completed by back-bonding of the nucleophilic oxygens to the antibonding *-orbital.
Q2. Which test can be used to distinguish between But-2-ene and Butane?
A. Tollen’s Test
B. Ammoniacal cuprous chloride
C. Baeyer’s Test
D. All of these
Solutions: But-2-ene being terminal Alkene and Butane being alkane can be distinguished with baeyer’s tests as in option (A).
Tolen’s test and option (B) Ammoniacal cuprous chloride give precipitates with terminal Alkynes only.
Baeyer’s Reagent Brown precipitates
When alkaline potassium permanganate is added to an unsaturated hydrocarbon, the pink colour of potassium permanganate disappears. The colour fades from purplish-pink to brown when it reacts with double or double bonds (-C=C- or -C≡C-). Baeyer’s reagent will give brown precipitates with both terminal and Non- terminal Alkenes. Hence, this test can be used for the detection of Alkenes.
Hence, the correct option is (C).
Q3. Predict the product of reaction when ethylene is treated with ozone molecule in the presence of hydrogen peroxide?
A. Formaldehyde
B. Formic Acid
C. Carbon dioxide
D. Buta-1,3-diene
Solution: This type of alkene ozonolysis involves the addition of an ozone molecule to an alkene to form an ozonide, which is then oxidized to smaller molecules such as acid. Hence, when ethylene is treated with O3 and H2O2, the product should be formic acid.
But formic being unstable further decomposed to form carbon dioxide and water. Hence, the correct answer is option (C).
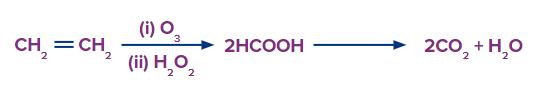
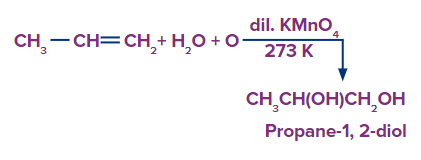
Q4. Predict the product for the given reaction
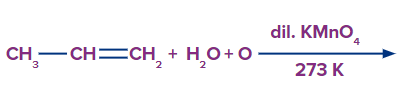
A. Propanal
B. Propane-1,2-diol
C. Ethanoic acid
D. Formic acid
Solution: Alkenes can be gently or strongly oxidised depending on the reaction environment. In neutral permanganate solution, for example, Alkenes form vicinal diols.
Hence, the product is a vicinal diol, Propane-1,2-diol. A correct answer is option (B).
Frequently Asked Questions
Q1. What applications does alkene have?
Answer: They are used in the production of plastics such as polythene for buckets, bowls, and bags and polystyrene for car battery cases and refrigerator parts. They are used in the production of ethane-1,2-diol, which is used as an anti-freeze agent in automobile radiators.
Q2. What role does zinc play in ozonolysis?
Answer: Zinc dust is used in ozonolysis to prevent further oxidation of the chemical. Zn halts the reaction by preventing the molecule from forming additional oxygen bonds.
Q3. Is ozone an electrophile or a nucleophile?
Answer: Molecular ozone has the properties of a dipole, an electrophilic agent, and a nucleophilic agent. The ozone molecule's dipolar structure may result in 1-3 dipolar cycloadditions on unsaturated bonds, resulting in the formation of primary ozonide.
Q4. Which reagent is more useful, osmium tetroxide or potassium permanganate?
Answer: Because OsO4 is toxic, potassium permanganate, KMnO4, was used instead. Although syn diols are produced by the reaction of KMnO4 and an alkene, potassium permanganate is less useful due to low product yields due to overoxidation.


