-
Call Now
1800-102-2727
Oxidation Number Method- Introduction to Redox Reaction, Oxidation Number Method, Practice Problems & FAQs
You must be aware of the fact that your parents get income in the form of salary and they divide the money in a certain proportion that they invest some amount of it and spend the rest of their salary in day to day life. The invested money is savings which helps in the future in case of an emergency and money spent depreciates our savings. For a balanced life, investing and spending should go hand in hand.
The same goes with the redox reactions, these reactions need to be balanced. But why do we need to balance the redox reactions?
Let’s see what is the importance of balancing a redox reaction and learn about an important method to balance these reactions.
Table of Contents
- What is an Oxidation Number Method?
- Balancing a Redox Reaction by Oxidation Number Method in Acidic Medium
- Balancing a Redox Reaction by Oxidation Number Method in Alkaline Medium
- Practice Problems
- Frequently Asked Questions-FAQs
What is an Oxidation Number Method?
This method is based on the change in the oxidation number of the reducing agent and the oxidizing agent followed by balancing the elements in which the oxidation state does not change by the normal hit and trial method.
Generally, there are two different mediums in which redox reactions can occur.
- In acidic medium
- In alkaline medium
Balancing a Redox Reaction by Oxidation Number Method in Acidic Medium
Let’s see the rules for balancing a redox reaction by oxidation number method in an acidic medium:
Take this reaction as an example which is occurring in an acidic medium.
Step 1: Identify the atoms which are oxidized and reduced with the help of the oxidation state of the element present in the reactant and product side.
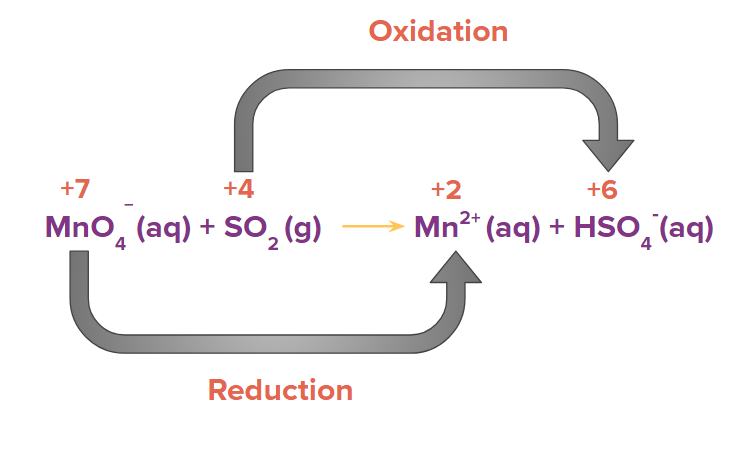
Step 2: Calculate the change of electrons (i.e., loss or gain) in the identified species which are getting oxidised and reduced.
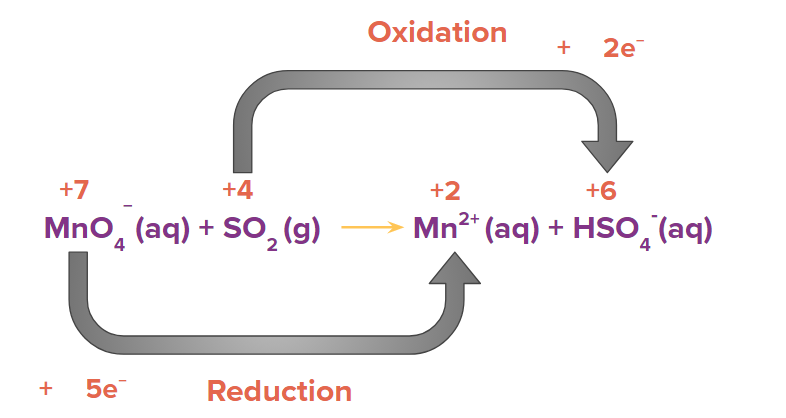
Step 3: Equate the number of electrons gained and lost in the given reaction by multiplying it with a suitable factor such that the number of loss of electrons and the number of gain of electrons becomes equal.

Step 4: Balance all other atoms except O and H by hit and trial method but in this case, all other atoms are already balanced.
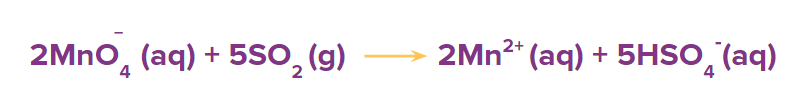
Step 5: Balance the oxygen atoms by adding the H2O molecules at the oxygen-deficient side of the reaction.
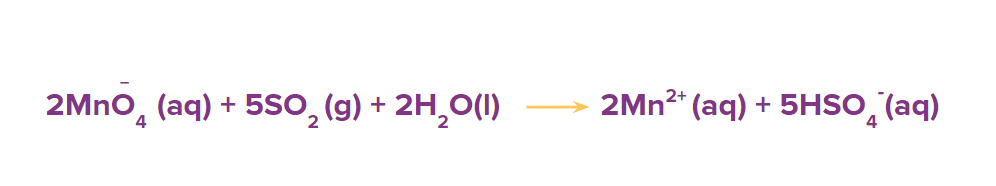
Step 6: To balance the hydrogen atoms, add H+ ions to the hydrogen atoms deficient side of the reaction.
Balancing a Redox Reaction by Oxidation Number Method in Alkaline Medium
Let’s see the rules for balancing a redox reaction by oxidation number method in alkaline medium:
Take this reaction as an example which is occurring in alkaline medium.
Step 1: Identify the atoms which are oxidized and reduced with the help of the oxidation state of the element present in the reactant and product side.
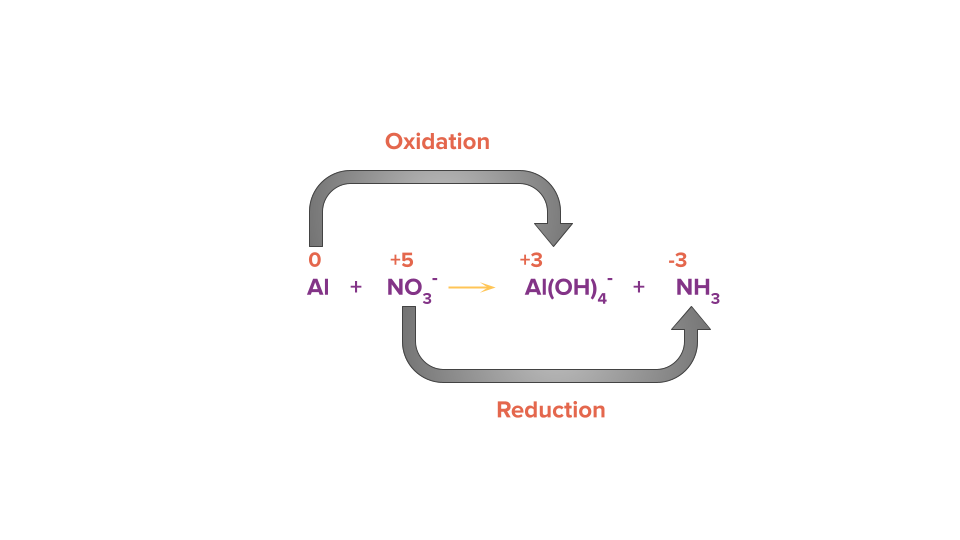
Step 2: Calculate the change of electrons (i.e., loss or gain) in the identified species which are getting oxidised and reduced.
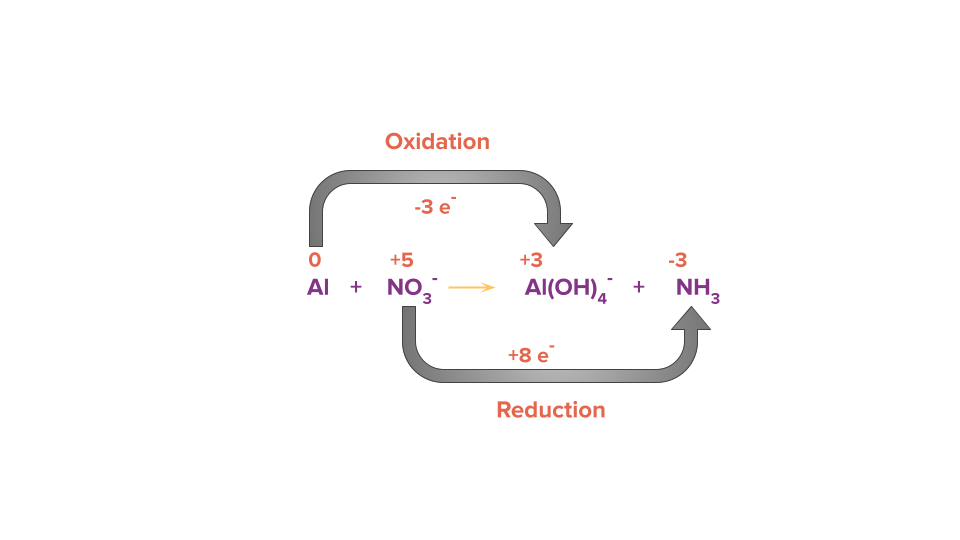
Step 3: Equate the number of electrons gained and lost in the given reaction by multiplying it with a suitable factor such that the number of loss of electrons and the number of gain of electrons becomes equal.
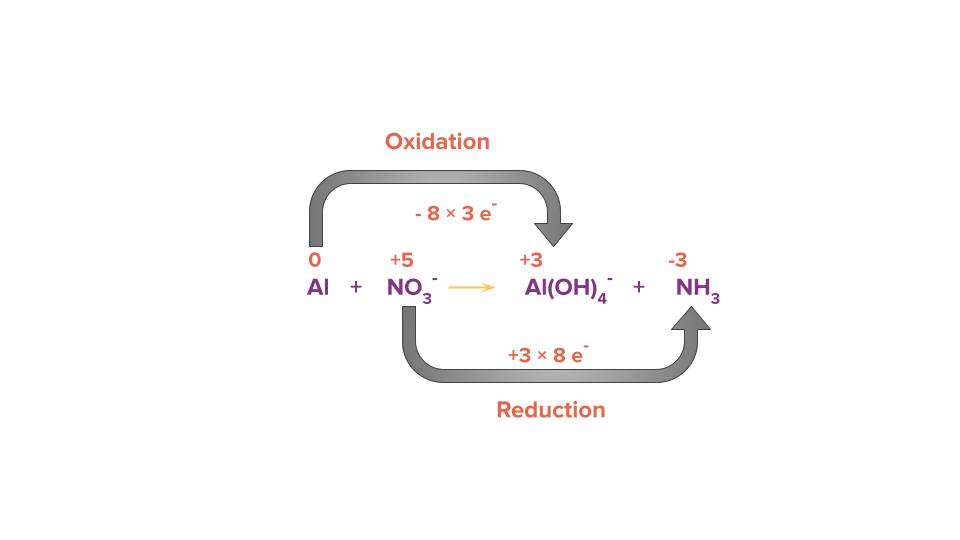
Step 4: Balance all other atoms except O and H by hit and trial method but in this case, all other atoms are already balanced.

Step 5: Balance the oxygen atoms by adding the H2O molecules at the oxygen-deficient side of the reaction.

Step 6: To balance the hydrogen atoms, add H+ ions to the hydrogen atoms deficient side of the reaction.

Step 7: Since the reaction is taking place in the basic medium, add the same number of OH- ions to both sides of the reaction which is equal to the number of H+ ions added in the previous step.

The final reaction would look like:
![]()
Practice Problems
1. Find the coefficients of the reaction. a Fe2+(aq) + b Cr2O72-(aq)c Fe3+(aq)+d Cr3+(aq) (acidic medium)
a. a=6, b=1, c=6, d=2
b. a=2, b=6, c=6, d=1
c. a=4, b=4, c=5, d=2
d. a=1, b=3, c=6, d=3
Answer: (A)
Solution: According to the given question,
Step 1:
|
Chemical species |
Reactant (O.S) |
Product (O.S) |
|
|
Fe |
+2 |
+3 |
oxidation |
|
Cr |
+6 |
+3 |
reduction |
Step 2:
Number of electrons involved in oxidation = 1
Number of electrons involved in reduction = 3 2=6
Multiply both the Fe by 6 to equalise the number of electrons gained and lost in the reaction.
Step 3: Balance the other atoms except for O and H by hit and trial method. In the given reaction, Cr is balanced by multiplying by 2 on the product side.
Step 4: Balance the oxygen atoms by adding H2O molecules in the product side.
Step 5: As the reaction takes place in the acidic medium, hydrogen atoms are balanced by adding (H+) on the hydrogen-deficient side.
Comparing equations (i) and (ii),
a=6, b=1,c=6,d=2
So, option (A) is the correct answer.
2. Find the coefficients of the reaction,
a H2O2+b ClO2+c OH- d ClO2-+e O2+f H2O ( basic medium).
a. a=2, b=2, c=2, d=2, e=1, f=2
b. a=1, b=2, c=2, d=2, e=2, f=2
c. a=1, b=1, c=2, d=2, e=3, f=2
d. a=1, b=2, c=2, d=2, e=1, f=2
Answer: (D)
Solution: According to the equation,
a H2O2+b ClO2+c OH- d ClO2-+e O2+f H2O....(iii)
Step 1:
|
Chemical species |
Reactant (O.S) |
Product (O.S) |
|
|
O (H2O2) |
-1 |
0 |
oxidation |
|
Cl ( ClO2) |
+4 |
+3 |
reduction |
Step 2: Number of electrons involved in oxidation = 2(0-(-1)=2
Number of electrons involved in reduction = 1
Multiply both the Cl by 2 to equalise the number of electrons gained and lost in the reaction.
H2O2+ 2ClO2+ OH- 2ClO2-+ O2+ H2O
Step 3: Now balance other atoms except O and H as the chlorine atom is already balanced. Therefore the reaction will be represented as;
H2O2+ 2ClO2+ OH- 2ClO2-+ O2+ H2O
Step 4: Balance the oxygen atom by adding the H2O molecule, but since the number of oxygen atoms are same at both sides. No, water molecule is added.
H2O2+ 2ClO2+ OH- 2ClO2-+ O2+ H2O
Step 5: To balance the hydrogen atoms, add H+ ions at the hydrogen atoms deficient side of the reaction.
H2O2+ 2ClO2+ OH- 2ClO2-+ O2+ H2O+2H+
Step 6: Since the reaction is taking place in the basic medium, add the same number of OH- ions to both sides of the reaction which is equal to the number of H+ ions added in the previous step.
H2O2+ 2ClO2+ 2OH- 2ClO2-+ O2+2 H2O.....(iv)
Comparing equations (iii) and (iv), we get;
a=1, b=2,c=2,d=2, e=1, f=2
So, option (D) is the correct answer.
Q 3. What will be the value of abcd in the given reaction a H2O2+b I2c HIO3 +d H2O if the reaction takes place in acidic medium?
a. 5
b. 85
c. 8
d. 58
Answer: (D)
Solution: According to the equation,
a H2O2+b I2c HIO3 +d H2O.....(v)
Step 1:
|
Chemical species |
Reactant (O.S) |
Product (O.S) |
|
|
O (H2O2) |
-1 |
-2 |
oxidation |
|
I ( I2) |
0 |
+5 |
reduction |
Step 2: Number of electrons involved in oxidation = |2(-2-(-1)|=2
Number of electrons involved in reduction = 5 x 2 = 10
Multiply both the O by 5 to equalise the number of electrons gained and lost in the reaction.
5 H2O2+ I2 HIO3 +5 H2O
Step 3: Now balance other atoms except O and H. In the given reaction balancing the iodine atoms by hit and trial method, we get;
5 H2O2+ I2 2HIO3 +5 H2O
Step 4: Balance the oxygen atom by adding the H2O molecule. In the given reaction adding one in the reactant side, we get;
5 H2O2+ I2+H2O 2HIO3 +5 H2O
Can be written as:
5 H2O2+ I22HIO3 +4 H2O
Step 5: To balance the hydrogen atoms, add H+ ions at the hydrogen atoms deficient side of the reaction. In the above reaction hydrogen atom is already balanced and the overall reaction becomes,
5 H2O2+ I2 2HIO3 +4 H2O.....(vi)
Comparinig equations (v) and (vi), we get;
a=5, b=1,c=2,d=4,
Therefore, the value of (abcd) will be 58.
So, option (D) is the correct answer.
Q 4. Select the incorrect statement regarding the redox reaction.
a. All decomposition reactions are redox in nature
b. Sodium chloride formed from the constituent element is an example of redox reaction
c. Decompostion of lithium nitrate is a redox reaction
d. None of these
Answer: (A)
Solution: All decomposition reactions are not redox reactions.
CaCO3(s)CaO(s)+ CO2(g)
|
Element |
Initial O.S |
Final O.S |
|
Ca |
+2 |
+2 |
|
O |
-2 |
-2 |
|
C |
+4 |
+4 |
No change in O.S of atoms involved in the reaction.
So, option (A) is the correct answer.
Frequently Asked Questions-FAQs
Q. What is an oxidising agent?
Answer: A substance that can oxidise others and reduce itself in a redox chemical reaction or a reagent that gains electrons in a redox reaction is called an oxidising agent.
Q . What is a reducing agent?
Answer: A substance that can reduce others and oxidise itself in a redox chemical reaction or a reagent that loses electrons in a redox reaction is called a reducing agent.
Q. How can we determine if the oxidation or reduction has taken place in a redox reaction?
Answer: The oxidation number, also known as oxidation states, keeps track of the electrons gained and lost when a substance is reduced and oxidised. An oxidation number is assigned to each atom in a neutral molecule or charged species. If the element accepts the electrons, it is said to be reduced, and if it loses the electrons, it is said to be oxidised.


