-
Call Now
1800-102-2727
Osmotic Pressure: Osmotic Pressure, Semipermeable Membrane, Measurement of Osmotic Pressure, Colligative Nature and Derivation of Osmotic Pressure, Examples of Osmotic Pressure, Practice Problems & Frequently Asked Questions
You are given a vessel with a partition which will allow only water molecules to pass through both sides. Now, you add water to one part of the vessel only. After some time what will be the level of water on the other side of the vessel? Still empty or level less or equal or more than the water containing part?
Now take water such that the water levels on both sides of the partition are the same. Add, say 10 gram of NaCl salt to one side of the vessel. Will the water level on both sides be the same or different? If different, which side salt containing or pure water side will be higher in height? Difficult to guess unless you know the term osmotic pressure.
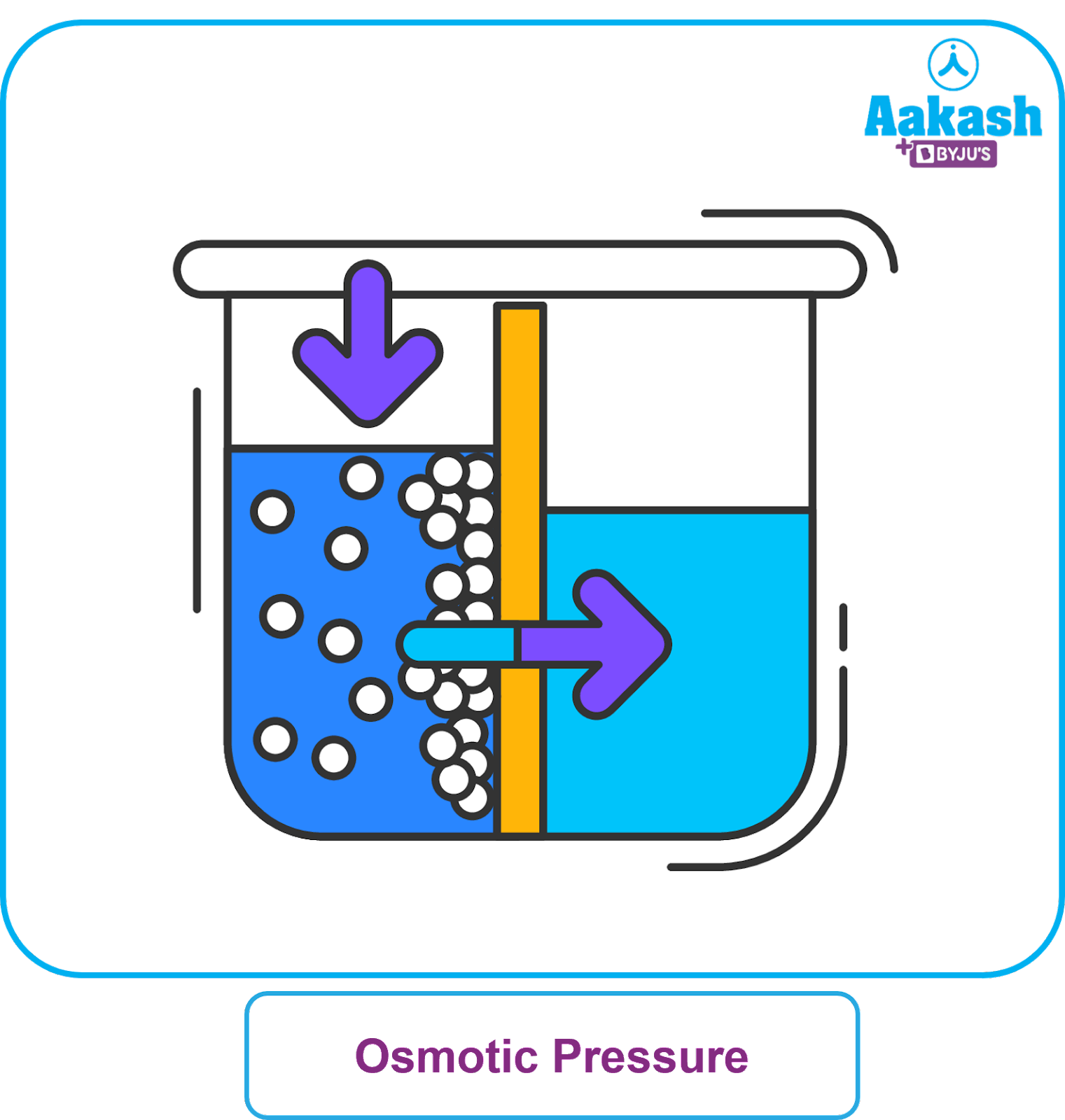
Is it just an experiment? No. It is of crucial importance for living who has to maintain a certain amount of water in and around the cells of the body.
During exercises or dehydration, the body loses water increasing the salt content in and around cells, which unless otherwise compensated will cause seizure, shock, coma or even death.
Ok, what if I drink excess water? That is also bad, because of the reverse effect, more water entering the cell leading to confusion, headaches, high blood pressure, low heart rate, bursting of cells, coma and even death.
It is all because the water exerts an amount of pressure called Osmotic pressure.
Come and know about this life deciding factor-Osmotic pressure
Table of content:
- Osmotic pressure
- Semipermeable membrane Types
- Measurement of osmotic pressure
- Colligative nature and derivation of osmotic pressure
- Examples of osmotic pressure
- Practice problems
- Frequently asked questions
Osmotic pressure:
Osmosis is the flow of solvent molecules from a high solvent concentration region to a low solvent concentration region through a semipermeable membrane.The force or pressure which is required to prevent such flow of solvent across semipermeable membrane to the solution side is called osmotic pressure.
A semipermeable membrane is a selective barrier containing microscopic pores that allow only the small solvent molecules but not large solute molecules to flow through it.
The partially permitting semi permeable substance used initially was an animal leather and so called semi permeable membrane. But it can be either natural from plant or animal sources or synthetically made polymers.
Semipermeable membrane types:
Natural semi-permeable membrane: The natural semi-permeable membrane is made up of animal and plant membranes found beneath the outer skin of animals and plants, respectively.
Artificial or synthetic semi-permeable membrane: Artificial or synthetic semi-permeable membranes include parchment paper, cellophane, and freshly created inorganic compounds such as copper ferrocyanide supported by the walls of a porous pot.
Measurement of Osmotic pressure:
Osmotic pressure of a solution is measured by the force or pressure to be applied to stop the movement of solvent molecules from solvent side to solution side.
- Take equal volume of pure solvent and a solution in a vessel separated by a semipermeable membrane.
- The volume of solvent and the level of the pure solvent decreases while the volume and the level on the other side of the solution proportionately increases initially and comes to a halt after some time.
- Now apply enough pressure on the solution side to make once again the level on both solvent and solution side to be equal as before.
- The force exerted to achieve this is the osmotic pressure of the solution.

![]()
The osmotic pressure of a solution is proportional to its concentration.
Colligative nature and Theoretical derivation of Osmotic pressure:
Van't Hoff observed that osmotic pressure of a dilute solution is proportional to the molarity C of the solution at a given temperature T. Osmotic pressure being dependent on the concentration , it is a colligative property. The relation can be written as,
∝ C T
= C R T … … … … … (1)
Where = Osmotic pressure
R = is the proportionality constant = Universal gas constant
… … … … … (2)
Where V is the volume of the solution in L containing nB mol of solute.
If WB grams of the solute of molar mass MwB is present in the solution, then
… … … … … (3)
Substituting (3) in (2),
Or … … … … … (4)
Thus, if other quantities like WB, T, and V are known, we can calculate the molar mass of the unknown solute.
The influence of solutes on the colligative properties of solutions is explained by the Van't Hoff factor. The Van't Hoff factor is represented by the letter "i." The amount that a substance associates or dissociates in a solution is described by the Van't Hoff factor.
The relationship between van’t Hoff factor and osmotic pressure is given by the below equation
= Osmotic pressure
R = is the proportionality constant = Universal gas constant
i= Van’t Hoff factor
C = concentration of the solution.
Types of solutions based on osmotic pressure
- Isotonic solution: Two solutions having the same osmotic pressure are termed isotonic solutions.
- Hypertonic solution: A solution having a higher osmotic pressure relative to some other solution is called a hypertonic solution.
- Hypotonic solution: A solution having a lower osmotic pressure relative to some other solution is called a hypotonic solution.
Examples of osmotic pressure:
Transport in plants: Plants absorb water through their roots, and water travels from the roots to various areas of the plant. Every root works as a semipermeable barrier, allowing water molecules to move from a high concentration (soil) to a low concentration (plant) (roots). Hair on the roots improves surface area and thus water intake by the plants.
Wrinkled fingers: When we sat in the bathtub or submerged our fingers in water for an extended period of time, they became wrinkly. This is due to osmosis. The skin on our fingertips absorbs water and expands or bloats, resulting in pruned or wrinkled fingers.
Wilting plants: The majority of plants rely on osmotic pressure to keep their stems and leaves in shape. If we have kept potted plants, we are surely aware that if they are not watered for an extended period of time, they might get very wilted. They can perk them right back up within minutes after watering!
Many plant stems and leaves are basically "inflated" by osmotic pressure; the salts in the cells allow water to be sucked in through osmosis, making the cell plump and hard. If there is insufficient water, the plant will wilt because its cells get "deflated." They are "hypertonic," which implies "the concentration of solute is too high" in scientific words.
Plants, too, can demonstrate the strength of osmotic pressure as they grow. Plants may have sprung through asphalt, or tree roots may have grown through concrete or bricks. This could also be caused by osmotic pressure: when plants expand, their cells draw in more water. The slow but constant pressure of water passing through the membranes of plant cells can really push through asphalt!
Practice problems:
Q.1. 0.2 L of an aqueous solution contains 1.26 g of a polymer. The osmotic pressure of solution is 2.57 ✕10-3 bar at 300 K. Calculate the molar mass of the polymer.
Solution:
= 1.26 ✕ 0.083 ✕ 300 0.2 ✕ 2.57 ✕10-3
Q.2. What is the volume of solution containing 1 g mole of sugar that will give rise to an osmotic pressure of 1 atm at 273 K?
Solution:
V= 1 ✕ 0.0821 ✕ 273 1
V= 22.4 L
Q.3. Calculate the concentration of an aqueous solution of a nonelectrolyte having an osmotic pressure of 2.0 atm at 300 K ?
Solution: = C R T
Q.4. 0.6 g urea (molar mass =60 g mol-1) and 1.8 g glucose (molar mass =180 g mol-1) are dissolved in 100 mL of water at 27oC to prepare a solution. The solution's osmotic pressure is (R=0.0821 L atm K-1 mol-1).
Solution: Number of moles of urea
Number of moles of glucose
Frequently asked questions:
1. Which colligative property is appropriate for determining protein and polymer molar mass?
Answer: Osmotic pressure is the most accurate method for determining the molar mass of proteins and polymers. Because a small amount of solute causes a significantly larger change in this quantity than in the boiling point, freezing point, or vapor pressure. Even a very small
10-6 molar solution would have measurable osmotic pressure. Proteins and other high molecular weight polymers are regularly subjected to molecular weight determinations. Because of their huge molecular size, these compounds are only sparingly soluble in most solvents, hence measuring osmotic pressure is often the only feasible approach to determine their molecular weights.
2. Why is osmotic pressure temperature-dependent?
Answer: Osmotic pressure, as we already know, is the external pressure provided to a solution to prevent the solvent from migrating from a higher concentration area to a lower concentration area.
We know that
At constant concentration we can write as
At constant temperature we can write as
As a result, osmotic pressure is proportional to temperature and solute molar concentration.
3. How is osmosis used to preserve food?
Answer: Sugar is used to preserve fruits. To keep fruits from deteriorating, honey is widely used to preserve them. Sugar is often employed as a preservative in jams and jellies made from fruits nowadays. Jellies are clear liquids made of fruit juice or fruit extract. Because sugar produces a hypertonic solution, it draws water from food, rendering it unavailable to germs. Microbial metabolism is inhibited, and they perish as a result of water loss.
4. Is Osmosis a Cause of Cholera?
Answer: Osmosis enables bad things to occur. Cholera would not be possible without osmosis. Choleric bacteria populate our intestines and begin to invert the ionic orientation of the intestinal cells. In other words, it alters the way ions and, as a result, water transit in our intestines. As a result, cholera pulls out a flawless coup.
For starters, as our ions' orientations change, the intestinal cells lose their ability to absorb water into the body. Osmosis now occurs in the other manner, with water moving from our intestinal cells into our intestines. This results in cholera's infamously lethal watery diarrhea. Second, this increases the rate at which you dehydrate. You are not only unable to absorb water, but you are also being drained dry. As a result, cholera can kill you considerably faster because it is not affected by how much water you consume.


