-
Call Now
1800-102-2727
Limitations of Octet Rule – Octet Rule, its Limitations, Practice Problems and FAQ
Various hypotheses have been proposed to explain the periodic table, atomic structure, the electronic configuration of elements, and molecule bonding. The Kossel-Lewis theory, the octet rule, the valence bond theory (VBT), the valence shell electron pair repulsion theory (VSEPR), and the molecular orbital theory (MOT) are a few of them.
Although each of these ideas has advantages and disadvantages, they frequently provide the right answers. Some of them are helpful in anticipating the structure of molecules that have not yet been discovered or identified as well as in describing the forms of molecules that are known to exist.
Understanding these theories is crucial for comprehending the nature of molecular bonding.
On this concept page, we will be discussing one such theory known as the ‘Octet Rule’ and its limitations.
TABLE OF CONTENTS
- Octet Rule – Introduction
- Octet Rule – Limitations
- Sugden's View of Singlet Linkages
- Practice Problems
- Frequently Asked Questions – FAQ
Octet Rule – Introduction
In 1916, W. Kossel and G. N. Lewis created the essential theory of chemical combination between atoms known as the electronic theory of chemical bonding. According to this theory, atoms can combine by either transferring valence electrons (gaining or losing) from one atom to the other or by sharing the valence electrons, so as to gain octet configuration (stable noble gas configuration) in their valence shell. octet (stable noble gas configuration). This is known as the octet rule.
Alternatively, an atom is more stable when itrs valence shell is complete. The K shell (n=1) is complete when it contains two electrons, and the other shells are complete when they have eight electrons. This is called the Lewis octet rule.
Octet Rule – Limitations
The octet rule has many applications, but there are also many situations in which it is not appropriate.
Below are a few significant exemptions to the octet rule:
1. Hydrogen is not able to complete its octet due to a lack of higher shells
In its first shell, hydrogen possesses one electron (n = 1). Since the first shell can only hold two electrons, this shell just needs one more electron to fill it. The configuration after filling becomes 1s2 and by comparing it with the noble gas helium, we can say it is quite stable. Therefore, an octet is not required in this situation to ensure a stable configuration.
2. Incomplete octet of the central atom
Certain molecules of lithium, beryllium, boron, and aluminium do not form compounds with a complete octet. They are basically electron-deficient compounds in which the central atoms possess less than eight electrons in their valence shell.
Examples
LiCl: Li has 1 e- and Cl has 7 e- in their outermost shell respectively. By sharing, Cl attains a noble gas configuration, but Li contains only 2 e- in its valence shell, and has an incomplete octet.
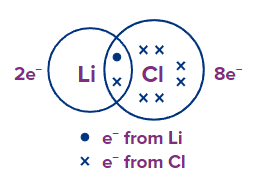
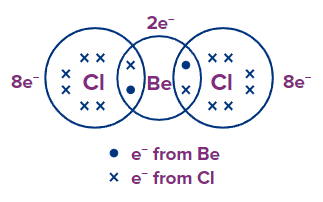
BeCl2: Be has 2 e- and each Cl has 7 e- in their outermost shell respectively. By sharing, each Cl atom attains an octet configuration, but Be contains only 4 e- in its valence shell, and has an incomplete octet.
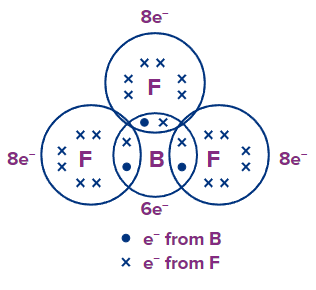
BF3: B has 3e- and each F has 7 e- in their outermost shell respectively. By sharing, each F atoms attain an octet configuration, but B contains only 6 e- in its valence shell, and has an incomplete octet.
3. Extended octet of the central atom
Certain molecules of phosphorous, sulphur, iodine, etc, do not form compounds with an octet. They are basically electron-rich compounds in which the central atoms possess more than eight electrons in their valence shell.
Examples
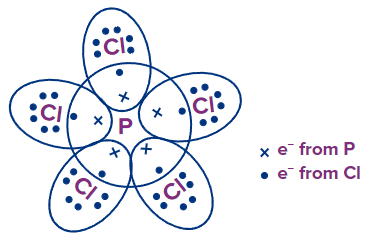
PCl5: P has 5 e- and each Cl has 7 e- in their outermost shell, respectively. By sharing, each Cl atom attains an octet configuration but P contains 10 e- in its valence shell, and hence, has an extended octet.
SF6: S has 6 e- and each F has 7 e- in their outermost shell, respectively. By sharing, each F atom attains an octet configuration but S contains 12 e- in its valence shell, and hence, has an extended octet.
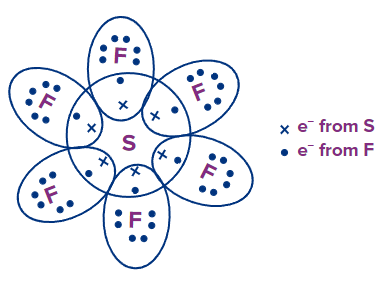
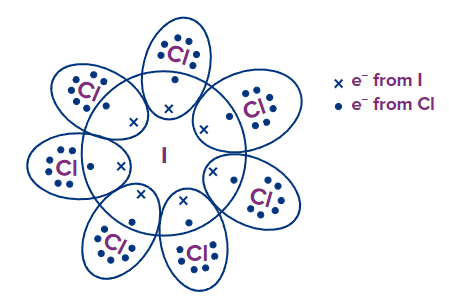
ICl7: I has 7 e- and each Cl has 7 e- in their outermost shell, respectively. By sharing, each Cl atom attains an octet configuration but I contains 14 e- in its valence shell, and hence, has an extended octet.
4. Odd electron atoms
Nitric oxide and nitrogen dioxide are two examples of molecules with an odd number of electrons. In certain situations, not all of the atoms satisfy the octet rule.
5. It should be remembered that the octet rule is based on the chemical inertness of noble gases. It has been discovered, nevertheless, that some noble gases, particularly xenon and krypton, can also interact with oxygen and fluorine to create a wide variety of compounds etc.
6. The shape of the molecules is not explained by this theory.
7. It is impossible to interpret the relative stability of the molecule in terms of energy.
Sugden's View of Singlet Linkages
According to Sugden's theory, the central atom's valence shell can only contain a maximum of 8 electrons. A complete octet is always present in a central atom, which is combined with other atoms not only by covalent bonds but also by singlet linkages, which involve the sharing of just one electron. This singlet linkage is also known as single electron linkage or half-bond.
This kind of electron linkage is represented by a half arrow (⇀).
Example: In PCl5, there are three covalent bonds which are formed by sharing one electron from phosphorous and one electron from chlorine. There are two singlet linkages in which one electron is shared by phosphorus with chlorine as shown below.
Recommended Videos
Kossel Lewis Theory Class 11 Chemistry Chemical Bonding and Molecular Structure (Ch-4) | JEE Man
Practice Problems
1. Which of the following compounds is considered to have an extended octet of the central atom?
a. NH3
b. CH4
c. PF5
d. PCl3
Answer: C
Solution: Certain molecules do not form compounds with a complete octet and their central atoms possess more than eight electrons in their valence shell. They are basically electron-rich compounds.
1. NH3: N has 5 e- and each H has 1 e- in their outermost shell, respectively. By sharing, the central atom contains 8 e- in its valence shell, and hence, has a complete octet.
2. CH4: C has 4 e- and each H has 1 e- in their outermost shell, respectively. By sharing, the central atom contains 8 e- in its valence shell, and hence, has a complete octet.
3. PF5 : P has 5 e- and each F has 7 e- in their outermost shell, respectively. By sharing, the central atom contains10 e- in its valence shell, and hence, has an extended octet.
4. PCl3: P has 5 e- and each Cl has 7 e- in their outermost shell, respectively. By sharing, the the central atom contains 8 e- in its valence shell, and hence, has a complete octet.
So, option C is the correct answer.
2. Which of the following compounds is considered to have an incomplete octet of the central atom?
a. H2O
b. AlCl3
c. SF4
d. OF2
Answer: B
Solution: Certain molecules do not form compounds with a complete octet and their central atoms possess less than eight electrons in their valence shell. They are basically electron-deficient compounds.
1. H2O: Each H atom has 1 e- and O has 6 e- in their outermost shell, respectively. By sharing, the central atom contains 8 e- in its valence shell, and hence, has a complete octet.
2. AlCl3: Al has 3 e- and each Cl has 1 e- in their outermost shell, respectively. By sharing, the central atom contains 6 e- in its valence shell, and hence, has an incomplete octet.
3. SF4: S has 6 e- and each F has 1 e- in their outermost shell, respectively. By sharing, the central atom contains 10 e- in its valence shell, and hence, has an extended octet.
4. OF2: O has 6 e- and each F has 1 e- in their outermost shell, respectively. By sharing, the central atom contains 8 e- in its valence shell, and hence, has a complete octet.
So, option B is the correct answer.
3. How many singlet linkages are present in IF7 as per Sugden’s theory of singlet linkages?
a. 5
b. 6
c. 7
d. 4
Answer: B
Solution: In IF7, there are single covalent bond present, which are formed by sharing one electron from iodine and one electron from fluorine. There are six (6) singlet linkages in which one electron is shared by iodine with fluorine.
So, option B is the correct answer.
4. Which of the following is/are the true statement(s) about Octet's theory and its limitations?
a. When atoms are joined together by chemical bonds, they form a stable octet.
b. The shape of the molecules is not explained by this idea.
c. Octet is not required in case of hydrogen to make a stable compound.
d. All of these
Answer: D
1. According to the Octet theory, atoms can combine by either transferring valence electrons (gaining or losing) from one atom to the other or by sharing the valence electrons, so as to gain octet configuration (stable noble gas configuration) in their valence shell. octet (stable noble gas configuration). This is known as the octet rule.
2. In its first shell, hydrogen possesses one electron (n = 1). Since the first shell can only hold two electrons, this shell just needs one more electron to fill it. The configuration after filling becomes 1s2 and by comparing it with the noble gas helium, we can say it is quite stable. Therefore, an octet is not required in this situation to ensure a stable configuration.
3. The shape of the molecules is not explained by this theory.
So, option D is the correct answer.
Frequently Asked Questions – FAQ
1. What are hypervalent compounds?
Answer: Hypervalent compounds are formed by main group elements that form more bonds than would be predicted by the octet rule. These compounds feature what is referred to as an "extended octet," which means that there are more than eight electrons surrounding each atom. They are basically electron-rich compounds.
2. What are hypovalent compounds?
Answer: Hypovalent compounds are formed by elements that form less bonds than would be predicted by the octet rule. These compounds feature what is referred to as an "incomplete octet," which means that there are less than eight electrons surrounding each atom. They are basically electron-deficient compounds.
3. What are electron-precise compounds?
Answer: Electron-precise compounds are formed by elements that form exactly the same bonds as would be predicted by the octet rule. These compounds feature what is referred to as a "complete octet," which means that there are eight electrons surrounding each atom.
4. What is the need for the octet rule?
Answer: The octet rule is adhered to by atoms because they constantly seek the most stable electron configuration. Hence, as a result of the octet rule, the outermost energy level of an atom has fully filled s- and p-orbitals which helps to gain stability.


