-
Call Now
1800-102-2727
Nucleophilic Substitution Reaction: Definition, Types, SN1, SN2, SNi, SNNGP, Practice problems and FAQs
Have you ever traveled in a metro or in a bus or in a local train? Getting a seat in a metro always feels like a victory. Sometimes when the metro is vacant, we get a seat easily but sometimes we have to wait for the other person for a seat to get vacant. Also, it may happen that people themselves offer us a seat if someone is not feeling well.
Consider this as an analogy for nucleophilic substitution reaction, where one atom/group of atoms (leaving group) from the substrate is replaced by another atom/group of atoms (nucleophile).
Let's study in detail what are nucleophilic substitution reactions and what all types of nucleophilic substitution reactions are possible?
TABLE OF CONTENTS
- What is Nucleophilic Substitution Reaction?
- Types of Nucleophilic Substitution Reaction
- SN1 Reaction
- SN2 Reaction
- SNi Reaction
- SNNGP Reaction
- Practice Problems
- Frequently Asked Questions - FAQs
What is Nucleophilic Substitution Reaction?
If a substitution reaction is brought about by the nucleophile, then it is known as a nucleophilic substitution reaction. The general reaction representing the nucleophilic substitution reaction is shown below:
Where Nu is a Nucleophile and lg is the leaving group.
Example-
Types of Nucleophilic Substitution Reaction
Based on the nature of substrate, reagents and reaction conditions nucleophilic substitution reactions are categorized into four types:
1. SN1 Reaction
2. SN2 Reaction
3. SNi Reaction
4. SNNGP Reaction
Let's study each of these reactions in detail.
SN1 Reaction
SN1 reaction is a unimolecular nucleophilic substitution reaction. Thus, the SN1 reaction mechanism follows a step-by-step reaction wherein the first step involves the formation of the carbocation which is formed by the removal of the leaving group. The carbocation intermediate thus formed, is attacked by the nucleophile. This is followed by the deprotonation of the protonated nucleophile to give the required product.
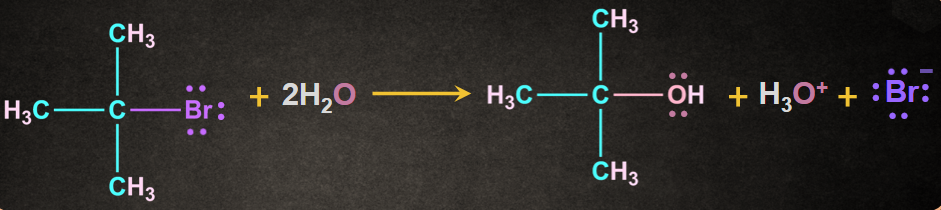
An intermediate formed during this reaction is the carbocation intermediate. SN1 reaction is generally given by tertiary and secondary alkyl halides. An example of SN1 reaction is shown as below:
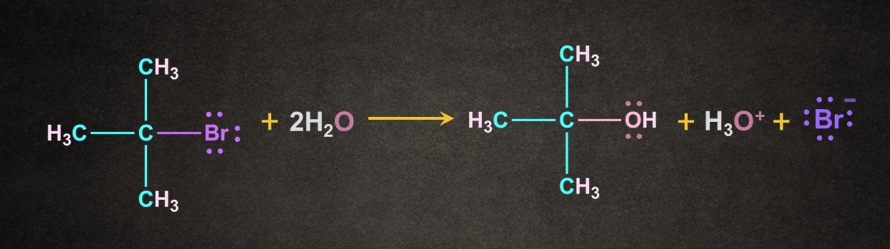
Mechanism of SN1 Reaction:
SN1 reaction mechanism involves two or more steps.
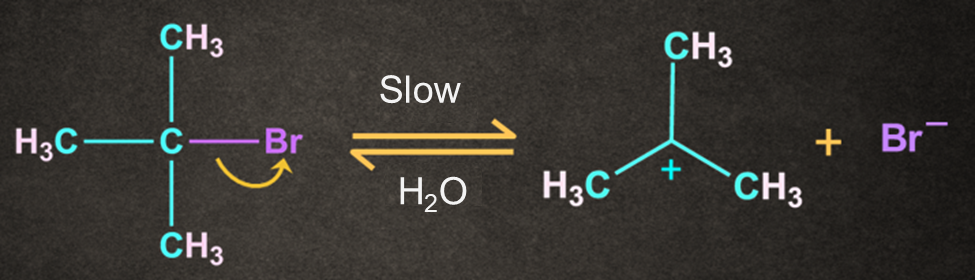
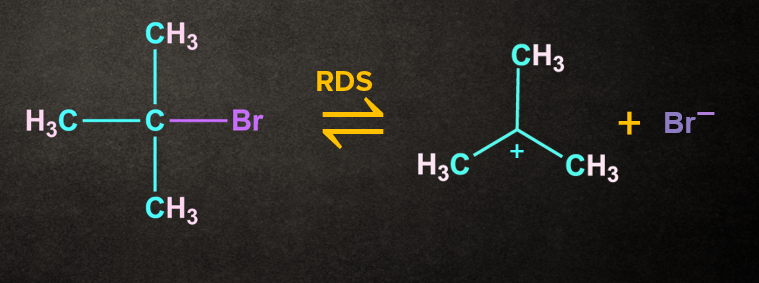
Step 1: Formation of carbocation
The carbon and bromine form a polar covalent bond. The cleavage of the carbon and bromine bond permits the leaving group to be removed as a bromide ion
A carbocation intermediate is formed when the bromide ion(Br-) departs from the tertiary butyl bromide.
This step in SN1 the mechanism is the slowest step, hence it is a rate-determining step.
The breaking of the carbon-bromine bond is endothermic in nature.
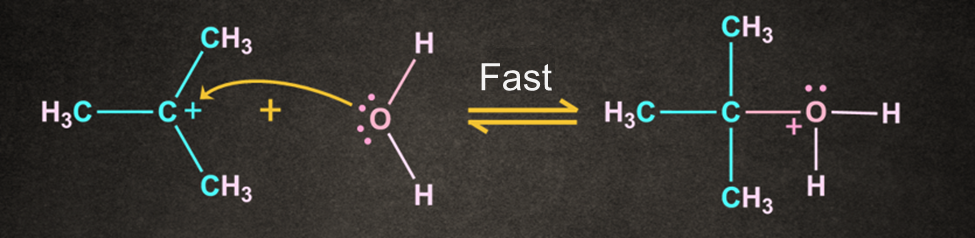
Step 2: Attack of the nucleophile
Second step of the SN1 reaction mechanism involves the nucleophilic attacks on the carbocation.
Water is used as a nucleophile which results in the formation of an oxonium ion intermediate after nucleophilic attack. In this step, the positive charge on the carbocation is transferred to the oxygen.
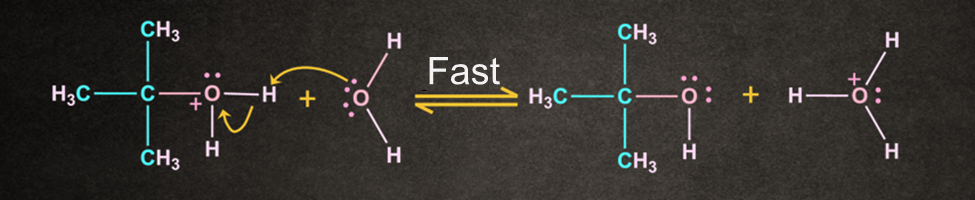
Since the solvent is neutral, deprotonation is required.
Water, which act as solvent, works as a base, by deprotonation of the oxonium ion and producing alcohol and a hydronium ion as the required product.
Energy Profile Diagram of SN1 Reaction:
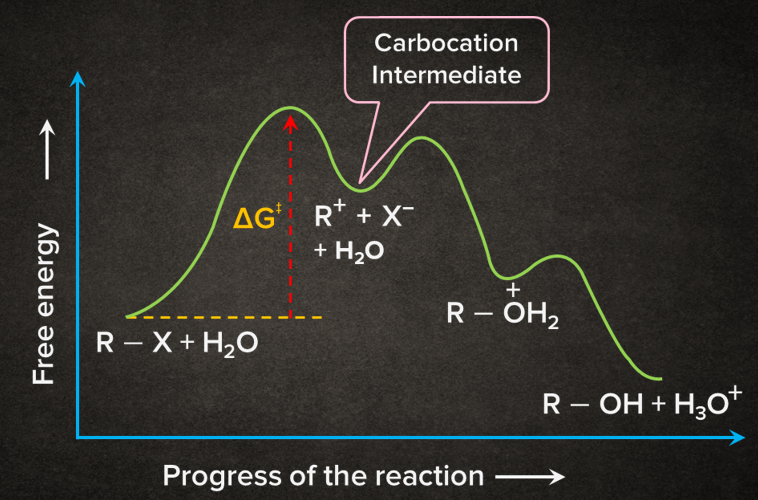
Rate of the SN1 Reaction:
In SN1 reaction, the first step being the slowest step is the rate-determining step.
As in the rate-determining step(RDS) only alkyl halide is involved, So it is a unimolecular reaction.
The rate of SN1 reaction is independent of the concentration of a nucleophile. A carbocation intermediate is formed, so there is a possibility of carbocation rearrangement in SN1 reaction. Due to the formation of a carbocation in the RDS the rate of SN1 reaction depends on the stability of carbocation.
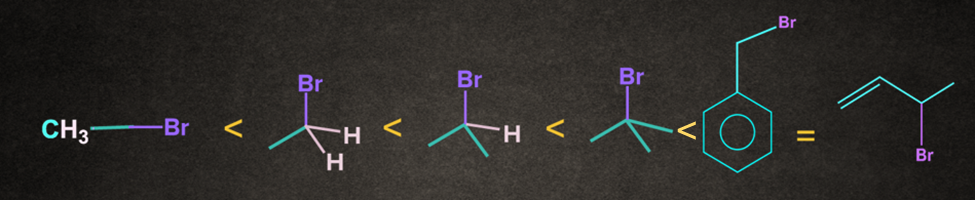
The nature of alkyl halide decides the reactivity of SN1 reaction. Increasing order of reactivity of alkyl halides is
|
Alkyl bromide |
Class of alkyl bromide |
Relative rate of alkyl bromide |
|
|
Tertiary |
12,00,000 |
|
|
Secondary |
11.6 |
|
|
Primary |
≈ 0 |

Also, rate of reaction depends on the R-X bond, for a given alkyl group(R). Weaker the R-X bond, more easily it can be cleaved for the further nucleophile to attack.
|
Bond |
C-X Bond enthalpy (kJ mol-1) |
Bond length(pm) |
|---|---|---|
|
CH3-I |
234 |
214 |
|
CH3-Br |
293 |
193 |
|
CH3-Cl |
351 |
178 |
|
CH3-F |
452 |
139 |
The order of reactivity of R-X bond towards SN1 nucleophilic substitution is given below: R-I > R-Br > R-Cl >R-F
Note: SN1 nucleophilic substitution is a regioselective reaction, such that that the most stable carbocation gives the major product.
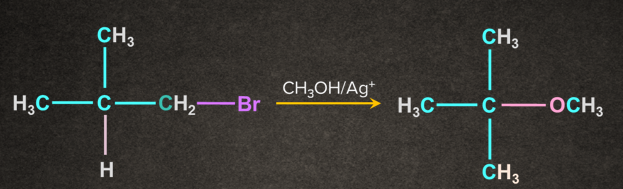
Examples of SN1 Reaction:
1. 
2. 
Note: If in a reaction, the solvent itself acts as a nucleophile and no other reagent is present, the reaction is known as solvolysis reaction.
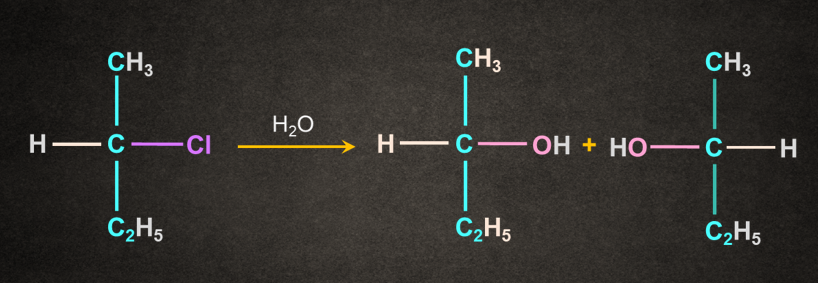
In the above case, water acts as both nucleophile and solvent.
Stereochemistry of SN1 Reaction
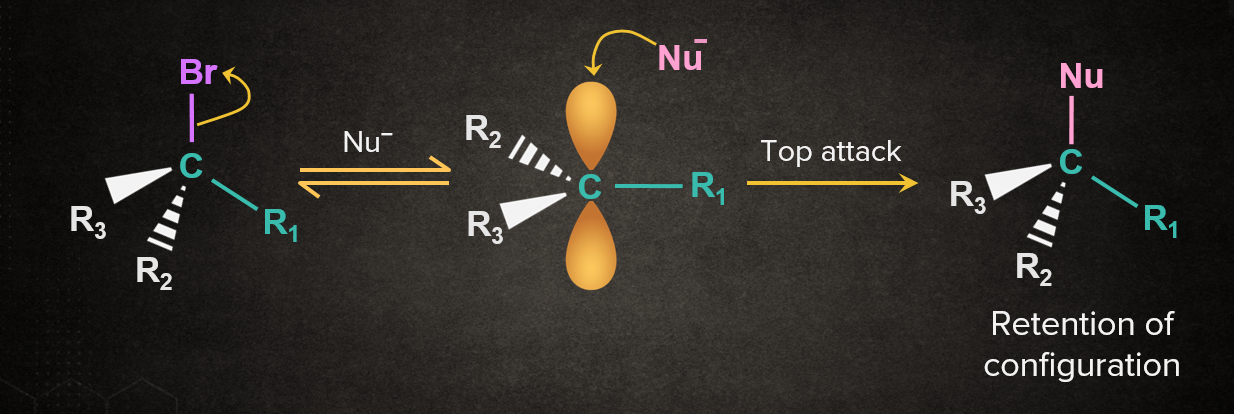
In the SN1 mechanism, a carbocation intermediate formed is sp2 hybridized and planar. Nucleophile can attack the carbocation from either face. If the reactant is chiral, the attack of the nucleophile from both the faces gives enantiomers as the product, known as racemisation. Reactions considering both the cases are discussed below.
Case 1: Nu- attacking from the same side as the leaving group.
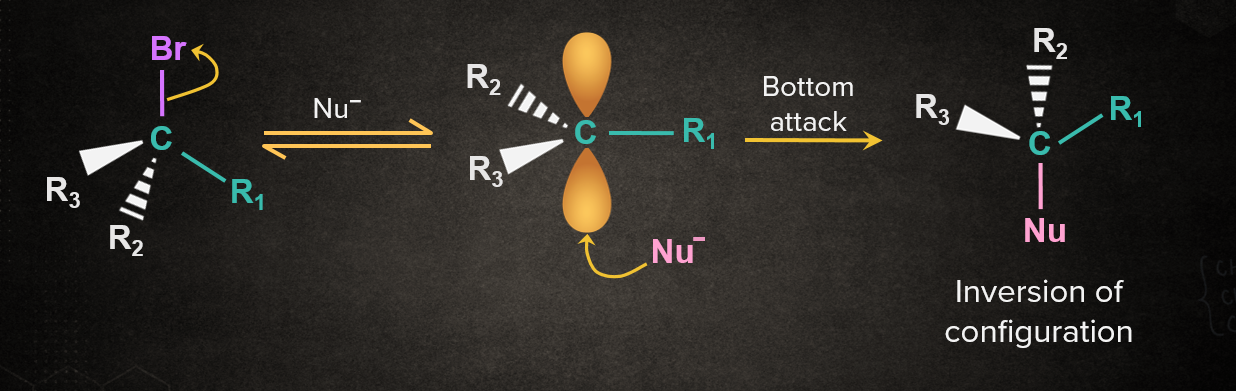
Case 2: Nu- attacking from the opposite side with respect to the leaving group.
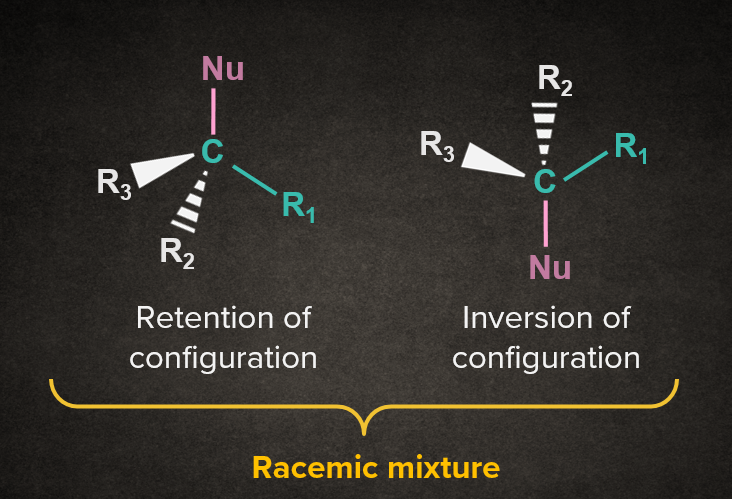
|
Note: Practically, we get a slightly higher proportion of inverted product in SN1 reaction. As we get one out of the two enantiomers as a major product, it is called partial racemization. |
Effect of Solvent:
The rate of the SN1 reaction can be increased by using a solvent that eases the formation of the carbocation intermediate.
Both polar and protic solvents are used for this type of reaction.
The polarity of the solvent helps in the stabilization of the positively charged carbocation intermediate formed in the reaction, whereas the polar bonds of the solvent help in the solvation and departure of the leaving group.
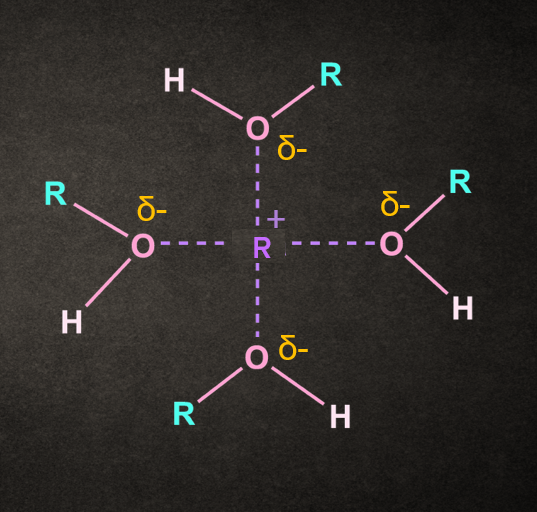
Alcohol and water are common examples of solvents used in SN1 reactions. These solvents also have nucleophilic properties and can act as a nucleophile.
SN2 Reaction
The SN2 reaction is referred to as a bimolecular nucleophilic substitution reaction because in the rate-determining step two reacting species are involved, i.e. the alkyl halide and the nucleophile. SN2 is a single-step reaction in which a bond is broken and a new bond is formed simultaneously.

Mechanism of SN2 Reaction:
The SN2 reaction mechanism occurs in a single step in which the substrate is being attacked by the nucleophile from the backside, which initiates the reaction. At an angle of 1800to the carbon-leaving group bond, the nucleophile approaches the given substrate through a transition state, the carbon-nucleophile bond is formed and the carbon-leaving group bond breaks at the same time and transition state is generated. Now, on the other side of the carbon-nucleophile bond, the leaving group is forced out of the transition state, generating the corresponding product. It's worth noting that the final product is formed by inverting the tetrahedral geometry at the central atom. Below is a diagram of the SN2 reaction mechanism for the nucleophilic substitution of chloromethane with hydroxide ion as the nucleophile.
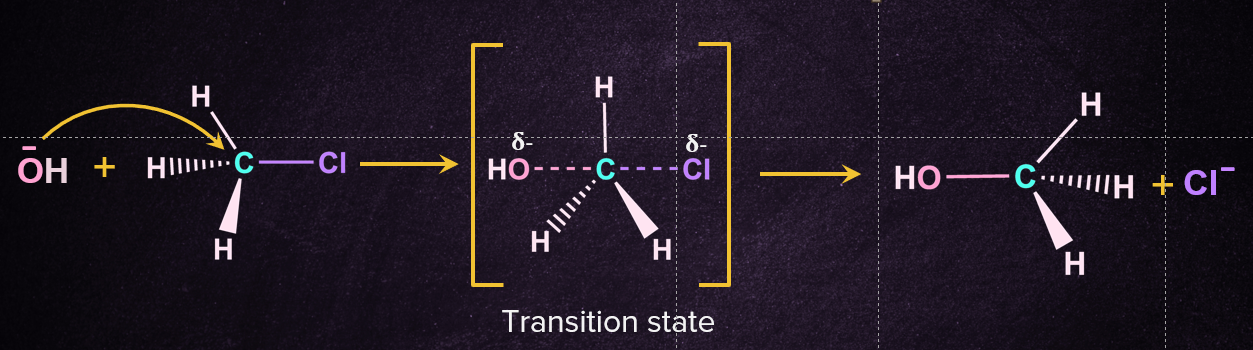
Energy Profile Diagram of SN2 Reaction:
In the transition state, a bond is partially formed between the nucleophile and the electrophilic carbon, and the bond between the carbon and the leaving group is partially broken as it involves simultaneous breaking and formation of bond.
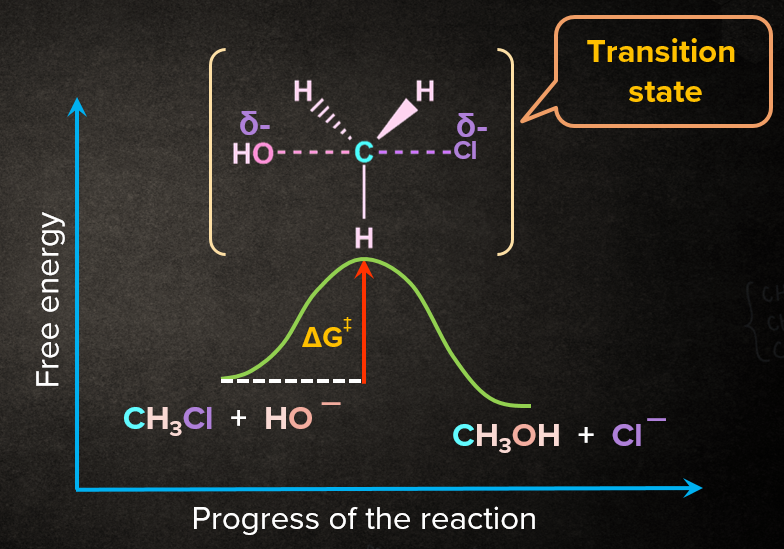
Rate of the SN2 Reaction:
The rate of SN2 reaction depends on the concentration of both alkyl halide and the nucleophile.
|
Rate ∝ [Alkyl halide][Nucleophile] |
It is a bimolecular, one-step process.
The nature of alkyl halide decides the reactivity of SN2 reaction. Very large and bulky groups can often hinder the formation of the required transition state. The crowding raises the energy of the transition state and slows down the rate of SN2 reaction.
Decreasing order of reactivity of alkyl halides is:
|
Substituent |
Compound |
Relative rate of SN2 |
|
Methyl |
30 |
|
|
10 |
1 |
|
|
20 |
0.02 |
|
|
30 |
∽0 |
Also, rate of reaction depends on R-X bond, for a given alkyl group. Weaker the R-X bond, more easily it can be cleaved for further nucleophiles to attack.
|
Bond |
C-X Bond enthalpy (kJ mol-1) |
Bond length(pm) |
|
CH3-I |
234 |
214 |
|
CH3-Br |
293 |
193 |
|
CH3-Cl |
351 |
178 |
|
CH3-F |
452 |
139 |
Order of reactivity of R-X bond towards SN2 nucleophilic substitution is: R-I > R-Br > R-Cl >R-F
Strong anionic nucleophiles speed up the rate of the SN2 reaction, And mostly anionic nucleophiles give SN2 reaction.
Example-
Stereochemistry of SN2 Reaction:
In an SN2 mechanism, the nucleophile attacks from the back side, that is from the side directly opposite to the leaving group. This causes an inversion of configuration at the chiral carbon atom. Also known as Walden inversion.
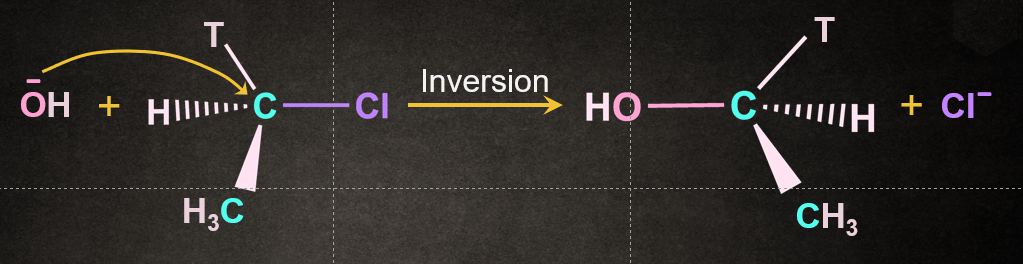
SN2 is a stereospecific reaction. Stereospecific reaction is a reaction in which a particular stereoisomeric form of the reactant reacts in such a way that it leads to a formation of specific stereoisomeric form of the product.
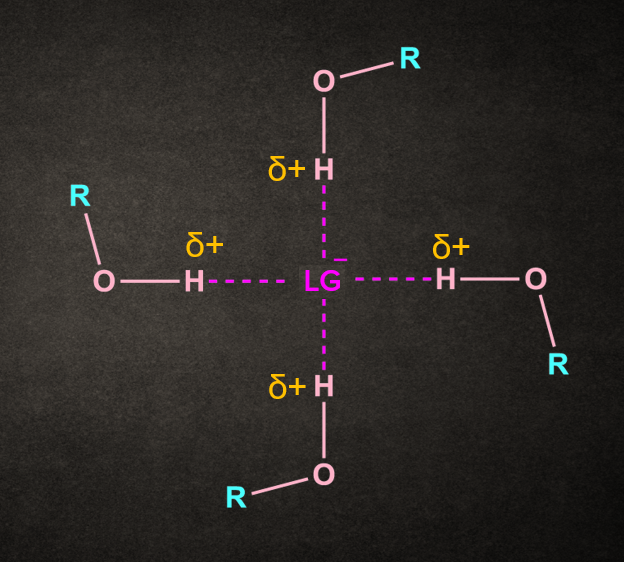
Effect of Solvent:
Polar aprotic solvents have a crowded positive center, so they do not solvate the anion appreciably. So they don’t help in the breaking of R-X bond or in the removal of leaving group. This rules out the possibility of SN1 reaction in this case. Hence, the rate of SN2 reactions increase when they are carried out in a polar aprotic solvent.

As we can see here, there is a repulsion between the methyls of DMSO and the nucleophile. As a result, the nucleophile remains unsolvated.
DMSO and acetone are commonly used solvents in SN2 reactions.
SNi Reaction
In SNi , S stands for substitution, N stands for nucleophilic, i stands for internal. In the SNi (internal nucleophilic substitution), a part of the leaving group must be able to attack the substrate detaching itself from the rest of the leaving group in the process. In the SNi reaction, the mechanism proceeds with the retention of configuration.
Example- Reaction of alcohol with SOCl2 Thionyl chloride (SOCl2) converts primary and secondary alcohols to alkyl chlorides with the retention of configuration.
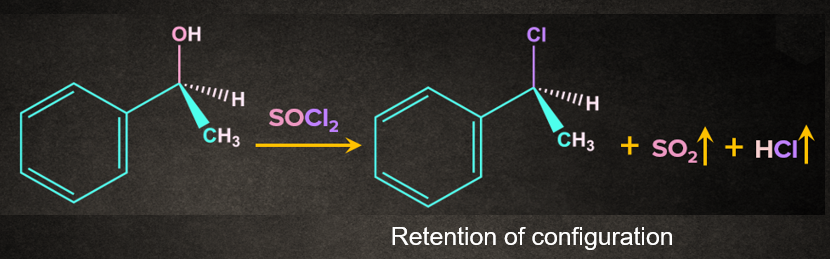
Mechanism of SNi Reaction:
Step 1: Reaction of alcohol with SOCl2
Alcohol reacts with thionyl chloride resulting in the formation of alkylchlorosulphite.
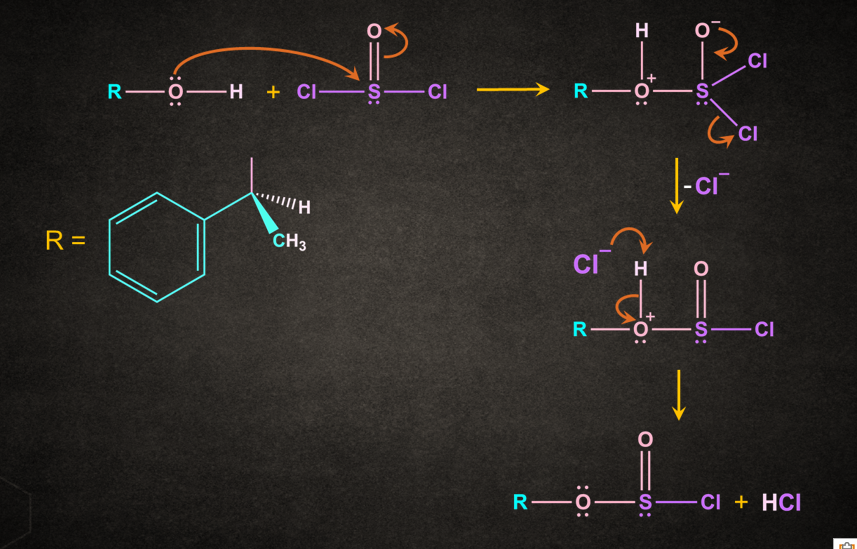
Step 2: Dissociation of alkylchlorosulphite
Second step is the same as the very first step of the SN1 mechanism i.e., dissociation into an intimate ion pair.

Step 3: Attack of the leaving group
Part of the leaving group attacks, necessarily from the front since it is unable to get to the rear side. It results in retention of configuration.
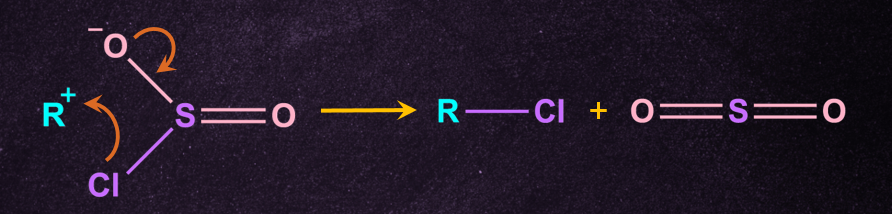
Note: If the same reaction occurs in the presence of pyridine, an inverted product is formed. Pyridine (C5H5N) is often included to promote the reaction.
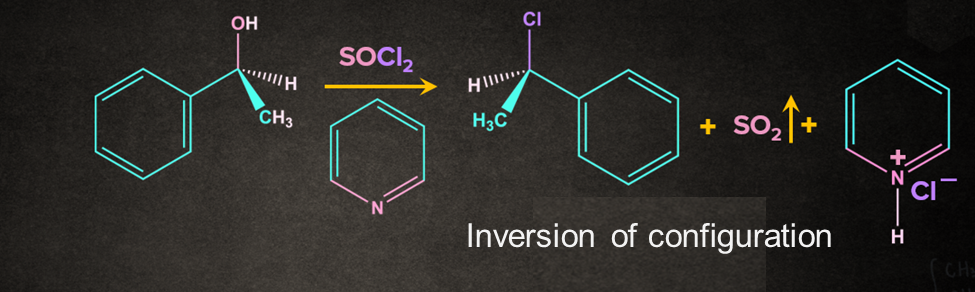
The mechanism involved in the above reaction is followed:
Step-1: Reaction of alcohol with SOCl2
Alcohol reacts with thionyl chloride resulting in the formation of an alkylchlorosulphite.

Step-2: Formation of pyridinium- alkylsulphite intermediate
The alkylchlorosulphite intermediate formed then reacts rapidly with another molecule of pyridine, to give a new intermediate called pyridinium alkyl sulphite.
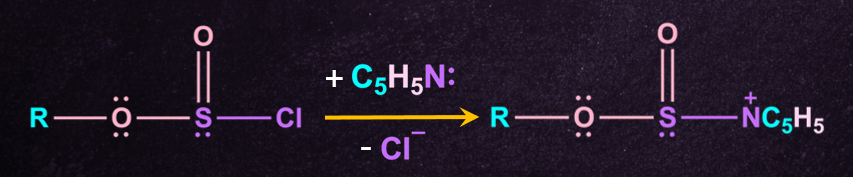
Step-3: Attack by chloride anion
A chloride anion then attacks the substrate carbon, displacing the sulphite leaving group.
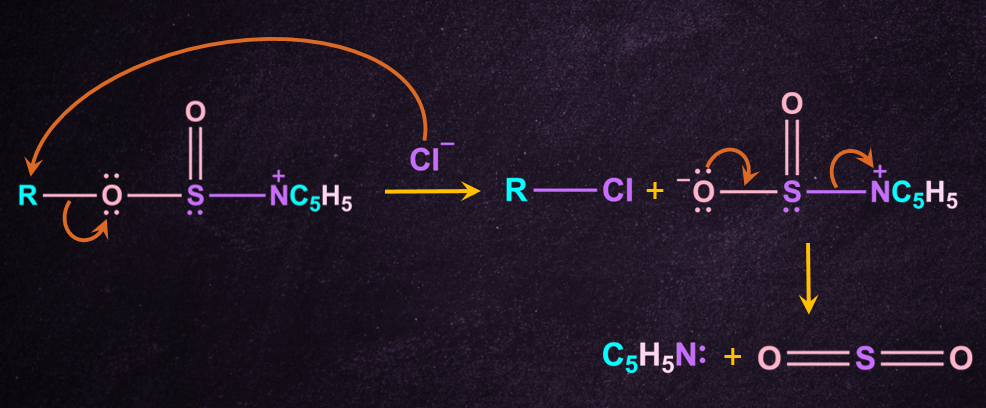
Inversion of the configuration takes place because free Cl- attack from the opposite side of the leaving group to give inverted R-Cl and - . - further cleaves to give pyridine(NC5H5 ) and SO2.
Example of SNi Reaction:
Reaction of Alcohol with PCl5

Reaction with PCl5 follows the same steps of mechanism as that of SOCl2 is followed.
SNNGP Reaction
In SNNGP, S stands for substitution, N stands for nucleophilic, NGP stands for neighboring group participation. It is also known as anchimeric assistance, which in Greek, it means “Adjacent part”.
Note: Rate of intramolecular reaction( i.e.when two reacting groups in the same molecule) is faster than intermolecular( i.e. when two reacting groups in separate individual molecules).
Characteristics of SNNGP Reaction:
1. The rate of reaction is greater than expected.
2. The configuration at a chiral carbon is retained such that the configuration at a chiral carbon is not inverted or racemised.
Conditions for SNNGP Reaction:
Nucleophile should be present within the molecule/ internally. Generally, the nucleophile and leaving group should be present anti to each other at 1,2 position. The concentration of nucleophile (external) should be less.
Mechanism of SNNGP Reaction:
Reactions, in which there is usually a group with an unshared pair of electrons, present at β-position to the leaving group (or sometimes farther away), the mechanism operating in such cases is called the neighbouring-group participation mechanism. It consists essentially of two SN2 substitution reactions. Each causes inversion so the net result is the retention of configuration.
Step 1: Intramolecular nucleophilic substitution reaction
The neighbouring group acts as a nucleophile, such that it pushes out the leaving group but still remains attached to the molecule.

Step 2: External nucleophilic substitution reaction
The external nucleophile then displaces the neighbouring group by attacking from the backside.
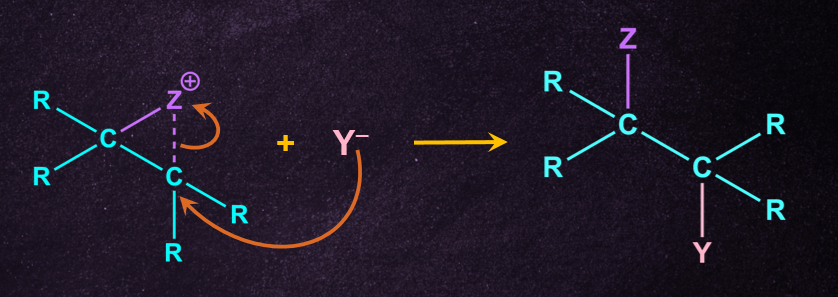
Below given is the table of groups behaving as Neighboring Groups(internal nucleophile).
|
Sulphur |
-SH, -SR, -Sˉ |
|
Carboxylic acid derivatives |
-COOˉ, -COOR, -OCOR |
|
Oxygen |
-OR, -OH, -Oˉ |
|
Nitrogen |
-NH2, -NHR, -NR2 |
|
Halides |
-I, -Br, -Cl |
Examples of SNNGP Reaction:
Neighbouring Group Participation by:
1. Involving species having lone pair
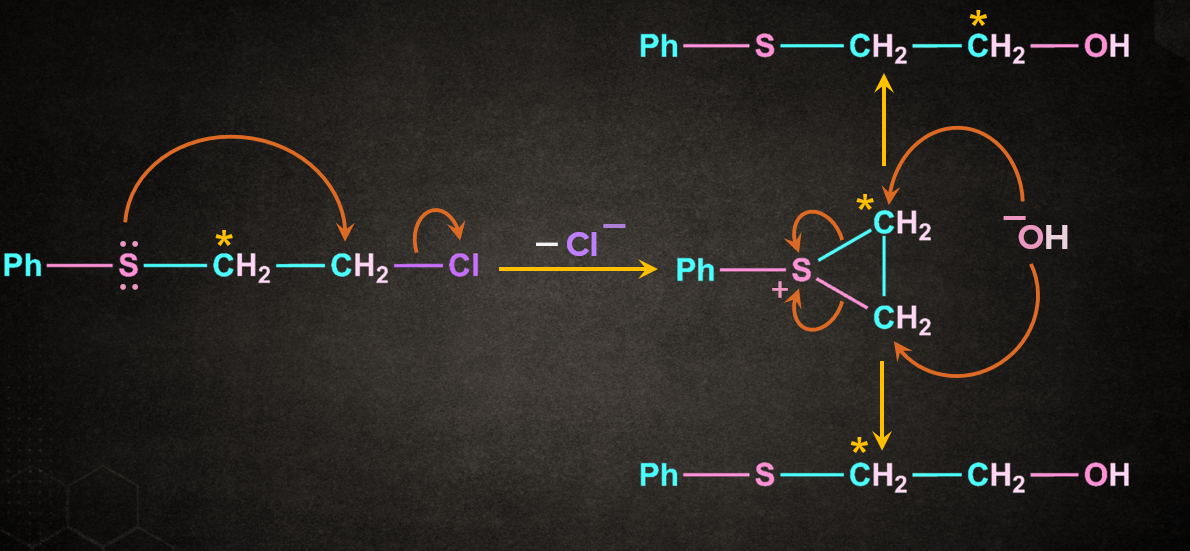
2. Aromatic rings as neighbouring groups

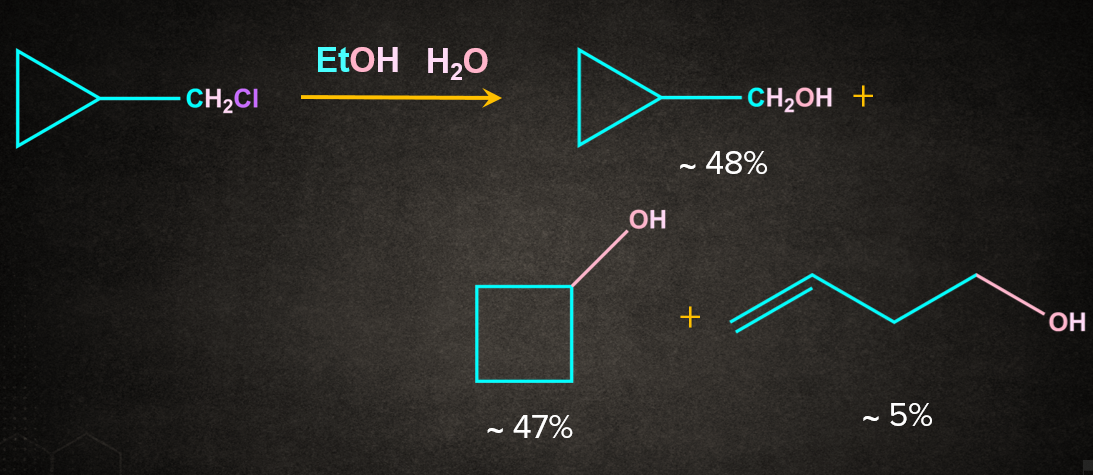
3. Cyclopropyl methyl system
Cyclopropyl methyl substrates solvolyze with abnormally high rates. The products often include not only cyclopropyl-methyl, but also cyclobutyl and homoallylic compounds.
Practice problems
Q1. Write the product of the following reaction with proper stereochemistry.
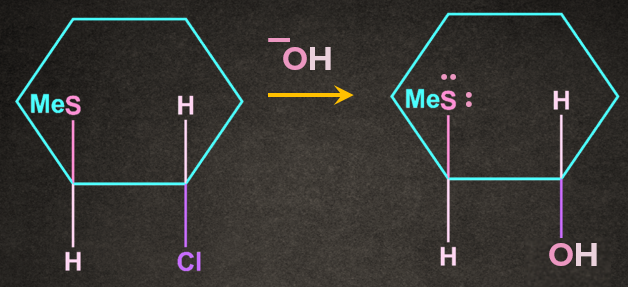
A). 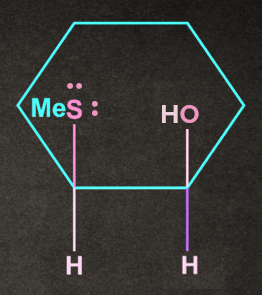
B). 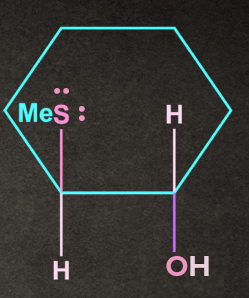
C). Both of the above
D). None of the above
Answer: B
Solution: 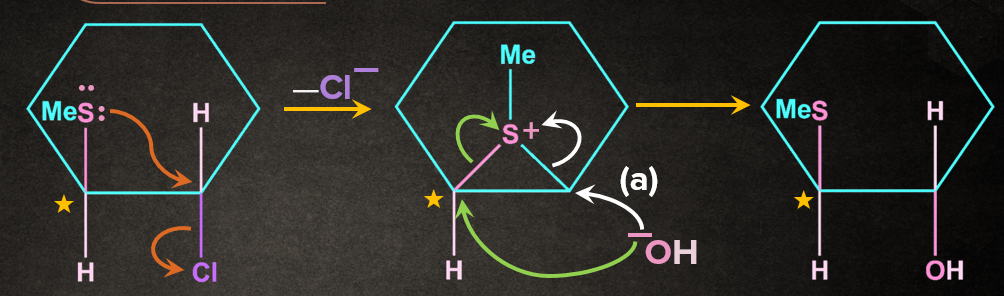
Q2. Arrange the following alkyl chlorides in the order of decreasing reactivity in an SN2 reaction?

A). P > Q > R > S
B). S > R > Q > P
C). S > Q > R > P
D). R > Q > S > P
Answer: A
Solution: Nature of alkyl halide decides the reactivity of SN2 reaction. Bulky groups often hinder the formation of the required transition state. The crowding raises the energy of the transition state and slows down the rate of reaction.
The decreasing order of reactivity of alkyl halides is:
Hence, the decreasing order of alkyl chloride is: P > Q > R > S
Q3. Which of the following gives the fastest SN1 reaction?
A). 
B). 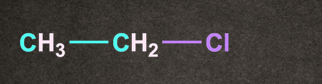
C). 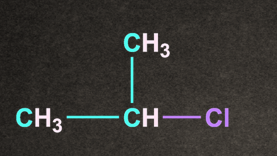
D). 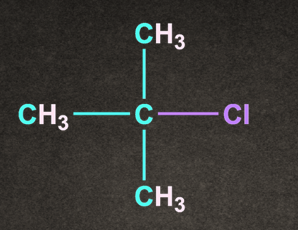
Answer: D
Solution: Degree of alkyl halide decides the reactivity of SN1 reaction.
More substituted the alkyl halide, it will rise to more stable carbocation.
Therefore option D, (CH3)3C-Cl generates more stable carbocation and will give the fastest SN1 reaction.
Q4. Reaction of alcohol with SOCl2 in the presence of pyridine occurs via _______ of configuration.
A). Inversion
B). Retention
C). Both of the above
D). None of the above
Answer: A
Solution: Reaction of alcohol with SOCl2 in presence of pyridine, gives the inverted product. Pyridine
(C5H5N) is often included to promote the reaction.
Frequently Asked Questions-FAQs
Q1. What is a substitution reaction? In a substitution reaction, one atom or a group of atoms is replaced by another atom or group of atoms, resulting in the formation of a completely new chemical compound.
Example:
In the above reaction, Y is substituted by Z. Such kinds of reactions in organic chemistry are said to be substitution reactions.
Q2. What is a leaving group?
Answer: The part of the reactant molecule which gets cleaved is called a leaving group. Generally poor bases are good leaving group.
1. Nucleofuge: Leaving group that carries away an electron pair or negative charge. For nucleofuge, weaker bases are good leaving groups.
2. Electrofuge: Leaving group that comes away without an electron pair or having positive charge.
Note: Generally, the leaving group carries away an electron pair.
Example:
Q3. What is a rate-determining step (RDS)?
Answer: If one step in a multistep reaction is intrinsically slower than all the others, then the rate of the overall reaction will be essentially the same as the rate of this slowest step. This slowest step is known as the rate-limiting step or the rate-determining step.
Q4. What is a regioselective reaction? A reaction which has more than one region to attack, Answer: which results in the formation of two or more products is called a regioselective reaction. Products obtained are constitutional (structural) isomers of each other. But in this mixture of products, one is the major product.