-
Call Now
1800-102-2727
Reduction of Nitro Compounds: Amine, Nitro compounds, Reduction- by Catalytic Hydrogenation, with Acid & Metal, by Electrolytic Reduction, Practice Problems, FAQs
You must have heard of dynamites.
It is a type of explosive which is used in mining areas to break big rocks into smaller pieces.
Which chemical do you think has been used to create such an explosion?
Nitroglycerin, commonly known as glyceryl trinitrate, is a potentially explosive and a crucial component of the majority of dynamite types.
Well, are all nitro compounds used in making explosives?
No, it has a wide range of applications in the pharmaceutical industry too. Nitro compounds can also be used to produce amines, which is a very important compound in organic chemistry.
Let’s understand some common methods of preparation of amines from the reduction of nitro compounds.
Table of Content
- Amine
- Classification of Amines
- Nomenclature of Amines
- Reduction
- Nitro Compounds
- Catalytic Hydrogenation of Nitro Compounds
- Reduction with Acid and Metal of Nitro Compounds
- Electrolytic Reduction of Nitro Compounds
- Practice Problems
- Frequently Asked Questions
Amine
Chemically speaking, the amine is a derivative of ammonia (NH3). In other terms, amines are derived from ammonia. A nitrogen atom with a lone pair is present in amines, which are organic nitrogen molecules. Aryl or alkyl groups often replace the hydrogen atoms of ammonia in amines.
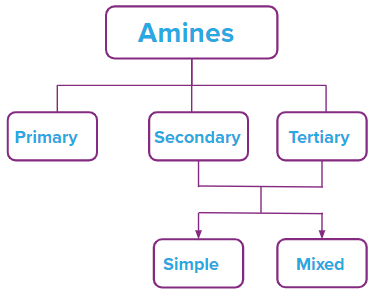
Classification of Amines
Amines can be classified according to the nature and number of alkyl or aryl groups attached to the Nitrogen.
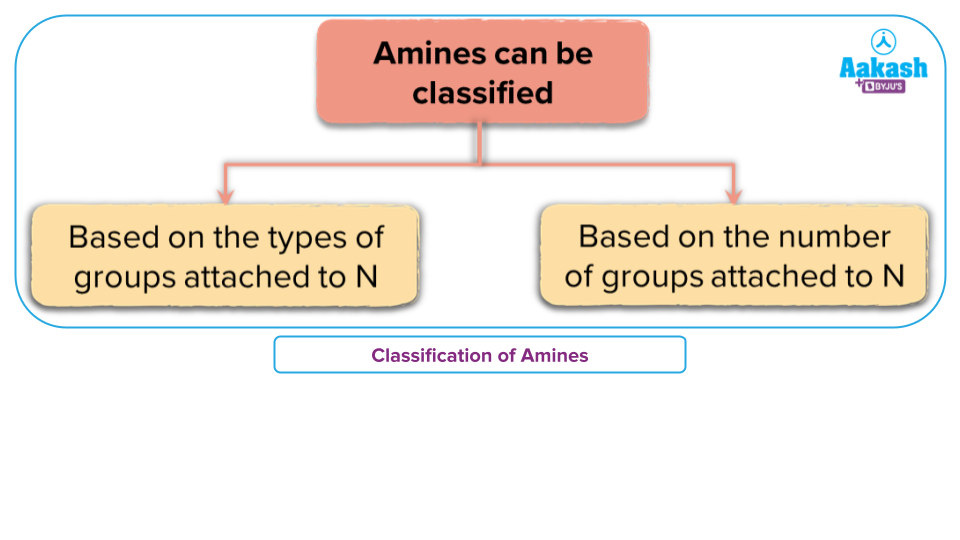
- Classification of the type of the hydrocarbon groups attached to N:
Amines are classified into three categories based on the types of groups attached to N
- Alkylamines
- Arylamines
- Heterocyclic amines
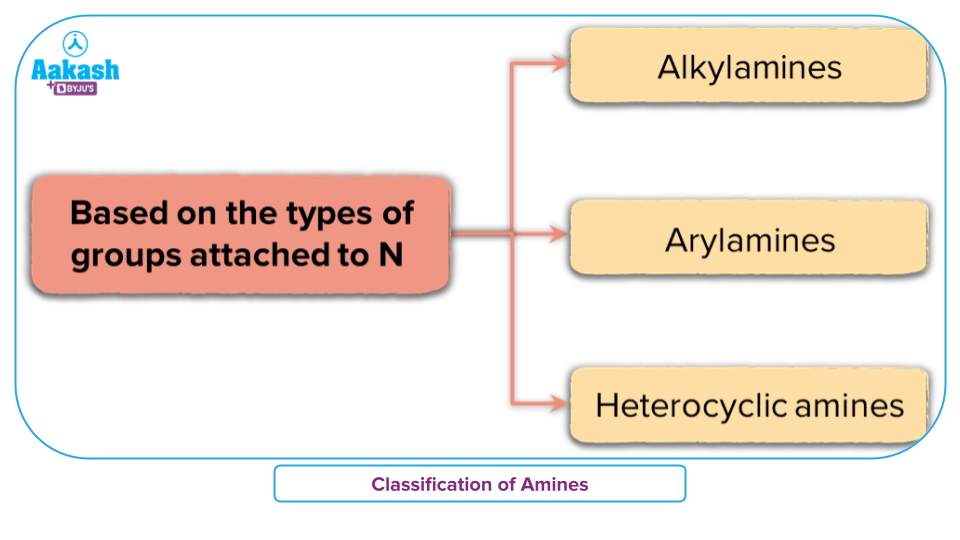
Alkyl Amines
An alkyl amine is a substance in which the nitrogen atom of an amine is joined to an aliphatic alkyl group. Alkyl amines are created when one of the ammonia's three hydrogen atoms is changed for an alkyl.
For example Methylamine and Ethylmethylamine
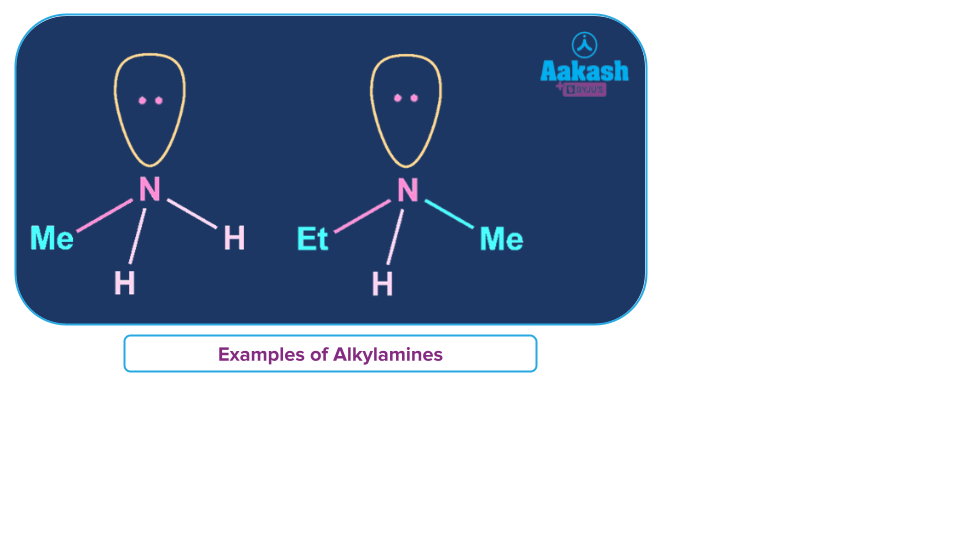
Aryl Amines
Aryl amines are a category of amines in which aromatic groups are used in place of one or more of the hydrogen atoms in ammonia.
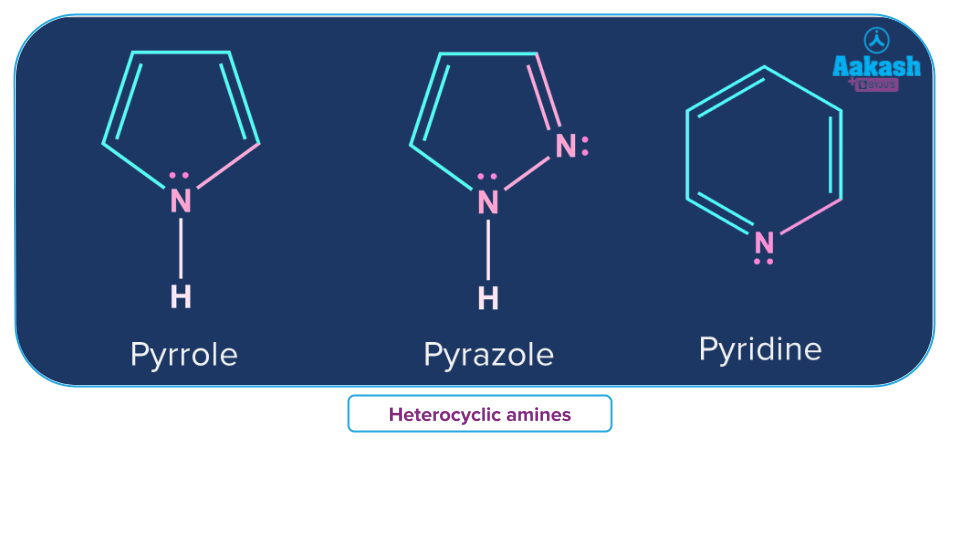
Heterocyclic Amines
Nitrogen of amine will be a part of the ring structure in heterocyclic amines.
- Classification of the number of hydrocarbon groups attached to N:
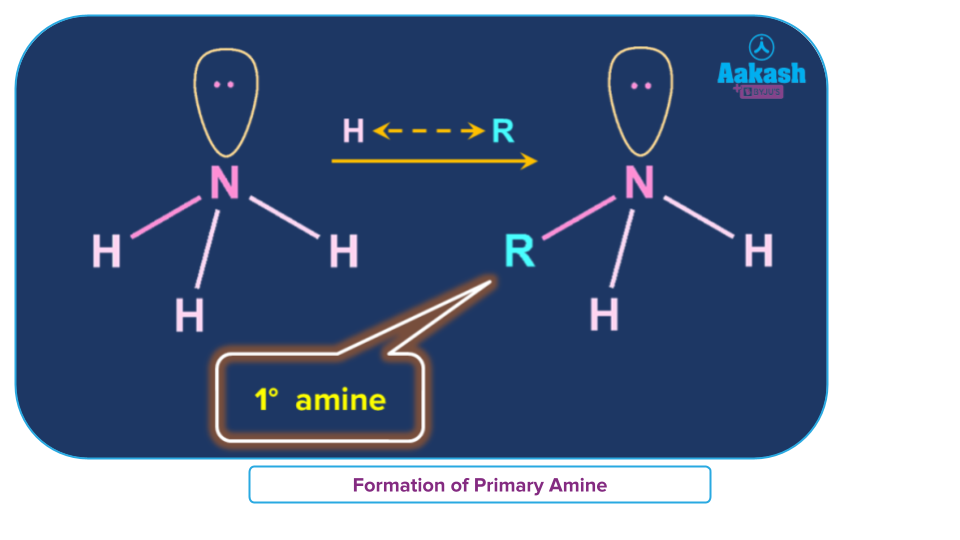
Amines are classified into three groups: primary (10), secondary (20), and tertiary depending on how many hydrogen atoms in ammonia are changed for an alkyl or aryl group (30). When one hydrogen atom is replaced, amines with the formula R-NH2 or primary (10) amines are created. Alkyl/aryl groups can replace two of the three hydrogen atoms to form secondary (20) amines. If an alkyl or aryl group is substituted for all three hydrogen atoms, tertiary (30) amines are created.
Primary Amines
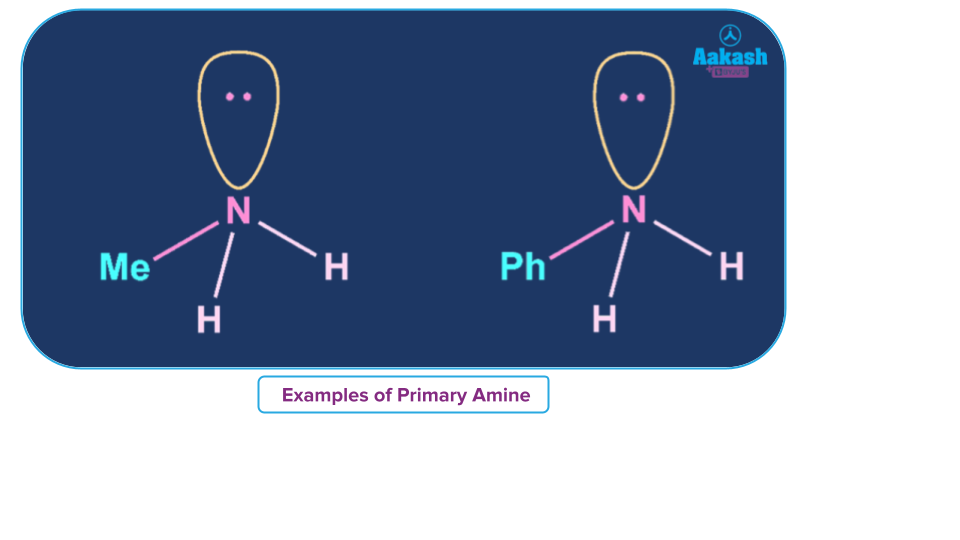
When an alkyl or aromatic group replaces one of the hydrogen atoms in ammonia, a primary amine is created. Amino acids and methylamine are examples of primary alkyl amines, whereas aniline is an example of a primary aromatic amine.
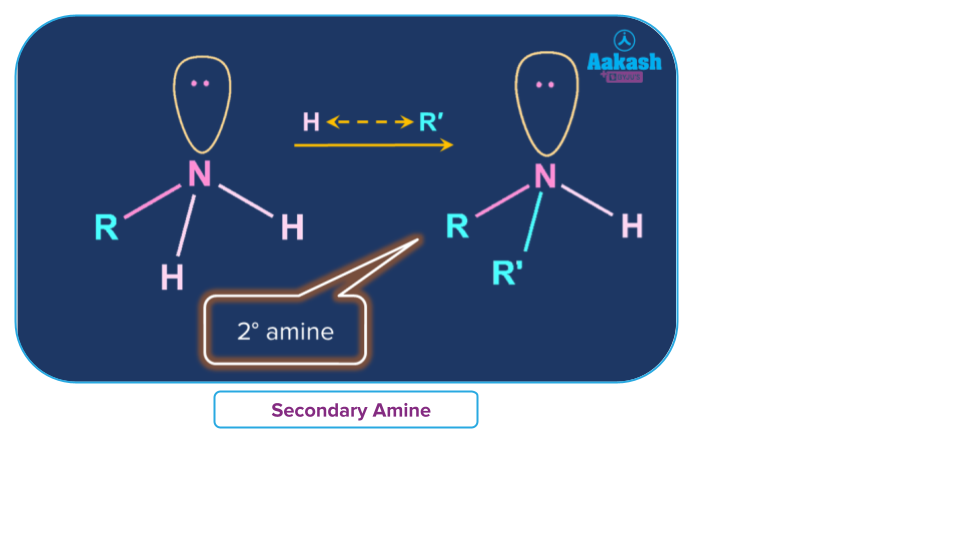
Secondary Amine
An amine in which the amino group has a direct link to two carbons of any hybridization, which takes place when an organic molecule with two amine groups. Secondary amines are also called as enamine.
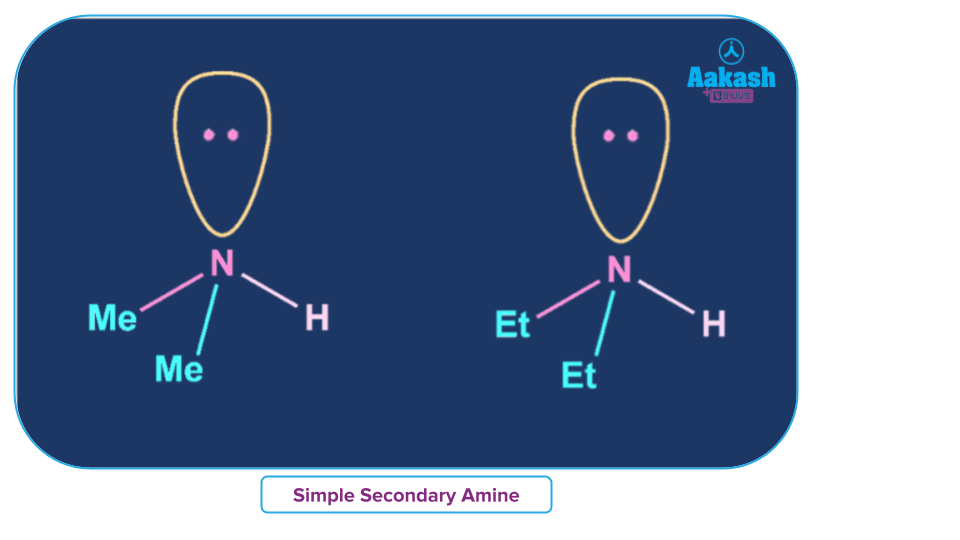
Simple Secondary Amine
An amine where all alkyl or aryl groups attached to the nitrogen atom are the same then it is called a simple secondary amine.
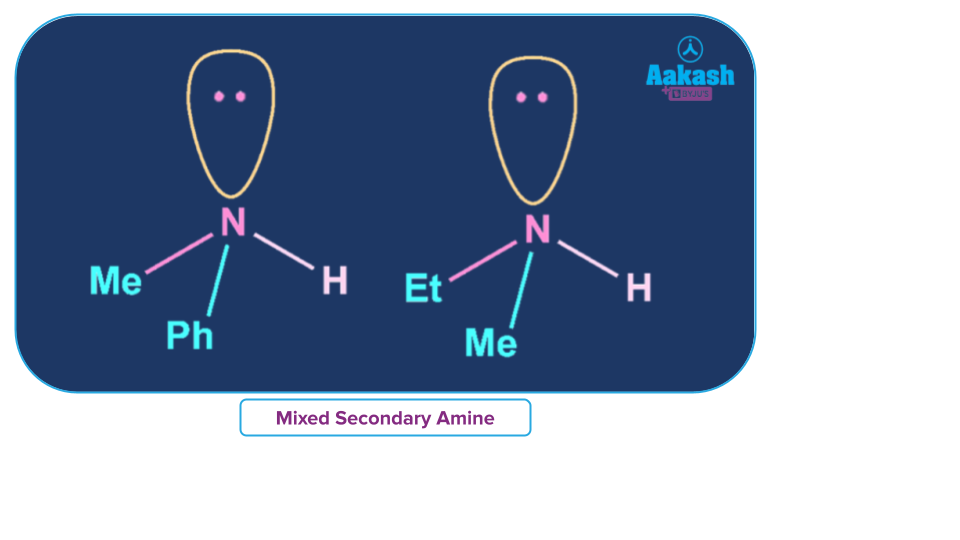
Mixed Secondary Amine
An amine where all alkyl or aryl groups attached to the nitrogen atom are not the same then it is called a simple secondary amine.
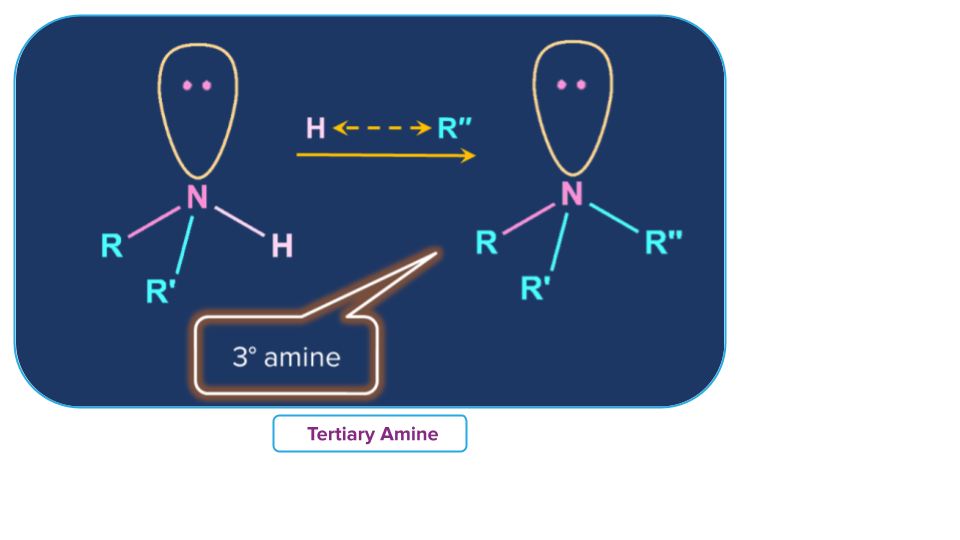
Tertiary Amine
An amine in which three carbons of any hybridization—but not carbonyl group carbons—directly link to the nitrogen atom. tertiary amine structure in general. C is equal to any carbon group besides carbonyl.
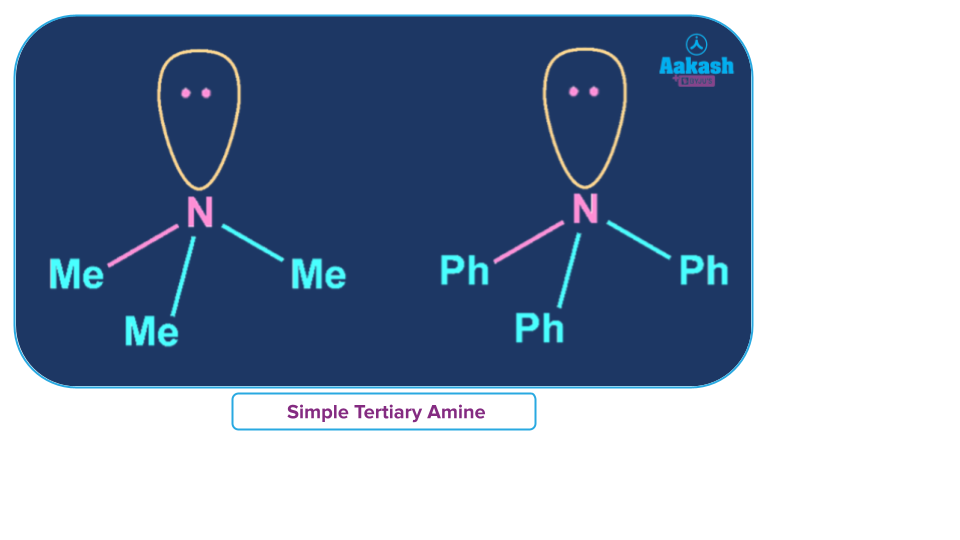
Simple Tertiary Amine
An amine Where all alkyl/aryl groups attached to N are the same then it is called a simple tertiary amine.
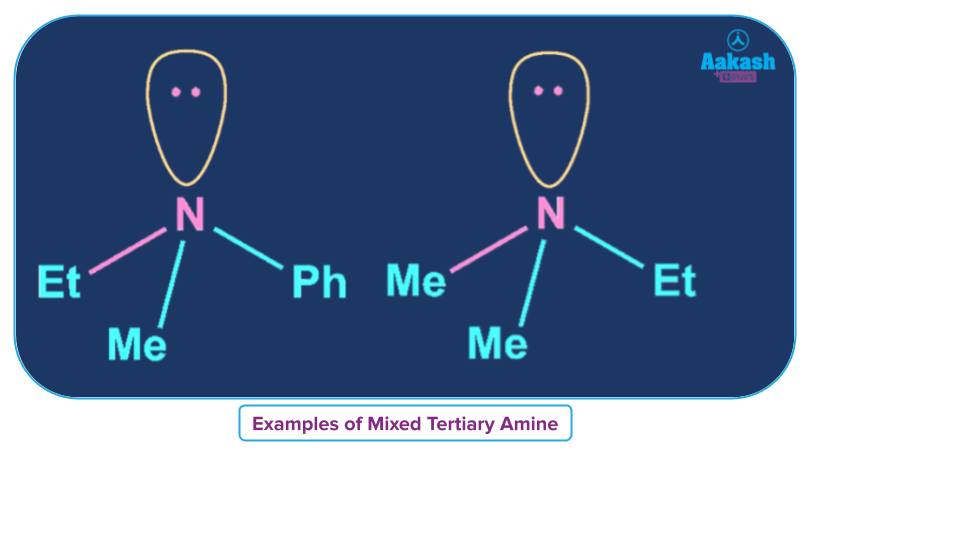
Mixed Tertiary Amine
An amine, where all alkyl/aryl groups attached to N are not the same then it is called a simple tertiary amine.
Nomenclature of Amines
The names of compounds that are widely accepted in organic chemistry are provided in accordance with the guidelines specified by IUPAC for the naming of organic compounds. Alkylamine is the form that aliphatic amine names adopt since the alkyl group is inserted before the amine during the naming procedure. For instance, the fact that CH3NH2 combines an amine with an alkyl group gives it the name "methylamine." The names of the alkyl groups are preceded by prefixes like di and tri when there are two or more comparable groups. If there are numerous amino groups present, the parent chain and the positions of the amino groups in the amine are identified by counting the carbon atoms.
Reduction
Reduction is a method used in chemistry to transfer electrons from one chemical to another. These electron-transfer procedures are referred to as redox reactions, also called oxidation-reduction reactions.
These reactions are accompanied by changes in energy in the form of heat, light, electricity, etc. Another step in the oxidation and reduction reactions is the addition of oxygen or hydrogen to different molecules.
- According to traditional or earlier conceptions, the reduction is a process that involves the addition of hydrogen or any other electropositive element or the removal of oxygen or any other electronegative element.
- According to the electronic notion, reduction is the process by which an atom or an ion gains one or more electrons.
Nitro Compounds
Nitro compounds are organic substances that include one or more nitro functional (-NO2) groups. Simply explained, a nitro compound is any class of chemical compounds in which the nitro group (-O-N=O) participates in the construction of molecules. Examples include organic compounds that feature a covalent connection between a carbon atom and a nitrogen atom from the nitro group.
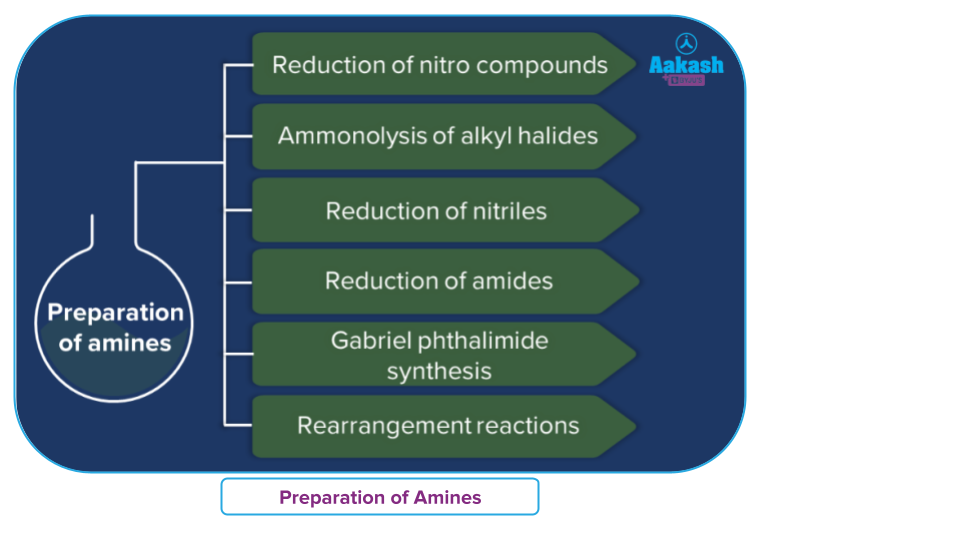
Preparation of amine
Amine is generally prepared by the following methods:
- Reduction of nitro compounds
- Ammonolyisi of alkyl halides
- Reduction of nitriles
- Reduction of amide
- Gabriel phthalimide synthesis
- rearrangement reaction
Reduction of Nitro Compounds
By reducing the appropriate nitro compounds, one can quickly generate primary amines that are both aliphatic and aromatic. There are numerous methods for achieving this reduction, including
- Catalytic hydrogenation
- Treatment with acid in the presence of a metal
- Electrolytic reduction
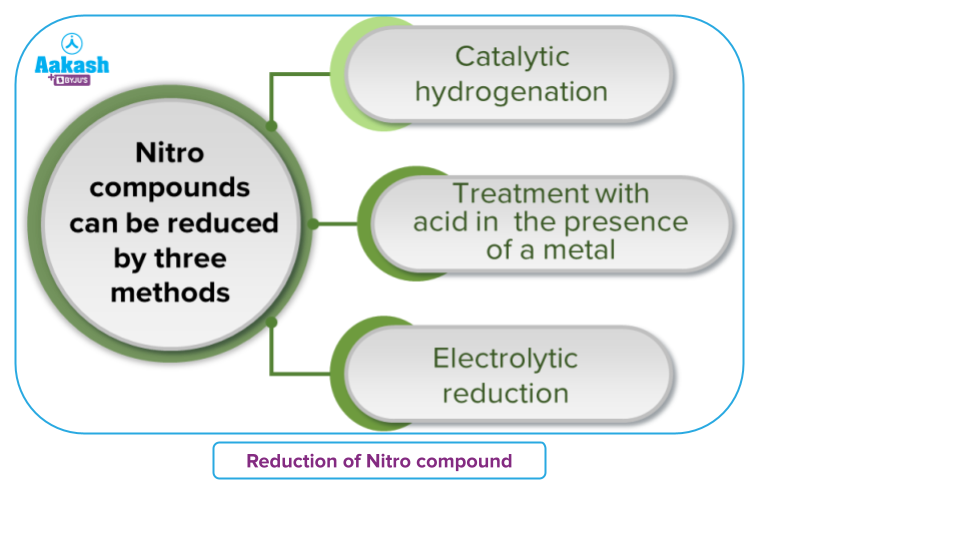
Catalytic Hydrogenation of Nitro Compounds
Nitro alkanes on passing hydrogen in the presence of Raney Ni, finely divided Pt or Pd or Pd/C, as catalyst at room temperature gives primary amines.
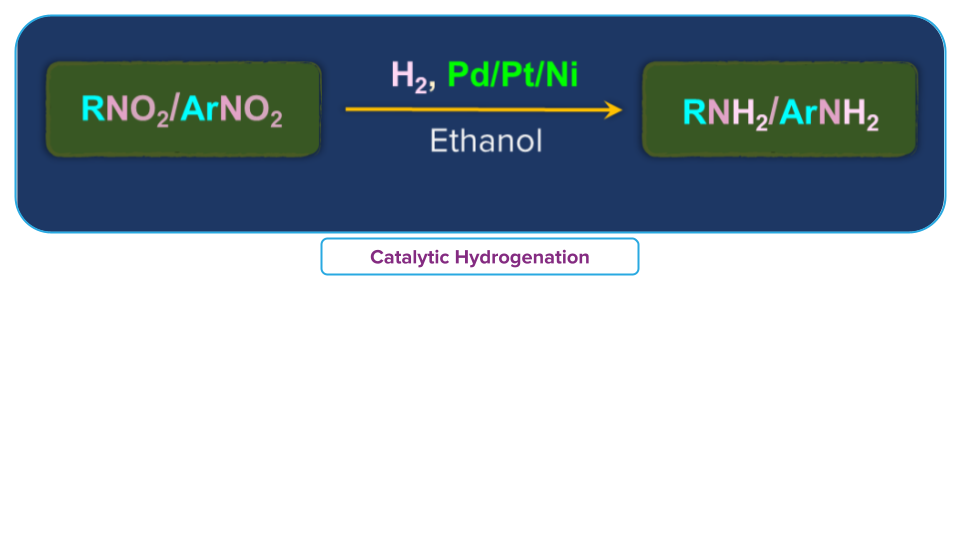
For example:
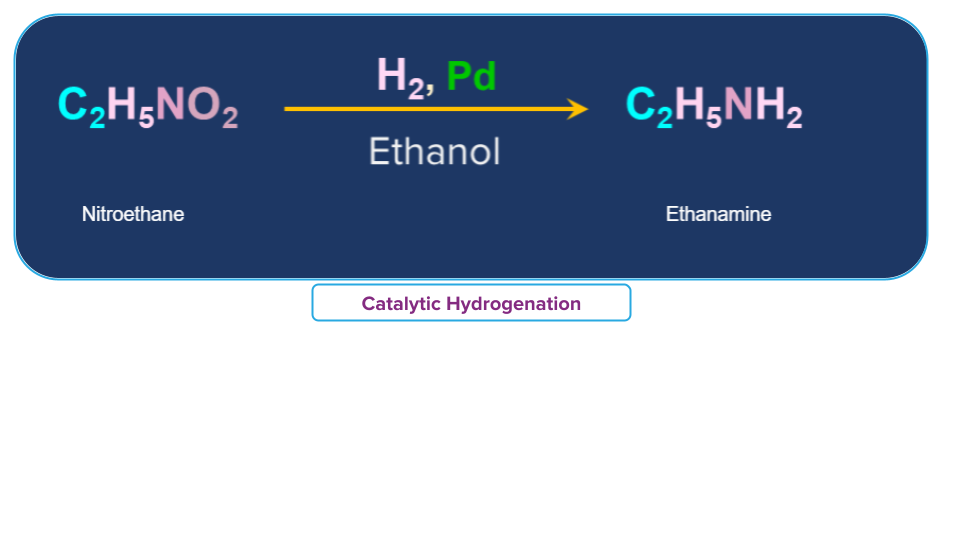
Nitro ethane (C2H5NO2) in presence of finely divided Pt or Pd or Pd/C gives ethanamine (C2H5NH2).
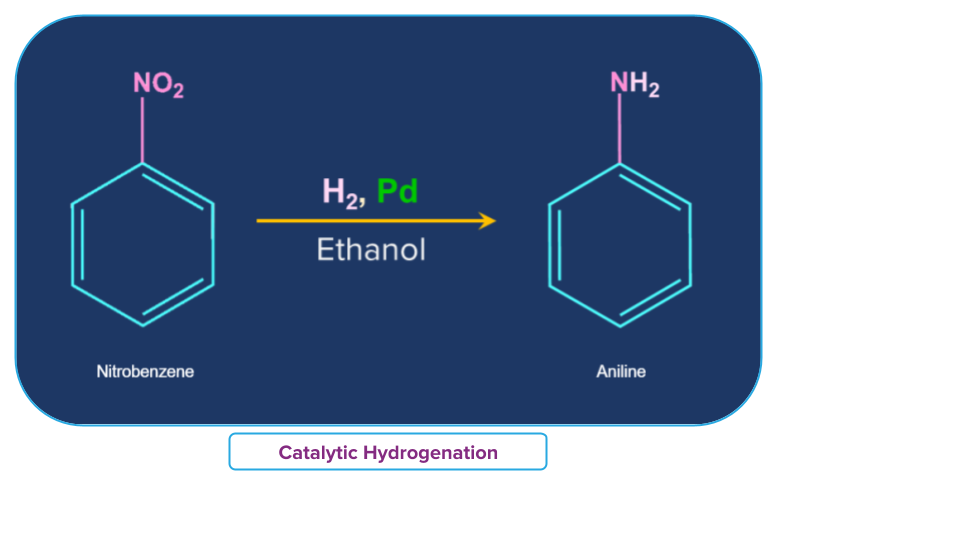
Another example can be considered where nitrobenzene is been converted to aniline by passing H2 gas in presence of finely divided Pt or Pd or Pd/C.
Treatment with Acid in presence of Metal of Nitro Compounds
In the presence of strong hydrochloric acid and an active metal like Fe, Sn, or Zn, nitroalkane is transformed into a primary amine.
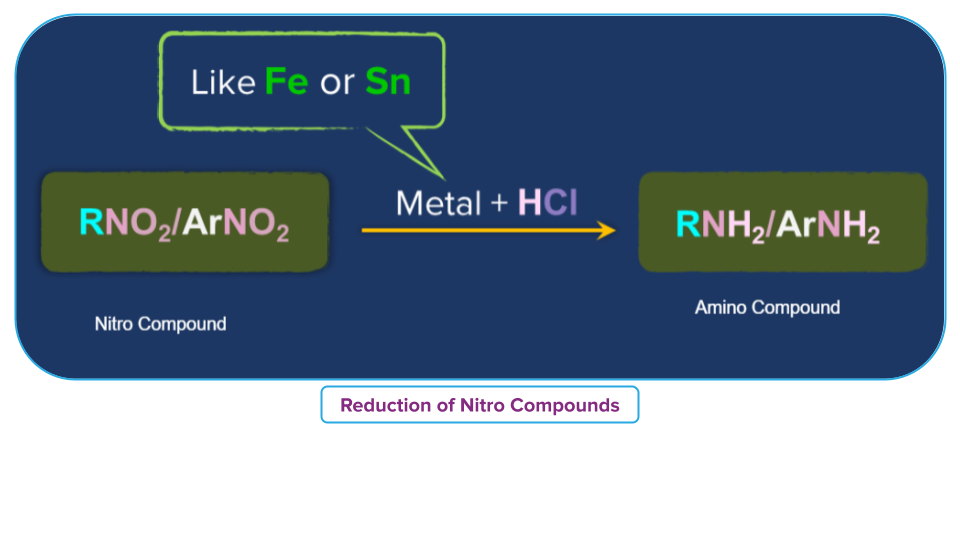
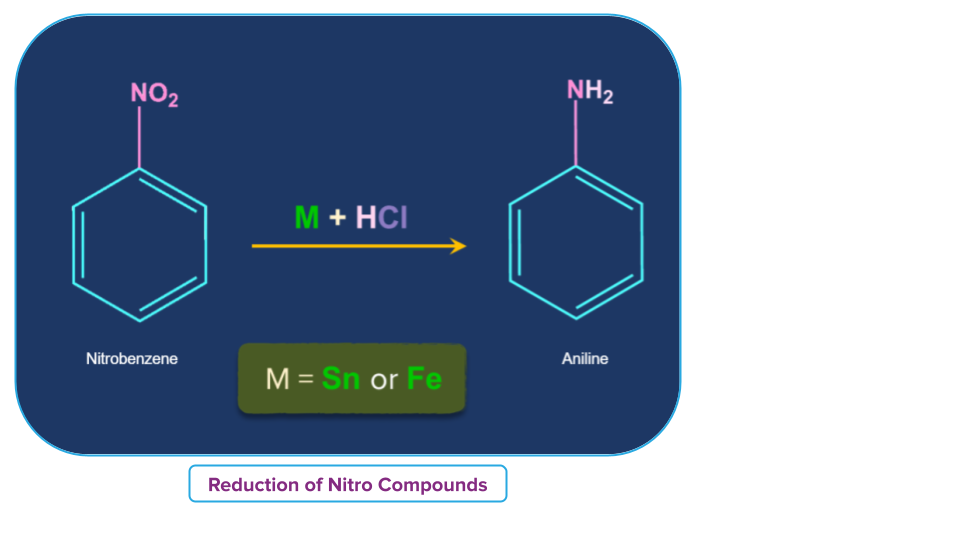
For example,
- Nitrobenzene is reduced to aniline when combined with strong HCl and metal, such as tin or iron.
The reduction of aromatic nitro compounds has also been accomplished using a combination of SnCl2 and conc. HCl. Since FeCl2generated during the reaction is hydrolysed to liberate hydrochloric acid, this reduction method is favoured to Sn/HCl. As a result, only a little quantity of hydrochloric acid is needed to start the reaction.
Since aromatic primary amines cannot be made from the equivalent aryl halides on treatment with ammonia, one of the most practical ways to make them is through the reduction of nitro compounds to primary amines. The nitration of arenes is a simple method for producing the necessary nitro compounds.
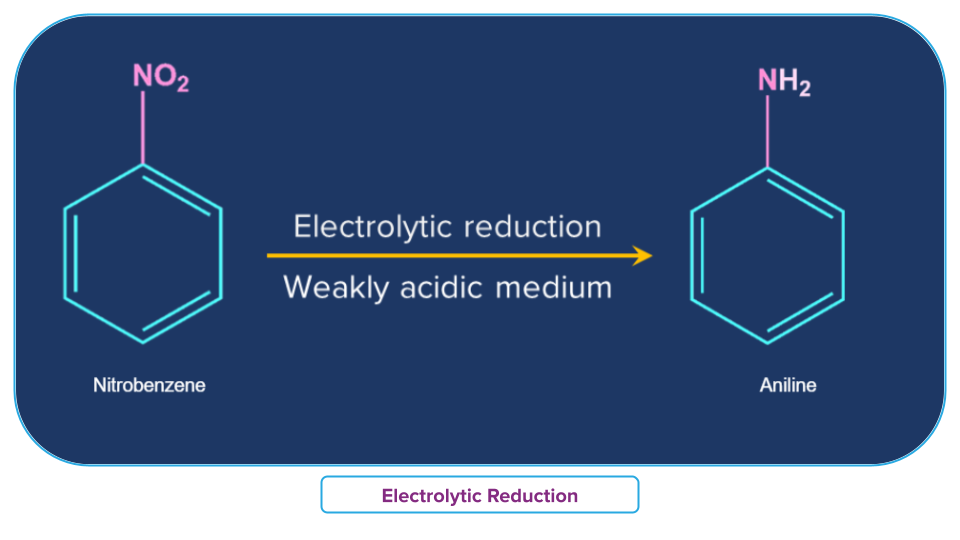
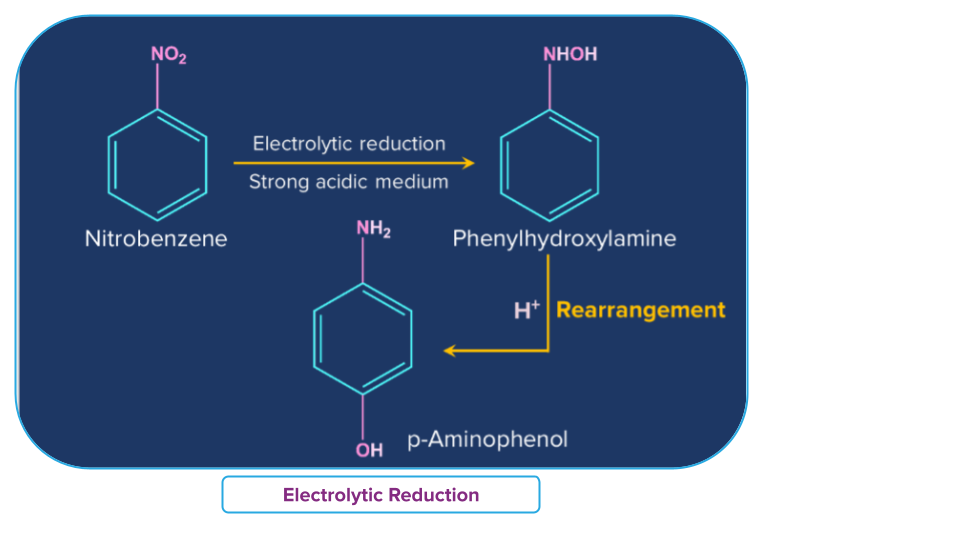
Electrolytic Reduction of Nitro Compounds
In a weakly acidic solution, electrolytic reduction of nitrobenzene produces aniline, whereas, in a strongly acidic condition, it produces p-aminophenol. Azoxybenzene and azobenzene are two examples of the many mono- and di-nuclear reduction products that can be produced in an alkaline media.
Practice Problems
Q1. Pyrazole is an example of
- Alkyl amine
- Heterocyclic amine
- Enamine
- Aryl amine
Answer: (B)
Amines are classified into three categories based on the types of groups attached to N
- Alkylamines
- Arylamines
- Heterocyclic amines
A chemical that develops during the high-temperature cooking of meat, poultry, or fish, such as when frying, broiling, or grilling. Carcinogens include heterocyclic amines (substances that may cause cancer). also known as heterocyclic amine. Examples of this category of amines are Pyrazole, Pyridine and Pyrrole.
Q2. Which of the following method is suitable for the preparation of amines from nitro compounds?
- Catalytic hydrogenation
- Electrolytic reduction
- Treatment with acid in presence of metal
- All of the above
Answer: (D)
Solution: Both aliphatic and aromatic primary amines can be easily prepared by the reduction of the corresponding nitro compounds. This reduction can be achieved in a number of ways such as
- Catalytic hydrogenation
- Electrolytic reduction
- Treatment with acid in presence of metal
Q3. What is the proper name for the substance that contains two amino groups positioned in opposition around a benzene ring?
- benzen1,4-diamine
- 3-aminobenzamine
- Both A & B
- None of the above
Answer: (A)
Solution: Given the equal significance, both amino groups (located in para position to one another) are called by prefixing the primary molecule with "di." The positions of the amino groups in the benzene ring are represented by numbers 1 and 4 on the prefix. Hence, the nomenclature would be benzene1,4-diamine.
Q4. Which of the following reducing agent can be used to obtain azo compound from nitro compounds?
- Sn/HCl
- H2 gas in the presence of Ni, Pt, Pd
- LiAlH4
- None of the above
Answer: (C)
Solution: Nitroalkanes can be easily reduced to the corresponding primary amines with LiAlH4. In contrast, aromatic nitro compounds on reduction with LiAlH4 give azo compounds and not primary amines.
Frequently Asked Questions (FAQs)
Q1. Why can't we reduce nitro compounds using Conc. HCl rather than Conc. sulphuric acid?
Answer: In contrast to hydrochloric acid, sulfuric acid is an oxidising agent. As a result, Conc. HCl is employed to decrease nitro compounds rather than Conc. sulphuric acid.
Q2. How basic or acidic are nitro compounds?
Answer: Acidic character: The -hydrogen linked to the carbon atom bearing the nitro group is acidic and can be readily extracted by a base due to the strong electron-withdrawing tendency of the nitro group (-I effect). Resonance stabilizes the resultant anion, which is frequently referred to as the nitronate ion.
Q3. Why do nitro compounds have an explosive nature at high temperatures?
Answer: This group's explosive properties result from the fact that when they thermally decompose, molecular nitrogen and a sizable amount of energy are also released.
Q4. How to detect nitro groups in a compound?
Answer: The Mulliken-Barker test is used to determine whether the compound has a mononitro group. The gleaming silver mirror reveals the presence of the nitro group.


