-
Call Now
1800-102-2727
Neutralization Titration - Definition, Practice Problems & FAQs
Have you ever been to Andaman and Nicobar Islands? Apart from the pristine beaches and clear sky, you know what else lies there? The Barren Island, home to one of the few active volcanoes in India.
Eruption of volcanoes lead to the spewing of lava, the viscous yellow thing which you might have seen in movies.
Have you ever wondered what happens when you pour water on to it?
Large fumes of heat rush out of the point of contact.
You know what else comes close to this sort of observation? When we mix an acid and a base, similar type of expulsion of heat is observed (albeit not of the same magnitude).
What is it called? What are the ways in which we can find out how acid and base react?
Lets beging to understand the reaction between an acid and base, termed as “Neutralization” reaction.

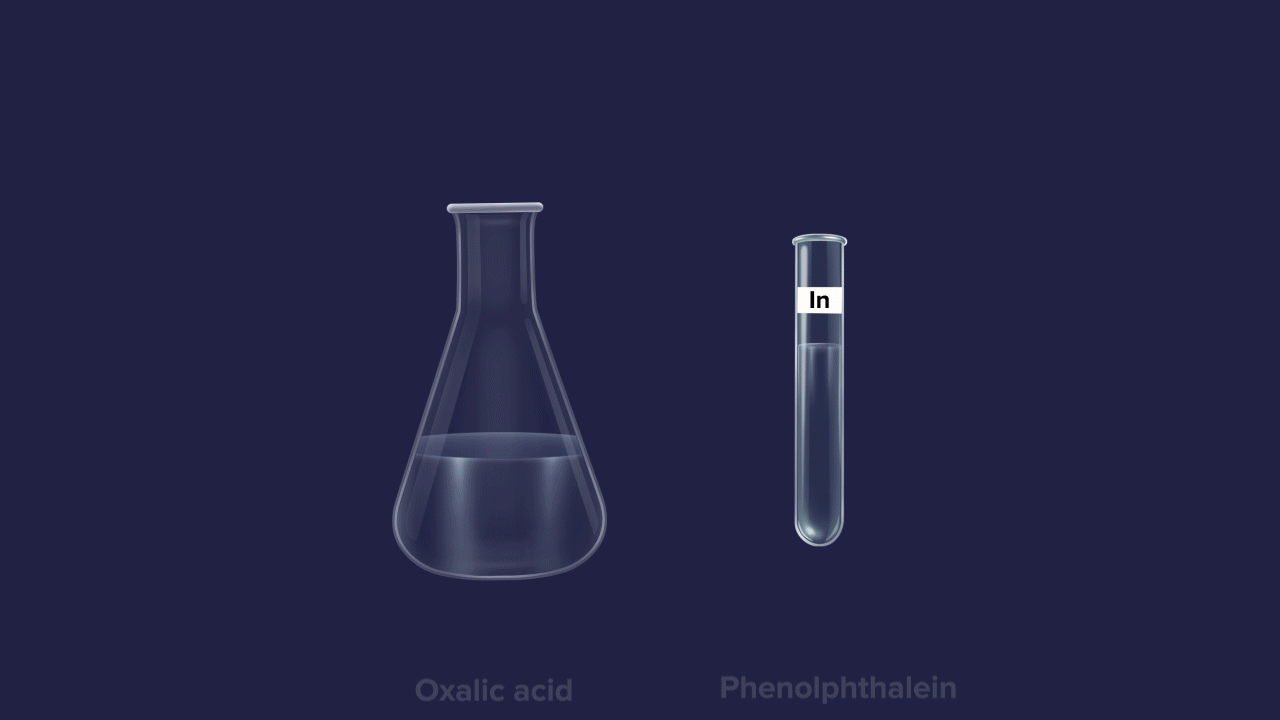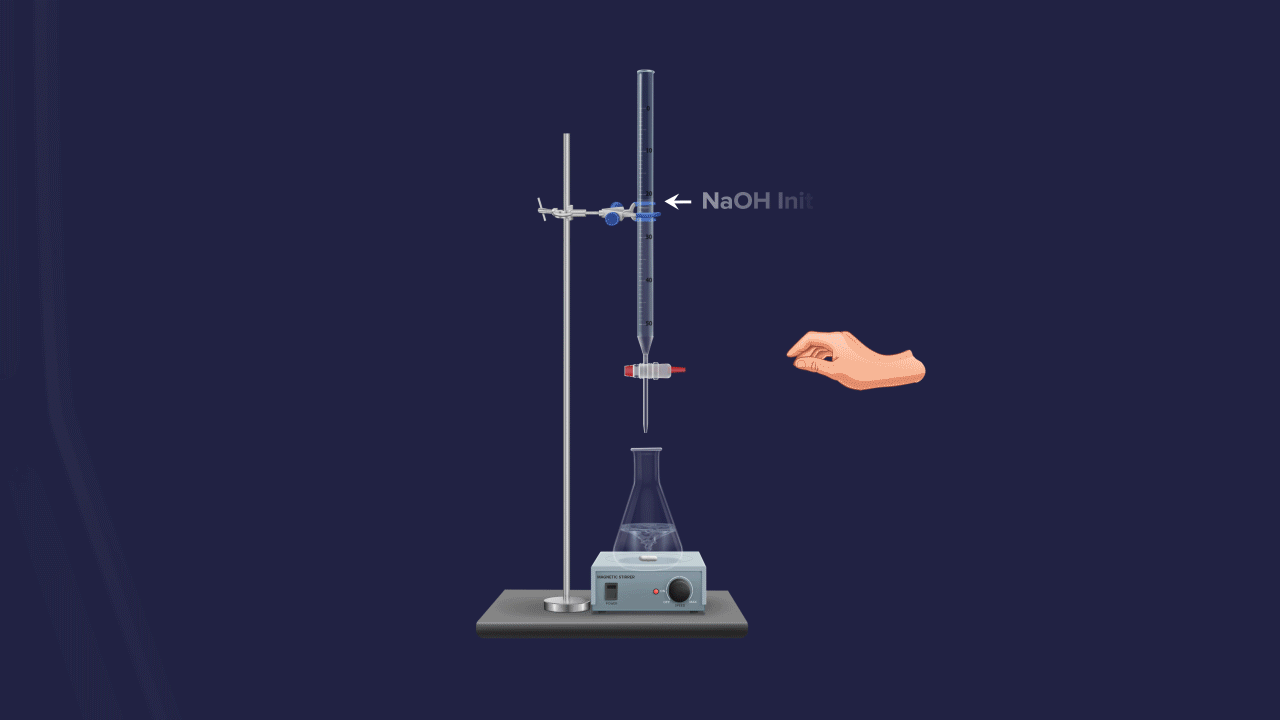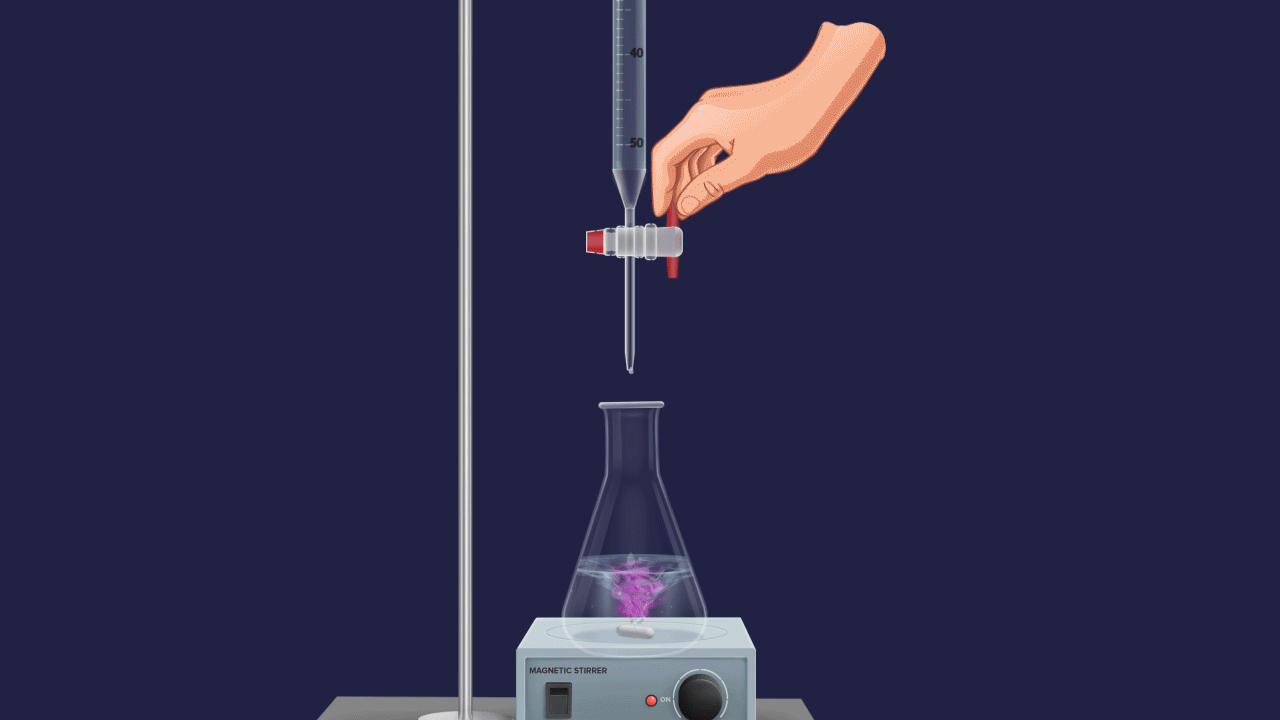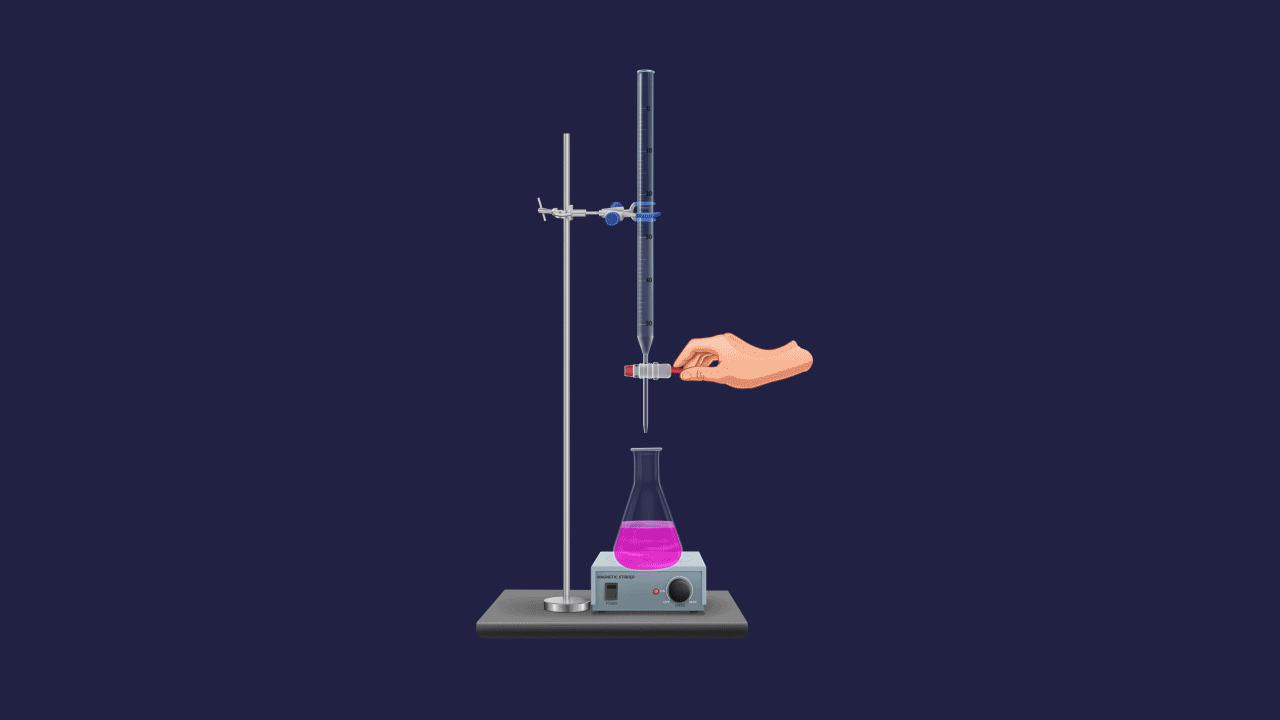
Table of contents
- Theory
- Neutralization titration
- Acid-base indicators
- Practice problems
- Frequently asked questions-FAQs
Theory
Strong or weak acids or bases are used in an acid-base titration. Specifically, acid-base titration can be used to figure out the following.
- The concentration of an acid or base
- Whether an unknown base or acid is weak or strong.
- Calculation of pKa of an unknown acid or pKb of the unknown base.
In water, the proton is usually solvated as H3O+. H2O is added to the base to lose (OH-) or gain (H3O+). Acid-base reactions are reversible.
The reactions are shown below.
Here conjugate base = [A-] , conjugate acid = BOH2+
Thus we say, Acid + Base Conjugate base + Conjugate acid
Neutralization titration
It is the method of finding the concentration of an unknown acid/base by titrating it against a base/acid with a known concentration.
For acid-base reactions,
Simple acid-base titration:
Acid + Base Salt + water (neutralization reaction)
⇒ At the equivalence point, the equivalent of Acid = equivalent base
The equivalence point is the point where:
Number of the equivalent of analyte = Number of the equivalent of titrant
Titre: Solution with an unknown concentration
Titrant: Solution with a known concentration
We can use relation, N1V1=N2V2 or NtitreVtitre=NtitrantVtitrant
Where N1=Normality of titre, V1= volume of titre
N2=Normality of titrant, V2= volume of titrant
Key terminologies
- Titration - When a solution of known strength is applied to a certain volume of a treated sample containing an indicator, it is referred to as titration.
- Titrant - A known concentration strength solution is used in the titration.
- Titre - Any solution to which the titrant is introduced and which contains the ion or species to be identified is referred to as the titre.
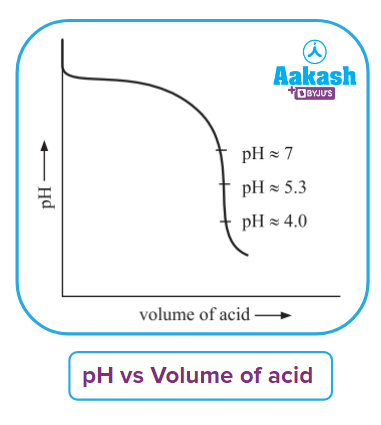
- Titration curve - A graph of pH vs. titrant millilitres that shows how pH varies vs. titrant millilitres during an acid-base titration.
- Equivalence point - The point at which just enough reagent is introduced to fully react with the material is known as the equivalence point.
- Buffer solution - A buffer solution resists pH changes even when a strong acid or base is added, or when it is diluted with water.
Titration: Endpoint refers to the point at which the indicator changes its colour in a titration.
Acid-base indicators: Indicators are generally weak organic acids or weak bases which show different colours in ionized and unionized forms or different colours in different pH ranges.
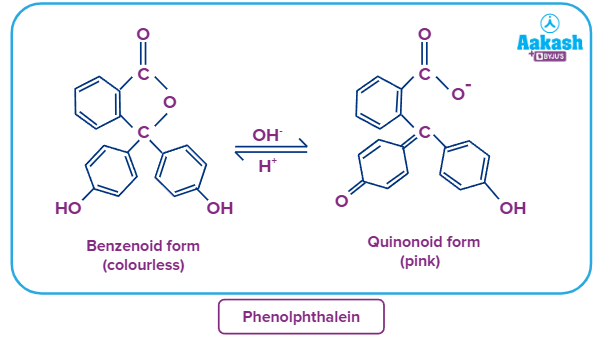
E.g-, phenolphthalein (HPh) is an organic weak acid (phenolphthalein ionized form is pink coloured and unionised form is colourless)
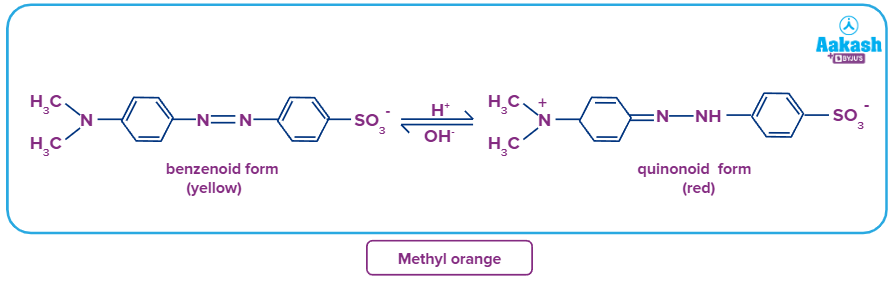
E.g- Methyl orange (MeOH) is a weak organic base (the ionized form red and the unionised form is yellow).
|
Types of titration |
Examples |
|
Strong acid - strong base |
HCl & NaOH |
|
Weak acid - strong base |
CH3COOH & NaOH |
|
Strong acid - weak base |
HCl & NH3 |
|
Weak acid - weak base |
CH3COOH & NH3 |
Titration curve can be seen from here.
List of some indicators and their colour according to pH
|
Indicator |
pH range |
Colour change |
pH range and corresponding colour change |
|
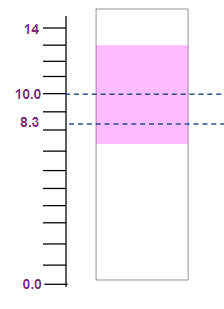
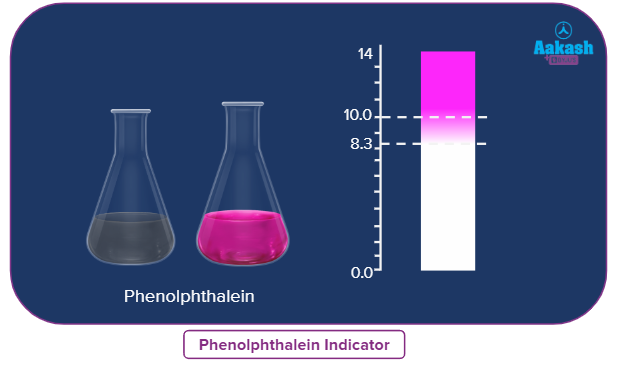
Phenolphthalein |
8.3-10 |
Colourless to Pink |
|
|
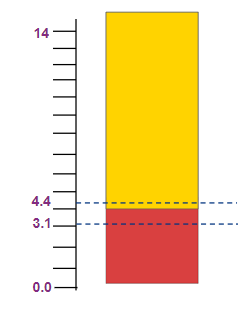
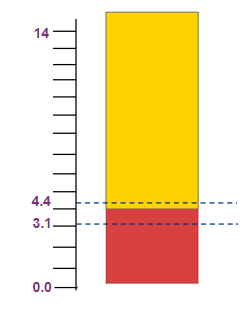
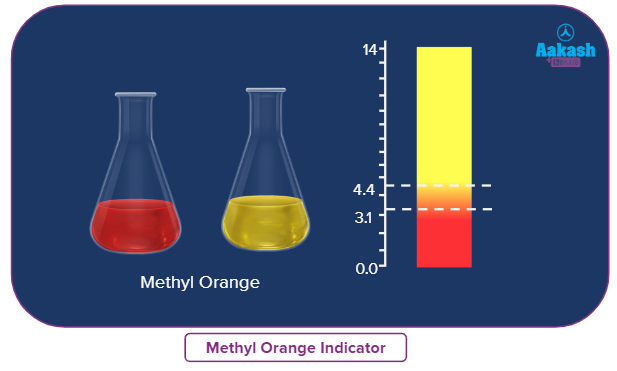
Methyl Orange |
3.1-4.4 |
Red to yellow |
|
|
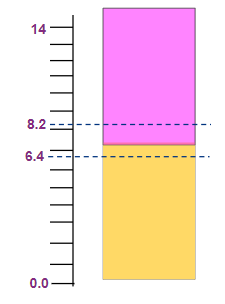
Phenol red |
6.4-8.2 |
Yellow to pink |
|
Generally, in acid-base titration phenolphthalein and methyl orange used as indicators.
Methyl orange:
Phenolphthalein:
Endpoint: The endpoint is defined as the point during titration at which the indicator changes its color.
Practice problems
Q 1. 20 g sulphuric acid is dissolved in water to prepare a 500 mL solution. 75 mL of this solution requires 75 mL of NaOH solution for complete neutralization. Find the molarity of NaOH solution.
- 0.5 M
- 0.62 M
- 0.76 M
- 0.98 M
Answer: (B)
Solution:
Weight of H2SO4 in 500 mL solution = 20 g
Weight of H2SO4 in 75 mL solution =
Molar mass of H2SO4=98 g mol-1
n-factor of H2SO4=2
n-factor of NaOH=1
Mole of H2SO4 in 75 mL solution
for complete neutralization; H2SO4+2NaOHNa2SO4+2H2O
equivalent of H2SO4=equivalent of NaOH
equivalent of H2SO4=equivalent of NaOH
We know, number of equivalent = molen-factor=NV
NNaOH= 0.62 N
Also, we know Normality = molarity n-factor
Molarity = 0.62 N x 1= 0.62 M
Q 2. 20 g sulphuric acid is dissolved in water to prepare a 500 mL solution. 75 mL of this solution requires 75 mL of NaOH solution for neutralization up to a single step. Find the molarity of NaOH solution.
- 0.41 M
- 0.62 M
- 0.34 M
- 0.23 M
Answer: (A)
Solution:
Weight of H2SO4 in 500 mL solution = 20 g
Weight of H2SO4 in 75 mL solution = (2050075) g = 3 g
Molar mass of H2SO4=98 g mol-1
n-factor of NaOH=1
Mole of H2SO4 in 75 mL solution = 398 mol
for complete neutralization; H2SO4+NaOHNaHSO4+H2O
n-factor of H2SO4=1
equivalent of H2SO4=equivalent of NaOH
equivalent of H2SO4=equivalent of NaOH
We know, number of equivalent = molen-factor=NV
NNaOH= 0.41 N
Also, we know Normality = molarity x n-factor
Molarity = 0.41 N x 1= 0.41 M
Q 3. Equal volumes of 10 g H2SO4 aqueous solution and 10 g NaOH aqueous solution are mixed together. The nature of the resulting solution will be
- Basic
- Neutral
- Acidic
- Can’t determined
Answer: (A)
Solution:
Molar mass of H2SO4=98 g mol-1
Molar mass of NaOH=40 g mol-1
Mole of H2SO4 = 1098 mol
Mole of NaOH = 1040 mol
for complete neutralization
We know, the number of equivalent = molen-factor
equivalent of NaOH> equivalent of H2SO4
So, the resulting solution is basic in nature.
Q 4. The most suitable indicator for the titration of a strong acid and weak base is
- Phenolphthalein
- Methyl orange
- Both of these
- None of these
Answer: (B)
Solution: methyl orange changes color between pH range 3.1 to 4.4.
|
Methyl Orange |
3.1-4.4 |
Red to yellow |
|
Frequently asked Questions - FAQs
Q1. How to identify which compounds could behave as an acid?
Answer: The most prevalent characteristics of acids is that the proton that is donated (or pulled off) is attached to an atom that is extremely electronegative. This atom is usually an oxygen or a halogen like chlorine (Cl), bromine (Br), or iodine (I). It's simple to spot the potentially acidic sites in a molecule once you know what to look for.
Q2. What role does the acid-base interaction play in food preservation?
Answer: In food, acid is used to delay or stop the growth of disease-causing or spoilage-causing organisms. This process can extend the shelf life of food while keeping the nutritional value approximately the same.
Q3. Describe any thermochemical evidence for the Arrhenius idea of heat of neutralization.
Answer: The heat of reaction, ΔHo, for neutralising a strong acid by a strong base is evidence for the Arrhenius Theory. The neutralising reaction is simply
and should always produce the same ΔHo per mole of water produced.
The same rules apply to all neutralizations involving strong acids and strong bases, ΔHo = -57.3 kJ per molof H2O (l) , according to research. This proves that, as anticipated by Arrhenius Theory, each neutralisation has the same reaction.
Q4. What are the different uses of acids and bases?
Answer: Following are some of the uses of acids :
- Vinegar is a diluted solution of acetic acid that has a variety of uses in the home. It's mostly used as a food preservative.
- Citric acid is a natural component of orange and lemon juice. It's also utilised in the food industry as a preservative.
- In batteries, sulfuric acid is more commonly used. This acid is commonly found in the batteries used to start vehicle motors.
Following are some of the uses of bases:
- A small amount of calcium hydroxide is used in dry mixtures for decorating or painting.
- Magnesium hydroxide, popularly known as magnesia milk, is primarily used as a laxative. It also acts as an antacid by reducing the amount of acid in the human stomach.
- Ammonium hydroxide is a commonly used reagent in laboratories.