-
Call Now
1800-102-2727
Concentration of Ore – Methods of Concentration of Ores, Practice Problems and FAQ
People have been involved in mining and quarrying since the beginning of time. The first material known to have been mined is flint, a fine-grained kind of quartz used to make weapons. One of the materials that prehistoric humans gathered for use as paint in ceremonial and artistic endeavours was ochre. Metals are mined nowadays to create items that benefit humanity. Mining produces metals including iron, copper, zinc, bronze, gold, and others that are used to create equipment ranging in size from a bell pin to a cargo ship.
We can extract these important metals from a variety of minerals, specifically ores, but these ores have impurities. The impurities must be taken out of the ore in order to increase its profitability. The metallic ores mined from the crust of the earth include non-metallic impurities such as sand, clay, and limestone. These contaminants are referred to as matrix or gangue. The concentration of the ore refers to the process of removing these contaminants from the ore. There are many techniques for concentrating ores, including hand-picking. Leaching, froth flotation, electromagnetic separation, hydraulic washing or gravity separation.
We will learn about the many techniques used and how the ore is concentrated in this article.
TABLE OF CONTENT
- Concentration of Ore
- Hydraulic Washing
- Magnetic Separation
- Froth Floatation Process
- Leaching or Hydrometallurgy
- Practice Problems
- Frequently Asked Questions – FAQ
Concentration of Ore
One of the most crucial phases in the extraction of metal from its ore is the concentration of ore, also known as the enrichment of ore or the benefaction process. To get rid of these undesirable compounds, the ores are concentrated based on the kind of impurities and their percentage proportion. By removing these impurities, we can get a concentrated ore that contains a significant amount of metal. After mining, the ores are typically found as large lumps. Using crushers or grinders, these are reduced to little fragments. This action is referred to as crushing. Then, either a ball mill or a stamp mill is used to grind these pieces into fine powder. Pulverization is the name of this process. Typically, the ores also include sand, stones, and other worthless silicates. These undesirable impurities found in ores are referred to as matrix or gangue (pronounced gang). Concentration, dressing, or benefaction of the ore are terms used to describe the process of removing these contaminants from the ores. Depending on the ore, both physical and chemical processes are employed to separate the valuable minerals from the gangue.
Concentration of Ore – Physical Methods
The process of segregating undesirable particles from the necessary components is the essence of the separation method. We will study in-depth information about several physical separation techniques in this particular article.
Hydraulic Washing
The difference in the specific gravities of the ore and gangue particles serves as the basis for this separation. A moving stream of water is used to wash or agitate the powdered ore. Sand, clay, and other lighter particles are washed away as the heavy ore particles settle down. Wilfley tables or hydraulic classifiers are employed for this. A wooden table known as a "Wilfley table" has a floor that slopes and are attached with long wooden strips called "riffles". A stream of water contains powdered ore suspended in it. The gangue particles are swept away by the water stream while the heavier ore particles gather behind the riffles. A powerful stream of water is injected from the bottom of the classifier while powdered ore is lowered from the top. This process is typically used to concentrate oxide and carbonate ores. For instance, the gravity method is used to concentrate iron ore (haematite) and tin ore (cassiterite).

Magnetic Separation
This technique can be employed for separation when one of the components, such as the ore or an impurity, has a magnetic property. One of the rollers in the magnetic separator, which has a belt rotating on two of them, is a powerful magnet. Non-magnetic ore powder is dumped onto the belt from one end, and when it reaches the magnetic end, magnetic particles are drawn to it and fall closer to the roller while non-magnetic particles fall farther away. This technique concentrates on ferromagnetic ores. This technique, for instance, can be used to separate the magnetic mineral wolframite (FeWO4) from the non-magnetic ore cassiterite (SnO2).

Froth Floatation Process
Froth flotation is a physical technique for separating minerals from gangue that relies on variations in the wettabilities of the two materials in a liquid solution. Sulphide ores are concentrated using this technique. The technique is based on the water and foaming agent's favoured wetting characteristics. The powdered ore is mixed with water that also contains sodium ethyl xanthate (a collecting agent) and pine oil (a foaming agent). Aniline and cresols are also included. They serve as stabilisers for froth. The liquid is now extensively stirred, and a strong stream of air is then sent through, dispersing the oil into particles the size of colloid.

An activator is a chemical that is applied to help the collector remain attached to the flotation particles, improving the ore's ability to float. For instance, ZnS has poorer floating characteristics as compared to CuS. Therefore, CuSO4 is employed as an activator to create a coating of CuS on the surface of ZnS and to enhance its floating properties.
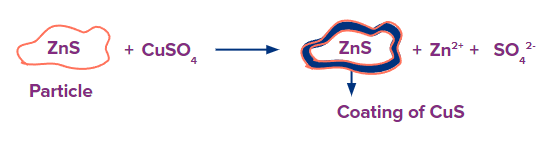
A depressant is a chemical that is used to decrease the ore particles' tendency to float. Galena (PbS), for instance, is connected to another sulphide impurity, ZnS, which similarly floats to the top with the froth. By generating a soluble compound with KCN, the reagent NaCN/KCN inhibits the floating properties of ZnS.
ZnS(s)+4[NaCN/KCN][Zn(CN)4]2-+4[Na+/K+]+S2-
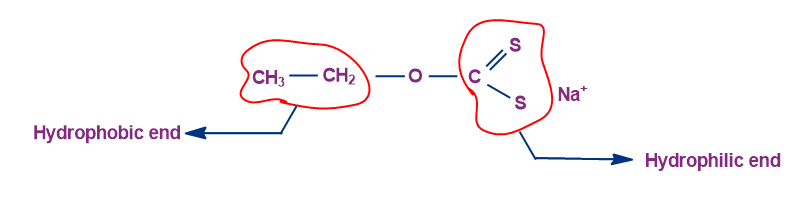
The hydrophilic end favours staying in the water, while the hydrophobic end likes staying in the air. The molecule exhibits floating properties because the air-water boundary is the optimum place for it to stay because it has both hydrophobic and hydrophilic components.
Concentration of Ore – Chemical Methods
Leaching or Hydrometallurgy
This separation involves chemicals to concentrate ore. It entails treating the ore with a suitable reagent in order to make it soluble while keeping impurities insoluble. The contaminants in this situation include those that are insoluble, and are separated by filtering. The process is demonstrated by the examples below.
1. Leaching of alumina from bauxite ore (Bayer’s process): Iron oxide, titanium oxide, and silica are impurities found in bauxite ore. When an aqueous solution of sodium hydroxide is used to digest the powdered ore (45%) under pressure at roughly 200°C, the alumina dissolves at (35–36 bar), generating soluble sodium aluminate. Iron oxide (Fe2O3), titanium dioxide (TiO2), and silica remain insoluble part.

Filtration is done on the sodium aluminate solution. It could also contain a small amount of free sodium hydroxide and sodium silicate. By passing CO2 gas and hydrated Al2O3, the filtrate is neutralised, and precipitation occurs. Currently, the remedy is infused with newly formed aluminium hydroxide that causes precipitation.

The sodium silicate remains in the solution. The hydrated alumina is filtered, dried and heated to approximately 1200°C to obtain pure alumina.

2. Leaching of silver and gold (MacArthur and Forest’s cyanide process): Leaching is a method used to extract gold and silver from their native ores or silver from argentite ore (Ag2S). The Mac-Arthur Forest cyanide procedure is another name for it. In this procedure, a diluted solution (0.5%) of sodium or potassium cyanide is applied to finely powdered argentite ore, native silver particles, or native gold particles while an air current is continually cycled. Silver and gold then dissolve by creating corresponding complex cyanides as a result. The impurities are not changed. These are removed via filtering.
Na2S is largely oxidised to Na2SO4
Au and Ag is recovered from the solution by the addition of electropositive metal like zinc.

Recommended Videos
Metallurgy Class 12 Chemistry One-Shot (Full Chapter) by Rakhi Ma'am | JEE Mains & Advanced 2023
Practice Problems
1. Which among the following separation method is the chemical method for the concentration of ores?
a. Leaching
b. Froth Floatation Process
c. Magnetic Separation
d. Hydraulic washing
Answer: A
Solution: There are two ways to methods to concentrate a ore, namely the physical and the chemical methods. Depending on the ore, both physical and chemical processes are employed to separate the valuable minerals from the gangue.
Physical separation techniques include levigation or gravity separation or hydraulic washing, magnetic separation, and froth floatation processes, whereas the chemical separation techniques include leaching or hydrometallurgy.
So, option A is the correct answer.
2. The concentration of the ore is carried out with __________in Bayer's purification process for aluminium ore.
a. HCl
b. NaOH
c. AgBr
d. KCl
Answer: B
Solution: Iron oxide, titanium oxide, and silica are impurities found in bauxite ore. When an aqueous solution of sodium hydroxide is used to digest the powdered ore (45%) under pressure at roughly 200°C, the alumina dissolves at (35–36 bar), generating soluble sodium aluminate. Iron oxide (Fe2O3), titanium dioxide (TiO2), and silica remain insoluble part.

So, option B is the correct answer.
3. Froth floatation process is generally used for __________ ores.
a. Oxide ores
b. Carbonate ores
c. Iron ores
d. Sulphide ores
Answer: D
Solution: Froth flotation is a physical technique for separating minerals from gangue that relies on variations in the wettabilities of the two materials in a liquid solution. Sulphide ores are concentrated using this technique. The technique is based on the water and foaming agent's favoured wetting characteristics. The powdered ore is mixed with water that also contains sodium ethyl xanthate (a collecting agent) and pine oil (a foaming agent). Aniline and cresols are also included. They serve as stabilisers for froth. The liquid is now extensively stirred, and a strong stream of air is then sent through, dispersing the oil into particles the size of colloid.

So, option D is the correct answer.
4. A ____________is a chemical that is used to decrease the ore particles' tendency to float.
a. Depressant
b. Activator
c. Froth
d. Gangue
Answer: A
Solution: A depressant is a chemical that is used to decrease the ore particles' tendency to float. Galena (PbS), for instance, is connected to another sulphide impurity, ZnS, which similarly floats to the top with the froth. By generating a soluble compound with KCN, the reagent NaCN/KCN inhibits the floating properties of ZnS.
ZnS(s)+4[NaCN/KCN][Zn(CN)4]2-+4[Na+/K+]+S2-
The hydrophilic end favours staying in the water, while the hydrophobic end likes staying in the air. The molecule exhibits floating properties because the air-water boundary is the optimum place for it to stay because it has both hydrophobic and hydrophilic components.
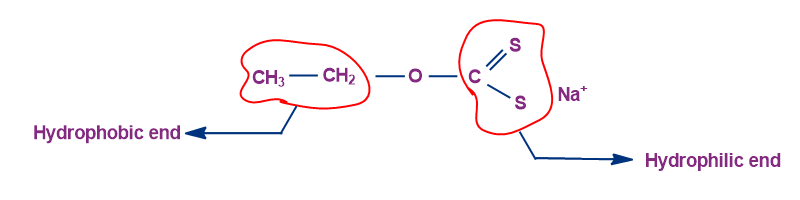
So, option A is the correct answer.
Frequently Asked Questions – FAQ
1. How is concentrated ore transformed into oxide for metal isolation?
Answer: For isolation, concentrated ore must be converted into a particular form that is suitable for reduction. Oxide ores such as sulphide ores, carbonate ores, and hydrated ores are easy to reduce. Generally, conversion of ore is carried out by either calcination or roasting. Calcination is generally used for used for carbonate ores. Calcination is the concentration of ore into its metal oxide by heating in the absence of air or limited supply of air. Roasting is generally used for used for sulphide ores. Roasting is the concentration of ore into its metal oxide by heating at a very high temperature below the melting point of the metal in excess supply of air in a reverberatory furnace.
2. Why is ore concentration important?
Answer: In order to purify the ore, gangue particles or other impurities are removed during the concentration process. This procedure is also known as benefaction and ore-dressing. The process of extracting pure metal from ore is extremely important.
3. How can a mineable mineral resource become unprofitable?
Answer: The ore's price may increase or decrease. The cost of mining the mineral may increase or decrease. For instance, if the ore is difficult to access and the cost of fuel increases. This could make mining the mineral deposit unprofitable.
4. What are the minerals that are found in the human body?
Answer: Minerals are those substances found in meals and in the ground that our bodies require for healthy growth and development. Selenium, manganese, molybdenum, fluoride, copper, chromium, iodine, zinc, iron, magnesium, sodium, potassium, phosphorus, and calcium are among the nutrients that are crucial for good health.


