-
Call Now
1800-102-2727
Mesomeric Effect - Electronic Displacement Effects, Types of Mesomeric Effect, Applications, Practice Problems & FAQs
Have you ever shared your things with your friends? I guess most of you have done this.
Let us consider a simple example and try to learn the concept behind that.
There was a group of 5 friends. They all studied together and were in the same school. All of them visited the library and saw a comic storybook. That book has only one copy left. They asked the librarian to assign that book to one of them. Librarian told the assigned person to share the book with each one among them so that everyone will get benefit.
All five friends rotated the book to each other after some particular interval of time. All are enjoying reading that comic book. All are equally benefited by sharing the book with each other. As the submission date came closer the librarian asked the group to submit the book. It was difficult for the librarian to guess the name of the bookholder person.

I guess you must ask what is the reason behind telling this story? Have you heard the term delocalisation?
The same process happens in chemistry also. Here we will study about delocalisation of electrons in conjugated systems. It is difficult to decide the current position of electrons if the electrons are delocalised in a conjugated system. This type of electronic displacement effect is known as mesomeric effect.
Let us study more about this effect and its application part in much more detail!
TABLE OF CONTENT
- Electronic displacement effect
- Mesomeric effect
- Types of mesomeric effect
- Applications - Mesomeric Effect
- Practice Problems
- Frequently asked questions-FAQs
Electronic displacement effect:
The presence of a suitable attacking reagent may cause electron displacement in an organic molecule. The bond becomes polarized as a result of this type of electronic displacement. The displacement of an electron in an organic molecule can occur in one of these ways either in the ground state, with the help of an atom or a substituent group, or a suitable attacking agent.
On this basis these displacements of electrons and effects are of two types which are categorized below:
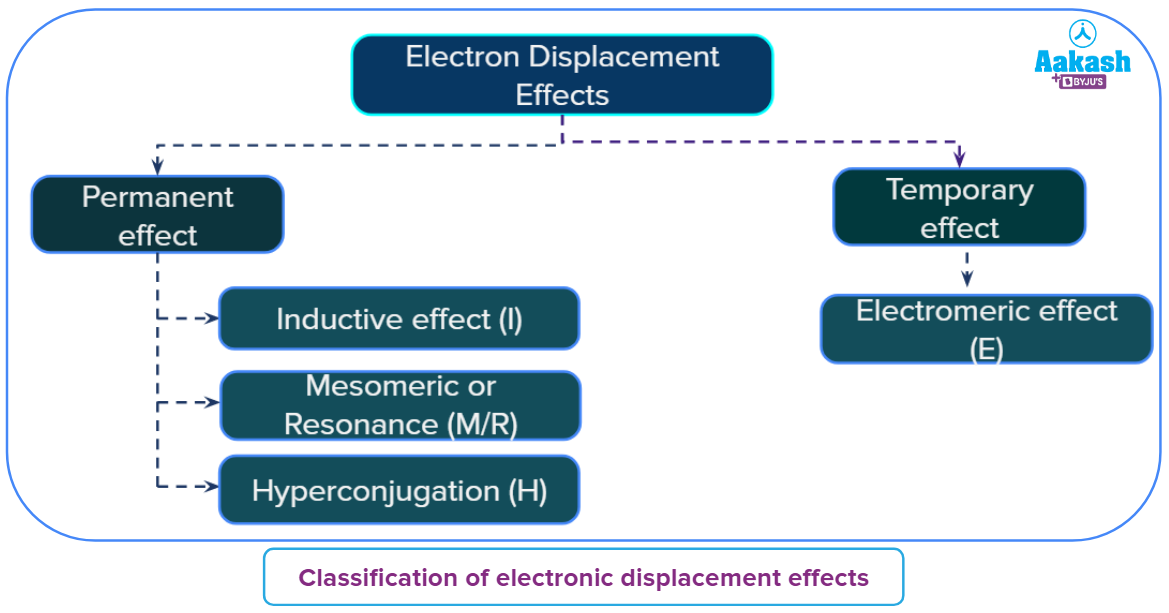
On this page, we are focusing on the Mesomeric effect which is categorized under the permanent effect.
Mesomeric effect:
- Scientist Linus Pauling came up with the term mesomerism, which is a synonym for resonance. However, the word "resonance" has gained popularity and is now widely used in English.
- Polarity is developed in conjugate systems by the delocalisation of non–bonding electrons or π bond electrons or by the interaction of two π-bonds or between a π-bond and lone pair of electrons present on an adjacent atom due to the phenomenon of resonance.
- Based on relevant resonance structures, the effect, which is represented by the letter M, is used to qualitatively describe the electron-withdrawing or releasing capabilities of substituents.
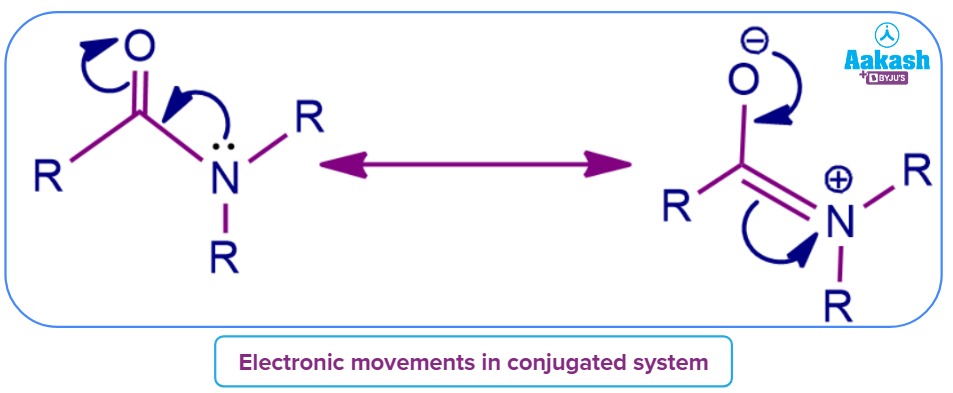
Types of mesomeric effect:
Mesomeric effect is further divided into two categories. There are
- +M effect (positive mesomeric effect)
- -M effect (negative mesomeric effect)
Let us discuss each in more detail!
+M effect (positive mesomeric effect):
- In this effect, an atom or substituent group connected to the conjugated system transfers electron density towards that system. Due to this electron displacement, some individual locations have large electron densities.
- The conjugate system develops a negative charge as a result of the +M effect, or its electron density rises. These conjugate complexes react more favorably with electrophiles and less favorably with nucleophiles.
- It is important to remember that for the group to exhibit the +M effect, there must be either a lone pair of electrons or a negative charge.
- Groups showing +M effect are mentioned here. R is considered an alkyl group.
- If we take any group G having a lone pair or an extra electron like an anion, then below are the possible cases. G can be any of these groups like -NH2,-OH,-Cl,-OR etc.
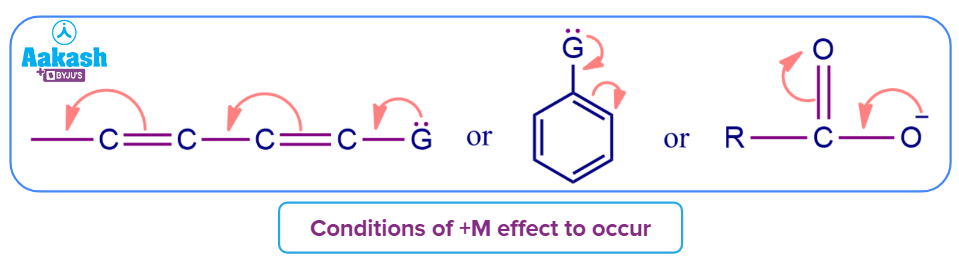
Example of +M effect are:
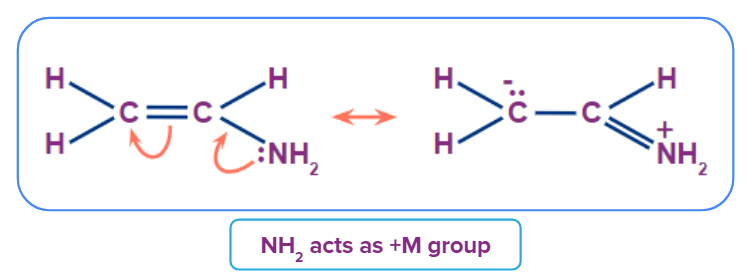
Example 1: Here -NH2 has lone pair present on Nitrogen atom which is involved in delocalisation in the open chain conjugated system.
Example 2: Here -X atoms have lone pairs present which are involved in delocalisation in the cyclic conjugated system. X represents Cl,Br,I.
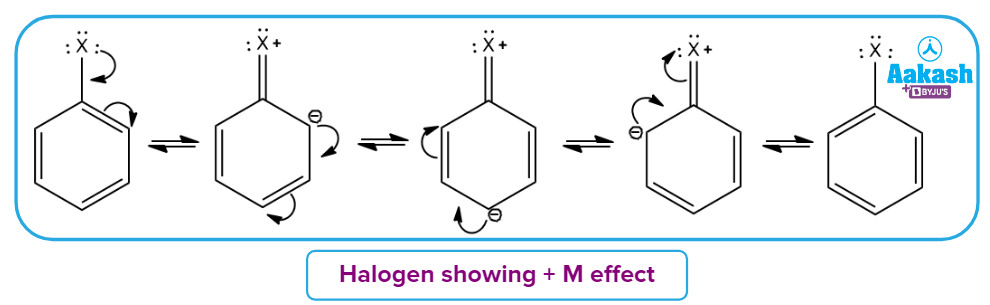
Example 3: When a –ive charge is present on the atom or substituent group attached to the conjugated system.
Order of +M group:
There are many groups having lone pairs or negative charges that when attached to a conjugated system show +M effect but it is difficult to decide which is showing more +M effect. There is a lot of confusion regarding this. To avoid this confusion, scientists have decided on an order.
Based on this order we can easily decide which group has more +M effect.
Examples:
- If there are two conjugated structures and both are showing +M effect then the above order can help us in deciding which group shows more +M effect.
Let's say we have two different conjugated structures. How can you decide which structure has more electron density? We can simply compare the +M order of the substituent group attached to the conjugated system and decide which system is rich in electron density.
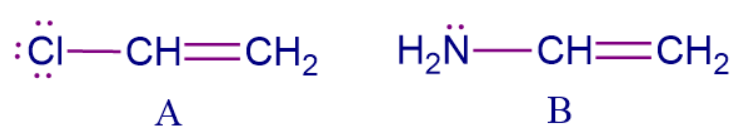
Here molecule B is more electron-rich than molecule A. The substituent attached to the conjugated system has lone pairs available and they are in conjugation with the 𝜋 electrons present in the double bonds, therefore both show +M effect but according to the +M order, -NH2 comes before -Cl hence, the correct order for electron-rich system is
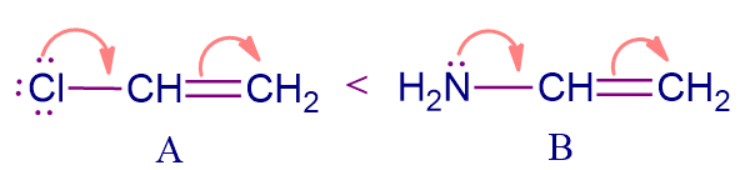
-M effect (negative mesomeric effect):
- When 𝜋-bond electrons are pulled off from the conjugate system to a particular group, the conjugate system's electron density decreases, which causes the negative mesomeric (-M) effect.
- It is important to remember that for the -M effect to occur, the group must either have a positive charge or a vacant orbital.
- By reducing the electron density in the conjugate system, the -M effect increases a molecule's reactivity toward nucleophiles while decreasing its reactivity toward electrophiles for the same reasons.
- Groups showing -M effect are mentioned here. These groups are:
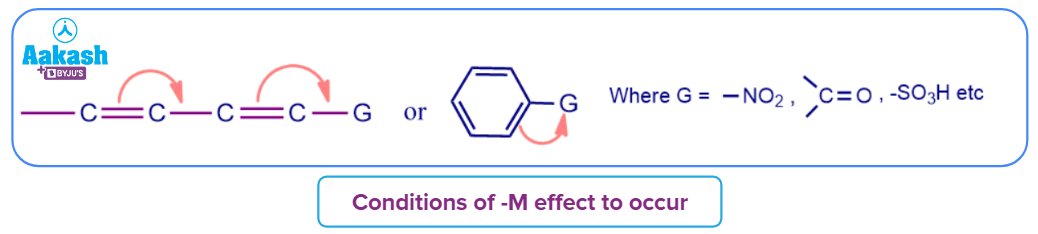
Example of -M effect are:
Example 1: Here a substituent or group attached to the conjugated system is having vacant orbital. This will show -M effect.
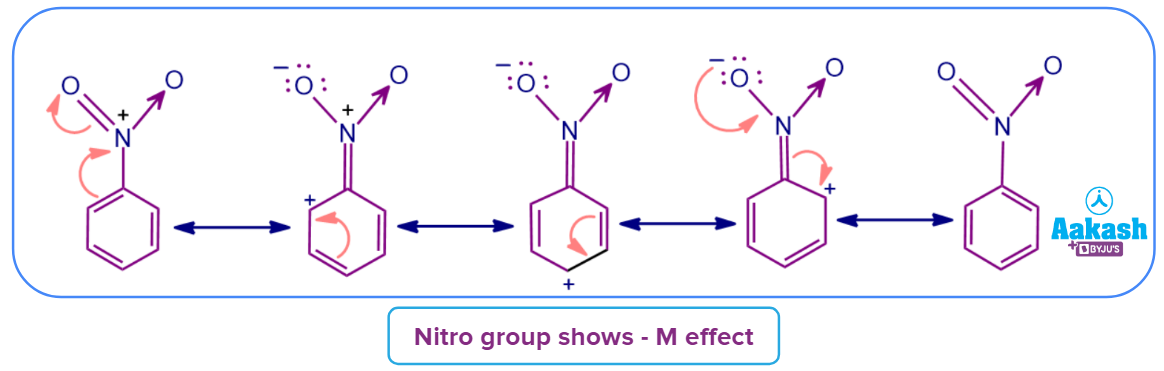
Example 2: Here the substituent or group attached with the conjugated system is -NO2. The nitro group in nitrobenzene, designated as -NO2, shows the -M effect as shown below as a result of the delocalization of conjugated electrons. Particularly at the ortho and para regions, the electron density on the benzene ring decreases.
Order of -M group:
There are many groups having vacant orbitals or positive charges that when attached to a conjugated system show -M effect but it is difficult to decide which is showing more -M effect. There is a lot of confusion regarding this. To avoid this confusion, scientists have decided on an order.
Based on this order we can easily decide which group has more -M effect.
Example:
- If there are two conjugated structures and both are showing -M effect then the above order can help us in deciding which group shows more -M effect.
Let's say we have two different conjugated structures. How can you decide which structure has more electron density? We can simply compare the -M order of the substituent group attached to the conjugated system and decide which system is rich in electron density. The group present in the beginning of the order will pull more electrons and make the system more electron deficient and vice-versa.
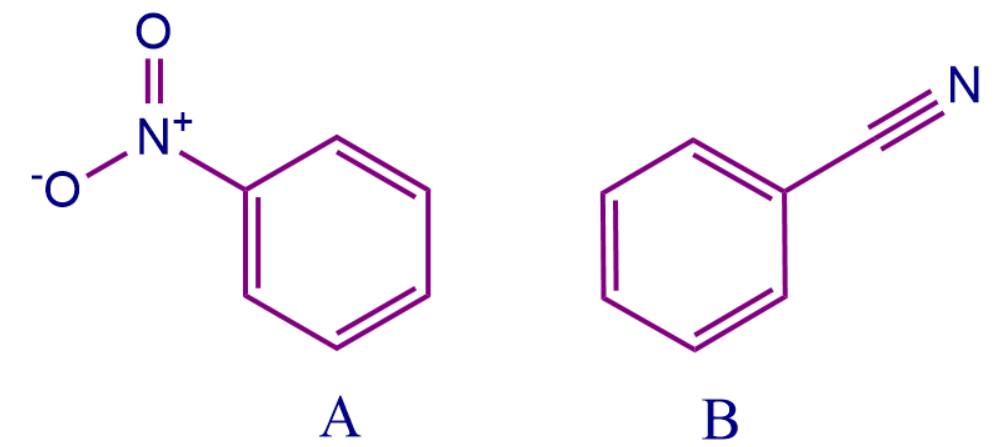
Here molecule B is more electron-rich than molecule A. The substituent -NO2 attached to the conjugated system are in conjugation with the + ve charge and 𝜋 electrons present in the double bond of the benzene ring whereas -CN the group attached is in conjugation with 𝜋 electrons, Both are pulling electrons from the benzene ring attached to them. Therefore both show -the M effect but according to the -M order, -NO2 comes before -CN Hence, the correct order for electron-rich system is A < B.
Applications - Mesomeric Effect:
The following are some of the mesomeric effect's numerous applications:
The Stability of Carbocation:
One use of the mesomeric effect is to enhance the stability of the carbocation. All aromatic compounds are always more stable than non-aromatic compounds due to the influence of resonance. More conjugated systems or the number of resonating structures greater will be stability.
- It has been discovered that the carbocation in which the positive charge is conjugated with a double bond is more stable. Delocalization of π-electrons of conjugated double bonds results in resonant structures, which leads to increased stability. For Example, An allylic carbocation is more stable than the corresponding alkyl cation due to the resonance. When there is true conjugation between the vacant p orbital and adjacent lone pair or electrons, strong stability is created.
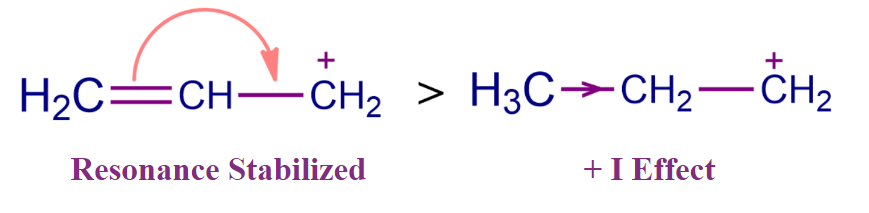
The Stability of Carbanion:
The stability of the carbanion is increased by the presence of double bonds or an aromatic ring close to a negatively charged carbon atom due to resonance.
For example, because of resonance, the negative charge on the benzyl carbanion was distributed over other carbon atoms, making it more stable than the ethyl carbanion.
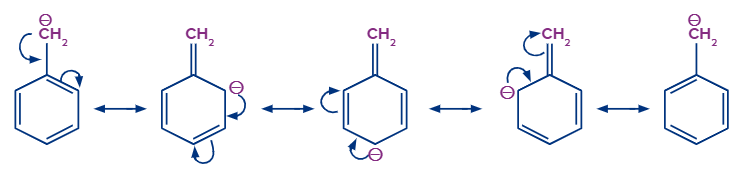
Stability of Free Radical:
Because of the delocalisation of the unpaired electrons over the system, simple alkyl radicals are less stable than allylic and benzylic forms of free radicals.
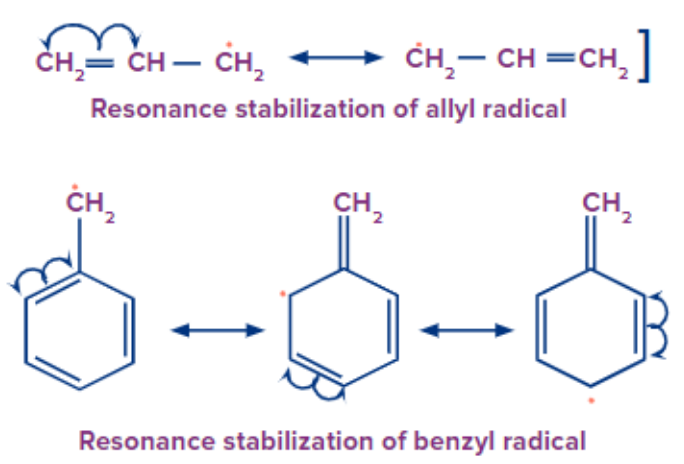
Acidic and Basic strength:
- The most prominent factor in determining the strength of an acid is the stability of its conjugate base—the more stable the conjugate base is, the stronger the acid will be.
- Based on electron pair availability, basicity may be seen in a practical manner. The easier it is for electrons to be transferred to create a new bond, the stronger the base will be.
- The conjugated system after a loss of proton forms a stable conjugate base then it is more acidic than another non-conjugated system. The mesomeric effect helps in deciding the acidic and basic nature of the system.
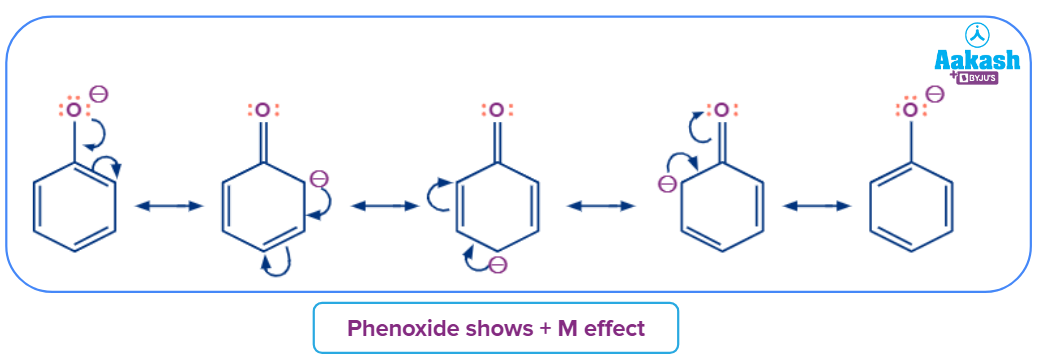
For example: To compare the acidic strength between cyclohexanol and Phenol, we check the stability of the conjugate base formed after the removal of the proton. Here phenoxide ion is stabilized by mesomeric effect and thus formed a more stable conjugate base than cyclohexanol.
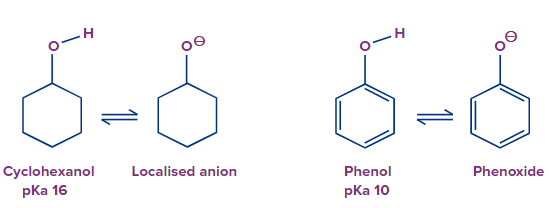
Example 2: When two compounds are showing Mesomeric effects then we should consider a few points.
Acidic strength ∝ -M group
Basic strength ∝ +M group
Let us understand a few important cases.
Case 1: Let us consider the below-mentioned system, and we have to arrange the acidic order here.
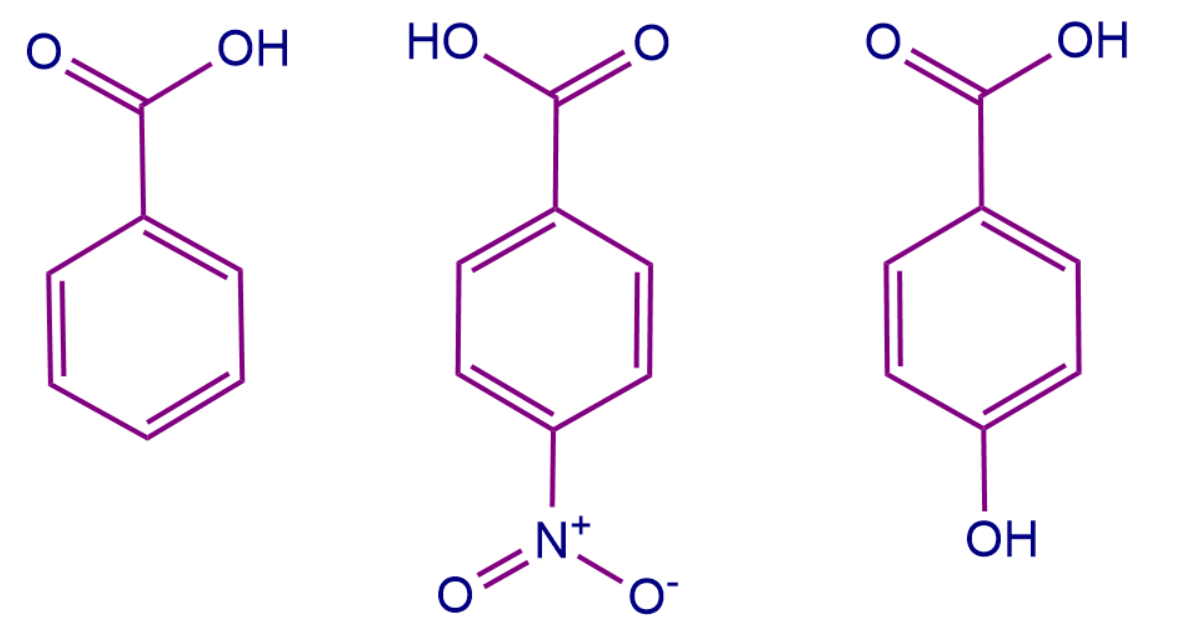
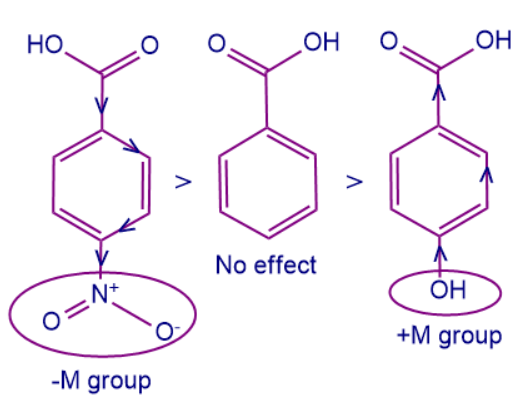
Here A is benzoic acid whereas B and C all are derivatives of benzoic acid. To arrange the acidic strength we should remember that -M group attached to the conjugated system withdraws more electron density and it is easy to remove protons from the conjugated system. Whereas +M group increases the electron density making it difficult to remove the proton and thus decreasing the acid strength of the system.
Case 2: Let us consider the below-mentioned system, and we have to arrange the basic order here.
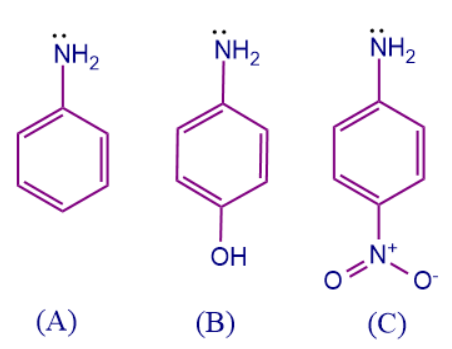
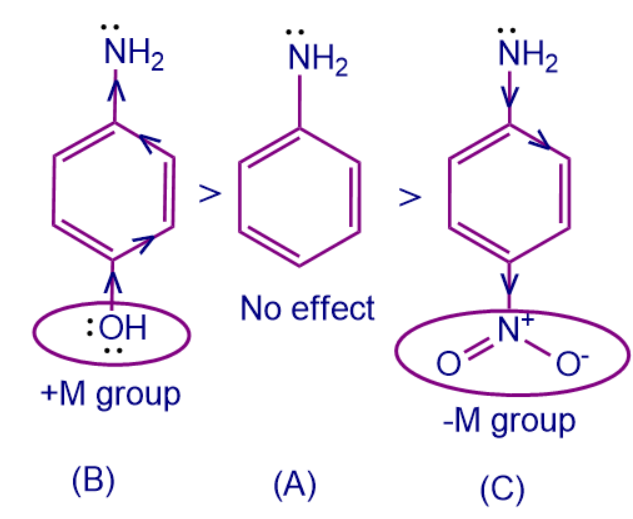
Here A is Aniline whereas B and C all are derivatives of aniline. To arrange the basic strength we should remember that +M group attached to the conjugated system donates more electron density and it is easy to provide electrons to an electrophilic species. Whereas -M group pulls the electron density making it difficult to donate electrons to the needed species and thus decreases the basic strength of the system.
Related video link: Mesomeric effect
Practice Problems:
Q1. Which among the following is -M group?
- -NO2
- -CN
- -SO3H
- All of the above.
Answer: D
Solution: When 𝜋-bond electrons are pulled off from the conjugate system to a particular group, the conjugate system's electron density decreases, which causes the negative mesomeric (-M) effect. In the mentioned example, all species pull electrons from the attached conjugated system so they behave as -M group.
Q2. From the below-mentioned figure, which type of effect is shown by the methoxy group in structure I?
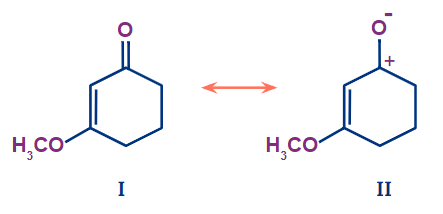
- Only +I effect
- Only -I effect
- -I effect and +M effect
- -I effect and -M effect
Answer: C
Solution: In the above figure, the attached group is -OCH3 which is a +M group because the lone pairs available on oxygen are involved in delocalisation with the conjugated system. -OCH3 is attached with a carbon atom developing polarity due to difference in electronegativity between oxygen and carbon. Oxygen is more electronegative than Carbon so the electron pull is towards -OCH3 group behaving as -I effect.
Q3. Which among the following is a +M group ?
- -O-
- -CN
- -SO3H
- -COR
Answer: A
Solution: It is important to remember that for the group to exhibit the +M effect, there must be either a lone pair of electrons or a negative charge. -O- have negative charge over oxygen atom. Other species behave as +M effect.
Q4. Arrange the acidic order in the below-mentioned compounds.
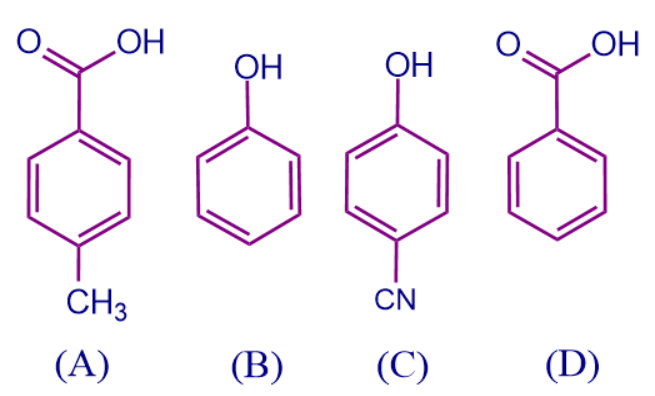
- A > B > C > D
- D > A > C > B
- A > D > C > B
- C > B > D > A
Answer: B
Solution: We can decide the order by checking the attached substituent groups and the electronic effects they are showing.
- In the above-mentioned compounds, compound A and D are derivatives of benzoic acid so they are more acidic than compound B and C.
- Among A and D, A is benzoic acid which has no substituent attached to it. In the compound, D -CH3 is attached to the benzoic acid ring, which shows +I effect and increases the electron density over the ring and thus removal of a proton is difficult. So acidic order among D and A is
D > A.
- Both compounds B and C are phenol derivatives where -CN group attached to C compound shows -M effect and there is no substituent attached to B, so comparing both -M effect increases the acidic strength of compound C than B.
- So the correct order of acidic strength is D > A > C > B
Frequently Asked Questions-FAQs:
1. Is resonance the same as mesomeric effect?
Answer: The primary distinction between the resonance effect and the mesomeric effect is that the former explains how a molecule's lone pair of electrons and bond pair of electrons define its chemical structure, while the latter explains how a functional group stabilizes a molecule's chemical structure.
2. Who discovered the mesomeric effect?
Answer: In his 1934 publication titled "Principles of an Electron Theory of Organic Reactions," Christopher Kelk Ingold is said to have made the discovery of the mesomeric effects and thus considered the discovery of the mesomeric effect.
3. Which has a bigger impact, inductive or mesomeric?
Answer: The mesomeric effect is thought to be more powerful and dominant than the inductive effect in the majority of substituents. Halogens are an apparent exception.
4. Is there a mesomeric impact with methyl?
Answer: Due to its +I inductive action, the methyl group is also known as an electron-donating group, but as it does not take part in conjugation, it cannot be said to have a +M impact.


