-
Call Now
1800-102-2727
Lunar Caustic - Definition, Preparation, Properties, Applications, Health and Risk Consequences, Practice Problems, FAQs
Many of you must visit chemistry labs and have performed many tests like salt analysis, titrations and distinguish tests. Your teachers helped you in performing these laboratory tests. Have you done major tests for differentiating aldehyde and ketones? I guess a few of you must have performed this “Tollens test” with the help of your teachers.
Do you remember the major component from which we prepare the tollens reagent? Most of you must have guessed it well, yes it is silver nitrate. A qualitative laboratory test called the Tollens test also referred to as the silver-mirror test, is used to distinguish between an aldehyde and a ketone. It takes advantage of the fact that aldehydes are easily oxidized whereas ketones are not.
Lunar caustic was previously the name given to it since early alchemists referred to silver as luna and connected it to the moon.
Let us study one of the most important compounds of silver i.e, silver nitrate in detail!
Table of Content:
- What is Lunar caustic?
- Preparation of silver nitrate
- Physical Properties of silver nitrate
- Chemical Properties of silver nitrate
- Applications of silver nitrate
- Health and risk consequences.
- Practice Problems
- Frequently asked questions-FAQs
What is Lunar caustic?
Here are a few points related to lunar caustic, some basic information is mentioned below:
- AgNO3 stands for silver nitrate in chemistry and its common name is lunar caustic.
- The silver cation (Ag+), as well as the nitrate anion (), forms an ionic connection.
- Due to its ionic nature, this chemical dissociates into its component ions and dissolves readily in water.
- When contrasted to silver halides, which are utilized in photography due to their extreme sensitivity to light, AgNO3 is relatively stable while exposed to light.
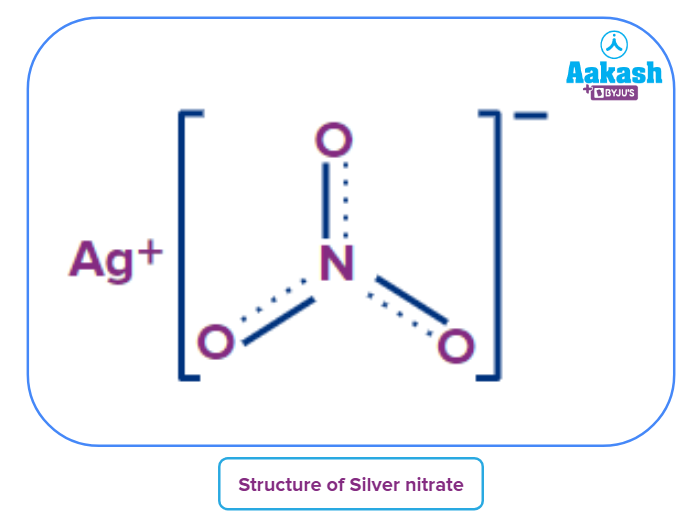
Structure of silver nitrate:
In the nitrate ion, three oxygen atoms are linked to one nitrogen nucleus.
- The strength and bond length of the nitrogen-oxygen bonds of this ion are comparable.
- Silver nitrate has a molar mass of 169.87 g mol-1
- The silver cation (Ag+) and nitrate ion () are present in their chemical structure.
- Silver ion has a net +1 charge and nitrate ion has a net charge of -1 because the core nitrogen atom is covalently linked to three oxygen atoms with a partial double bond character.
Preparation of Lunar caustic:
- Silver and weak nitric acid are heated to make it. When the silver nitrate crystals separate, the solution is concentrated and cooled.
Physical properties of Lunar caustic:
- Silver nitrate has an orthorhombic crystal structure.
- Under STP conditions, the melting and boiling points of silver nitrate are 482.8 K and 713 K, respectively.
- There is no color or smell of silver nitrate.
- Silver nitrate dissolves easily in water.
- At 0 °C, silver nitrate has a solubility of , whereas, at 25 °C, it has a solubility of .
Chemical properties of lunar caustic:
- At 440 °C, this substance completely disintegrates, releasing oxygen, nitrogen dioxide, and silver.
- One of the hazardous features of AgNO3 is that it is toxic and corrosive.
- When the silver in this mixture is taken out copper nitrate results.
- A white precipitate of silver cyanide forms when KCN is introduced to silver nitrate; this precipitate dissolves any excess KCN to generate the complex salt potassium Argento cyanide(KAg(CN)2).
- An useful laboratory reagent for identifying different acidic radicals is silver nitrate.
Applications of lunar caustic:
- Numerous industries, including biology, biotechnology, and medicine, use silver nitrate.
- This chemical has also been applied in the treatment and eradication of unwanted warts in humans.
- Silver nitrate can be used as an antiseptic in many healthcare settings once combined with water to produce a concentration of 0.5 percent.
- In analytical chemistry, this reaction is used to detect the presence of halide anions, such as iodide and chloride ions.
- A variety of silver-based explosives can be produced through the precipitation of silver nitrate.
- Due to the fact that other ligands can bind to silver ions in place of the nitrate ion, silver nitrate is a very adaptable chemical.
- Alkene mixtures can be separated with this chemical because the silver cation interacts with them in a reversible way.
- In the study of inorganic chemistry, this molecule is utilized to extract halides.
- This substance is used to create photographic films because, when exposed to halide ions, it can precipitate silver halides.
Risk and health consequences due to Lunar caustic:
- Silver nitrate should only be handled carefully because it is toxic and reactive. In extremely diluted concentrations, it is used in medical applications.
- Higher quantities of silver nitrate could result in damage, whereas low amounts generate a greyish-black coloring of the tissues on the surface or in the eyes.
- If inhaled, this will irritate and damage mucosal membranes.
- One must properly wash their hands with lots of fresh water after coming into contact with silver nitrate. They should seek medical attention as soon as possible if extended exposure is suspected.
- Additionally, any local laws and regulations that apply should be followed while disposing of this chemical.
- Silver nitrate exposure has been linked to cancer in some cases.
Practice problems:
Q 1: Silver nitrate when decomposed produces:
A. Oxygen and nitric oxide gas
B. Oxygen and nitrogen dioxide gas
C. Oxygen and Nitrous oxide gas
D. Oxygen, nitric oxide and Nitrous oxide gas
Answer: B)
Solution: Silver nitrate when decomposed produces silver, oxygen gas and nitrogen dioxide gas.Below is the mentioned reaction,
Q 2: __________is the technique of applying a uniform, thin layer of silver to a clean glass surface.
A. Pattinson's process
B. Polishing of mirrors
C. Silvering of mirrors
D. Electrolysing process
Answer: C)
Solution: For the manufacture of silver mirrors, silver nitrate is frequently employed. Silvering of mirrors is the technique of applying a uniform, thin layer of silver to a clean glass surface.
Q 3:___________ precipitate is obtained when sodium phosphate and silver nitrate combine together.
A.White coloured
B. Red coloured
C. Yellow coloured
D. Black coloured
Answer: C)
Solution: Yellow coloured precipitate is obtained when sodium phosphate and silver nitrate combine.Below is the mentioned reaction when sodium phosphate combines with silver nitrate.
Q 4: Silver nitrate and iodine interact in two different ways, but when too much silver nitrate is consumed, ___________is formed.
A. periodic acid
B. silver iodate
C. iodic acid
D. Hypoiodous acid
Answer: B)
Solution: Silver nitrate and iodine interact in two different ways, but when too much silver nitrate is consumed, silver iodate is formed.This is the reaction which takes place when silver nitrate is taken in excess.
Frequently asked questions-FAQs
Q 1: Does lunar caustic pose a risk for humans?
Solution: Because silver nitrate (lunar caustic) is an oxidant, organic substances should not be exposed to it. Silver nitrate is frequently extremely poisonous and corrosive, despite its widespread use (particularly in shallow doses) for the prevention of gonorrhoea and to halt bleeding from the nose. The only immediate negative effect of short-term exposure to this substance is the development of a violet, brown, or black stain on the area of the skin that came into contact with the silver nitrate. However, ocular damage frequently occurs when this compound is exposed for an extended period of time. This substance is frequently categorised as an eye and skin irritant.
Q 2: Why are dark bottles used to store silver nitrate?
Solution: In the presence of sunlight, silver nitrate breaks down. Silver nitrate is put in brown-coloured bottles to prevent this breakdown since sunlight cannot penetrate through them.
Q 3: How was silver nitrate discovered?
Solution: The German chemist Johann Schulze (1687–1744) produced discoveries that led to the use of silver nitrate in printing and photography in the 1720s. Schulze discovered that when exposed to light, a solution of silver, nitric acid, and chalk turns purple or black.
Q 4: Does silver nitrate dissolve in water?
Solution: Due to its potent ionic characteristics, it quickly dissolves in water. At higher temperatures, it can break down and turn into elemental silver. When silver nitrate is dissolved in water, silver ions are created that are positively charged and can bond to other negatively charged ions like halides.


