-
Call Now
1800-102-2727
Liquefaction of Gases- Introduction, Factors Affecting Liquefaction of Gases, Andrew’s Isotherm, Critical Point, Significance of Critical Temperature
I think you all must be aware of the importance of oxygen during the recent pandemic that had devastated the whole world. One of the most common symptoms of patients was shortness of breath. Medical oxygen played a crucial role in the treatment of such patients suffering from severe Covid-19. Not only this but medical oxygen is also used in hospitals and medical institutions in major trauma surgeries to prevent oxygen deficiency in the body. But do you know which form of oxygen is supplied for medical purposes? Yes! It might sound weird but it is in liquid form. Medical liquid oxygen is supplied and stored in a liquid form at low temperature in storage tanks, it is then converted to medical oxygen gas at normal temperature for patients to breathe when required. Do you know some other ways to liquefy the gas apart from lowering the temperature? Well! Let's learn about how the liquefaction of gas takes place and the condition required for the liquefaction of gases.
Table of contents
- Introduction to liquefaction of gas
- Factors affecting liquefaction of gas
- Andrew’s isotherm
- Critical point
- Significance of critical temperature
- Practice problems
- Frequently asked questions-FAQs
Introduction to liquefaction of gas
Transporting gases is tough. It is nearly impossible for gas to be transported from one location to another because gases occupy a large volume in comparison to liquids. The gas is converted into a liquid form before transporting. The study of gas liquefaction reveals changes in a gas's characteristics. It also provides useful information on matter structure in general.
Liquefaction is the process in which gas is converted into liquid form by lowering the temperature or increasing the pressure or both.
Factors affecting liquefaction of gas
Liquefaction of gas depends on two factors:
- Pressure: On increasing the pressure, gas molecules come closer to each other and hence increasing the intermolecular forces and thus converting into the liquid state.
- Temperature: On decreasing the temperature kinetic energy of gas molecules decreases, movement of gas molecules slows down. The slow-moving molecule comes closer to each other due to the intermolecular forces thus aggregating and converting into the liquid form.
Note: Liquefaction of gas is not possible if the temperature is greater than a specific temperature known as critical temperature even if high pressure is applied.
Andrew’s isotherm
In 1869, Thomas Andrew studied the pressure-volume-temperature relationship for carbon dioxide gas and found that above a certain temperature it is impossible to liquefy a gas whatever the pressure was applied.
The result of Andrew’s experiment is shown below in which pressure is plotted against the volume at various temperatures for CO2 gas. Each pressure-volume plot is called an isotherm (as the temperature is constant for each curve).

Let us consider an isotherm curve at 13.1 ℃. At low pressure, carbon dioxide is entirely in the gaseous state and is represented by a point A in the isotherm. On increasing the pressure, volume decreases approximately in accordance with Boyle's law as shown by the portion AX of the isotherm curve. From point X to point Y, deviation from Boyle's law is observed and volume decreases rapidly at a constant pressure as the gas is getting converted into liquid. At a point, Y carbon dioxide gas is completely converted into liquid carbon dioxide. In between point X and Y both the liquid and gas phases of carbon dioxide are in equilibrium.
The pressure corresponding to the horizontal portion XY represent the vapour pressure of the liquid at that particular temperature of the isotherm. It is the maximum pressure that can be attained by gas at that temperature.
The curve between the point Y and Z is represented by an almost vertical line which shows that liquid is only slightly compressible and therefore pressure shoots up even with the slightest decrease in the volume.
When the similar experiment was repeated at a higher temperature below the critical temperature it was observed that the nature of the graph looked similar but the vapour pressure was higher than in the previous case. This is because when the temperature of the gas is increased, the kinetic energy also increases and therefore more pressure is required to condense the gas. The horizontal portion corresponding to the volume of gas that liquefies decreases as we move to the higher temperature and limit to a point at a temperature of 30.98 ℃ for carbon dioxide gas. The temperature above which no liquefaction of gas takes places is known as critical temperature.
Above the critical temperature, ( 30.98 ℃) liquefaction of gas does not take place, even if highest of pressure is applied.
Critical point
Van der Waals equation can be represented as:
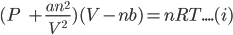
Here,
“P” determines the actual pressure of the gas
“b" is the excluded volume of molecules per mole of gas
“V” is the volume of the gas considering ideal behaviour
“n” represents the number of moles of gas
“T” represents the absolute temperature of the gas
“R” represents the universal gas constant
“a” represents the constant which is indicative of the strength of the intermolecular forces
For one mole of a gas van der Waals equation can be represented as:
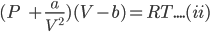
Above equation is rearranged to;
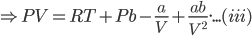
Multiplying the equation (iii) by V2, we get;
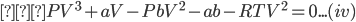
Dividing the equation (iv) by ‘P’ we get,
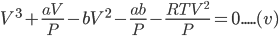
Equation (v) can be written as:
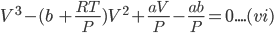
Equation (vi) is a cubic equation in ‘V' therefore, for any value of P and T there will be three roots of V, all of which may be real or one may be real and other two imaginary or one imaginary and other two real.
At the critical point, all the three roots of the equation (vi) are not only real and positive but they are also identical and equal to the critical value of volume i.e., Vc.
At a critical point,
V = Vc
[when V = Vc, pressure = Pc (critical pressure), temperature = Tc (critical temperature)]
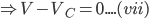
Cubing both the sides and expanding the equation (vii), we get;
(V-Vc)3 = 0
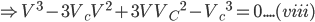
Comparing coefficients of equation (viii) and equation (vi),
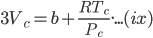
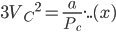
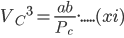
Dividing the equation (xi) by (x), we get;
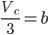
⇒ Vc = 3b
Substituting the value of Vc in equation (x) we get,
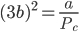
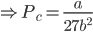
Substituting the value of Pc, Vc in equation (ix), we get;
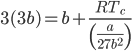
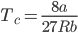
Using the equations of Pc, Vc and Tc, we get;
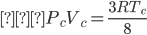
The above equation represented as an equation of critical state.
Where,
Pc represents the critical pressure
Vc represents the critical volume
Tc represents the critical temperature
Note: At the critical point,
- Surface tension becomes zero.
- Enthalpy of vaporization (△Hvap) becomes zero.
- Density of liquid = Density of vapour
Significance of critical temperature
Critical temperature (Tc) : Critical temperature of the gas is the maximum temperature at which the liquefaction of gas is possible and above critical temperature liquefaction of gas can not take place.
As we know that,
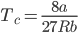
Using the above equation, it can be said that;
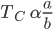
Here,
‘a’ represents the attraction force present between the molecules.
Generally, the higher the value of 'a', the more will be the force of attraction between the molecules, the easier for gas to liquefy, and the higher will be the critical temperature.
‘b’ represents the incompressible volume of gas molecules. It is also known as the excluded volume. Generally higher the value of “b", difficult to liquefy the gas because for liquefaction to to take place molecules need to come closer and compressed but when the value of ‘b’ increases the incompressible volume of gas increases which results in an increase in the repulsion between the molecules and as we know that critical temperature of the gas is inversely proportional to ‘b’ therefore it is generally difficult to liquefy the gas. So, the higher the value of "b", the lower will be the critical temperature.
Order of critical temperature for some of the compounds is:
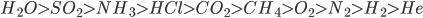
Stronger the forces of attraction between gas particles, greater will be the critical temperature.
The critical temperature of H2O is the highest among all these mentioned substances because in H2O hydrogen bonding is present, which is the strongest force of attraction between molecules. Whereas, in O2, N2, H2 and He weak London dispersion forces are present, hence lesser critical temperatures of these gases. Among non-polar molecules, the higher the molecular mass stronger will be the attraction forces and therefore, the higher the critical temperature.
Recommended video
Practice problems
Q1. Critical pressures and critical temperatures of three hypothetical gases are given below. Select the gases which cannot be liquified at a temperature of 50 K and at a pressure of 25 atm.
|
Gas |
Critical temperature(K) |
Critical pressure(atm) |
|
X |
33 |
4.3 |
|
Y |
127 |
7 |
|
Z |
145 |
10 |
- X and Y
- Y and Z
- X and Z
- Only X
Answer: (B)
Solution: As we know that a gas can be liquefied by applying the pressure but only when the temperature is below or equal to the critical temperature of the gas.
According to the given data, we can see that the critical temperature of the gas X is less than the given temperature (50 K). Therefore it cannot be liquefied while gas Y and gas Z can be liquefied at a given temperature.
Q2. Which of the following is the correct order of ease of liquefaction of gases CO2, H2O, O2,NH3?
- H2O > O2 > CO2 > NH3
- H2O > NH3 > CO2 > O2
- NH3 > CO2 > O2 > H2O
- H2O > CO2 > O2 > NH3
Answer: (B)
Solution:
The order of force of attraction between molecules:
H2O > NH3 > CO2 > O2
H2O: Strong intermolecular hydrogen bonding
NH3: Dipole-dipole interactions
CO2: London dispersion forces
O2: London dispersion forces
Among non-polar molecules (CO2 & O2), the higher the molecular mass stronger will be the attraction forces and therefore, the higher the critical temperature.
The stronger the forces of attraction between gas particles, the greater will be the critical temperature and easier for gas to liquefy.
So, the order of ease of liquefaction is:
H2O > NH3 > CO2 > O2
Q3. Given below are the values of van der Waals constant ‘a’ and ‘b’ for three different gases X,Y,and Z. Select the correct option for the gas which has the highest critical temperature.
|
Gas |
Van der Waal constant (a) in atm L2 mol-2 |
Van der Waals constant (b) in L mol-1 |
|
X |
6.2 |
0.025 |
|
Y |
3.15 |
0.15 |
|
Z |
2.3 |
0.02 |
- Gas X
- Gas Y
- Gas Z
- All the three gases have a same critical temperature
Answer: (A)
Solution: As we know that,
Critical temperature 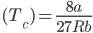
From the above equation, we know that;
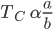

Q4. Which of the following relations is correct?
Here,
Pc represents the critical pressure
Vc represents the critical volume
Tc represents the critical temperature
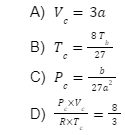
Answer: (B)
Solution:
As we know that the expression of critical constants are: 
We know that,
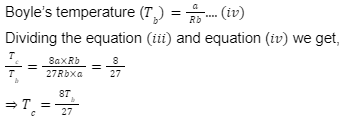
Frequently asked question-FAQs
Question 1. What is the difference between vapour and a gas?
Answer: Those substances that can be liquefied by applying pressure (below critical temperature) and the substances which can be converted into gaseous phase are known as vapours of those substances. Whereas, gas is referred to a substance which is present above the critical temperature and cannot be liquefied by applying pressure.
Question 2. What is the importance of the liquefaction of gas?
Answer: Liquefaction is the process in which gas is converted into liquid form by lowering the temperature or increasing the pressure or both. Transporting gases is tough. It is nearly impossible to transport them from one location to another due to their physical features. The gas is changed into a liquid for the same reason.
Question 3. What is the use of some important gases that are liquefied?
Answer: Some of the important uses of liquefaction of gas are:
- Liquid oxygen is used for different purposes like in hospitals and health institutions, welding metals etc.
- Liquefied petroleum gas (LPG) is used for different purposes like household purposes for cooking, as a fuel to run the vehicles etc.
- Liquid nitrogen is used in cryotherapy to remove unwanted skin, for transportation of food samples etc.
Question 4. What are the steps involved in the liquefaction process?
Answer: There are two major steps involved in the liquefaction process:
- Increasing the pressure: In the first step when the pressure is increased it brings the molecules closer such that the molecules now can interact.
- Decreasing the temperature: When the temperature is decreased the kinetic energy of the molecules decreases and increases the force of attraction and thus molecules aggregate and convert into liquid form.
Related topics
|
Van der Waal equation |
Gay-Lussac’s Law |
|
Kinetic theory of gases |
Dalton's Law of Partial Pressure |
|
Ideal Gas Equation |
Real Gas |


