-
Call Now
1800-102-2727
Stoichiometric Problems Based on Limiting Reagent-Determining Limiting Reagent - Definition, Examples, Practice Problems & FAQs
Imagine you are a car manufacturer and in stock, you have 100 wheels & 30 engines and rest other parts in excess. How many maximum cars you can manufacture in your plant?
Understand it with a chemical equation form, the other part required for manufacturing your branded car is in excess amount only the number of wheels and engines are the bottleneck.
Each car would require four wheels in order to be completed.
Wheel +Engine Four wheeler car
Initial number 100 30 0
Final number 0 30 - 25 = 5 25
Limiting reagents controls the amount of the product formed. In this case, the wheel is limiting reagents. The number of wheels will decide the number of cars produced. Available 100 wheels will use to manufacture only 25 cars.

Table of contents
- Limiting Reagent
- Rules to find limiting reagent
- Practice problems
- Frequently asked questions-FAQs
Limiting Reagent
The reactant that is consumed first and limits the amount of the product formed in the reaction is known as the limiting reagent, assuming the a complete reaction.
- The limiting reagent is present in the least stoichiometric amount.
- Limiting reagents controls the amount of the product formed.
- The remaining or left out reactant is termed an excess reagent.
The remaining or left out reactant is called the excess reagent.
Chemical reactions need to be balanced before applying the concept of limiting reagents.
In a balanced chemical equation if initially given moles of reactant are not in the same ratio of their stoichiometric coefficient ratio, then at least one reactant will act as a limiting reagent.
E.g- 2H2(g)+O2(g)2H2O(l)
Let us assume that the reaction started with 3 moles of hydrogen gas and 4 moles of oxygen gas.
the stoichiometric coefficient ratio of H2 & O2 = 21
Initial mol ratio of H2 & O2 = 34
stochiometric coefficient ratio of H2 & O2 Initial mol ratio of H2 & O2
Case 1 ⇒ If the stoichiometric coefficient ratio of reactants = Initial mol ratio of reactants
Example. 6 moles of Zn is combined with 12 moles of HCl to form ZnCl2 and H2 . Calculate the evolved volume of H2 gas at STP.
A. 136.2 L
B. 13.62 L
C. 68.1 L
D. 272.4 l
Answer: (A)
Zn(s)+2HCl(aq)ZnCl2(s)+H2(g) (balanced)
In this case, the stoichiometric coefficient ratio of Zn & HCl = 1/2
Initial mol ratio of Zn & HCl = 6/12=1/2
stochiometric coefficient ratio of Zn & HCl = Initial mol ratio of Zn & HCl . So, both reactants will consume.
Zn(s)+2HCl(aq)ZnCl2(s)+H2(g)
Initial moles: 6 12 0 0
Final moles: 0 0 6 6
Mole of H2 formed = 6
We know, the volume of 1 mole of any gas at STP is 22.7 L
So, the volume of H2 gas = 6 x 22.7 L=136.2 L
Case 2 ⇒ If the stoichiometric coefficient ratio of reactants Initial mol ratio of reactants
Example. 6 moles of Zn is combined with 18 moles of HCl to form ZnCl2 and H2 . Calculate the evolved volume of H2 gas at STP.
E. 136.2 L
F. 13.62 L
G. 68.1 L
H. 272.4 l
Answer: (A)
: Zn(s)+2HCl(aq)ZnCl2(s)+H2(g) (balanced)
In this case, stoichiometric coefficient ratio of Zn & HCl = 1/2
Initial mol ratio of Zn & HCl = 6/18=1/3
stochiometric coefficient ratio of Zn & HCl Initial mol ratio of Zn & HCl . So, one reactant will consume first act as limiting reagent and one will be left out (excess reagent).
Rules to find out limiting reagent:
Step 1- divide the initial moles of the individual reactants with their respective coefficient and compare the obtained value ( given initial molesstochiometric coefficient) for all reactants.
Step 2 - The reactant having a minimum value of given initial molesstoichiometric coefficient act as limiting reagent
Step 3 - Then all calculation should be based on limiting reagent
Now, check
|
Reactant |
Stoichiometric coefficient |
Given moles |
|
|
|
Zn |
1 |
6 |
|
Zn is limiting reagent |
|
HCl |
2 |
18 |
|
In this case Zn will act as limiting reagent and decide the amount of product.
Rules to calculate the amount of product formed and amount of remaining excess reagent
Moles of product formed = ![]() Stoichiometric coefficient of product
Stoichiometric coefficient of product
Moles of consumed excess reagent = ![]() Stoichiometric coefficient of excess reagent
Stoichiometric coefficient of excess reagent
Moles of ZnCl2 formed = ![]() Stoichiometric coefficient of ZnCl2
Stoichiometric coefficient of ZnCl2
Moles of ZnCl2 formed =![]()
Moles of H2 formed =  Stoichiometric coefficient of H2
Stoichiometric coefficient of H2
Moles of ZnCl2 formed = ![]() mol
mol
Moles of HCl consumed =  Stoichiometric coefficient of HCl
Stoichiometric coefficient of HCl
Moles of HCl formed = 
Moles of HCl remaining = 18 mol -12 mol=6 mol
Zn(s)+2HCl(aq)ZnCl2(s)+H2(g)
Initial moles: 6 18 0 0
Final moles: 0 6 6 6
Mole of H2 formed = 6
We know, the volume of 1 mole of any gas at STP is 22.7 L
So, volume of H2 gas = 6 x 22.7 L=136.2 L
Practice problems
Q 1. The reaction 2C(s) + O2(g)⟶ 2CO(g) is carried out by taking 24 g of carbon and 96 g O2. find the excess reagent and the amount of left reagent?
a. C & 12 g
b. O2 & 32 g
c. O2 & 64 g
d. C & 6 g
Answer: (C)
Step 1: find out limiting reagent:
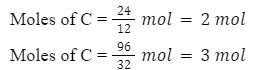
Now, check
2C + O2⟶ 2CO
|
Reactant |
Stoichiometric coefficient |
Given moles |
|
|
|
C |
2 |
2 |
|
C is limiting reagent |
|
O2 |
1 |
3 |
|
C is limiting reagent and O2 is excess reagent.
2C + O2 ⟶ 2CO
Initial moles: 2 3 0
Final moles: 0 2 2
Moles of product formed =  Stoichiometric coefficient of product
Stoichiometric coefficient of product

Q 2. The reactions 2SO2(g)+O2(g)2SO3(g) and 2SO3(g)+2H2O(l)2H2SO4(l) are carried out by taking 640 g of SO2, 64 g of O2 and 90 g of H2O. What are the limiting reagents for the two reactions?
a. SO2 & SO3
b.. SO2 & H2O
c. O2& H2O
d. O2 & SO3
Answer: (D)
For reaction, 2SO2+O22SO3
Step 1: find out limiting reagent:

|
Reactant |
Stoichiometric coefficient |
Given moles |
|
|
|
SO2 |
2 |
10 |
|
|
|
O2 |
1 |
2 |
|
O2 is limiting reagent |
O2 is limiting reagent and SO2 is excess reagent.
2SO2 + O2 2SO3
Initial moles: 10 2 0
Final moles: 6 0 4
Moles of product formed = initial moles of  Stoichiometric coefficient of product
Stoichiometric coefficient of product
Moles of SO3formed = ![]()
Now, for second reaction
For reaction, 2SO3+2H2O2H2SO4
Step 1: find out limiting reagent:
Molar mass of SO3= 80 g mol-1
Molar mass of H2O= 18 g mol-1
Molar mass of H2SO4= 98 g mol-1
Moles of SO3 = 4 mol
Moles of H2O = 9018 mol=5 mol
|
Reactant |
Stoichiometric coefficient |
Given moles |
|
|
|
SO3 |
2 |
4 |
|
SO3 is limiting reagent |
|
H2O |
2 |
5 |
|
SO3 is limiting reagent and H2O is excess reagent.
Q 3. P(g)+Q(g)P3Q2(g)(unbalanced)
P3Q2(g)+R(g)P3Q2R2(g)(unbalanced),
two reactions are carried out by taking 6 moles each of P and Q and 1 mole of R. Then find the incorrect answer

Answer: (A)
3P+2QP3Q2
|
Reactant |
Stoichiometric coefficient |
Given moles |
|
|
|
P |
3 |
6 |
|
P is limiting reagent |
|
Q |
2 |
6 |
|
3 P + 2Q P3Q2
Initial moles: 6 6 0
Final moles: 0 2 4
Moles of product formed = initial moles of  Stoichiometric coefficient of product
Stoichiometric coefficient of product

|
Reactant |
Stoichiometric coefficient |
Given moles |
|
|
|
P3Q2 |
1 |
2 |
|
|
|
R |
2 |
1 |
|
R is limiting reagent |

Q 4. The reaction N2 + 3H2⟶ 2NH3 is carried out by taking 2 moles of nitrogen gas and 4 moles of hydrogen gas. Find the amount of product formed and identify the limiting reagent?
a. 34 g & N2
b. 17 g & H2
c. 34 g & both N2 & H2
d. None of these
Answer: (C)
Now, for the second reaction
N2 + 3H2⟶ 2NH3
|
Reactant |
Stoichiometric coefficient |
Given moles |
given initial molesstoichiometric coefficient |
|
N2 |
1 |
2 |
|
|
H2 |
2 |
4 |
|
Both reactants will consume and both acts as limiting reagent.
Moles of NH3 formed = 2 mol
Weight of NH3 formed = 217 g=34 g
Frequently asked questions-FAQs
Q 1. What is the general procedure to solve the question by finding the limiting reagent method?
Answer: first balance the chemical reaction, then find the limiting reagent by dividing the initial moles of reactants by their stoichiometric coefficient, the reactant with the lowest ratio of the given mole by their coefficient is the limiting reagent.
Q 2. Why are we call limiting reagents that reactant first consumed in any reaction?
Answer: because limiting reagents will decide the amount of product formed in any chemical reaction. The limiting reagent is consumed first and the reaction stops other involved reactants acting as excess reagents.
Q 3. Are both reactants involved in a chemical reaction that can act as limiting reagents?
Answer: yes, in any chemical reaction all involved reactants can act as limiting reagents if their ratio of given moles and the stoichiometric coefficient is the same
Q 4. Can we apply POAC (principal of atom conservation) in a reaction in which one reactant is in excess?
Answer: We can’t apply POAC on atoms present in excess compounds. POAC is only applied for atoms present in the limiting reagents.
Related Topics:
|
Volume strength of H2O2 |
Mole |
|
Strength of oleum |
Normality |
|
Strength of solution |
Density |














