-
Call Now
1800-102-2727
Law of Multiple Proportions: Statement, Limitations, Practice Problems and FAQs
Which is your favourite outdoor sport?
There are sports which we play individually and there are team sports as well. In every sport one thing is very common that all these sports must be having a set of rules or laws for the smooth conduct of the game and to obtain an unbiased result.
Imagine playing a game of cricket without any rules or laws, it is not even possible to play the game.
Similarly in chemistry, we know that elements combine with others to form compounds. The combination can’t be random; it should follow certain laws. Hence, by observing the pattern of combination of some common compounds, scientists developed laws of chemical combination.
In this article, we will discuss the law of multiple proportions.
Table of Content
- Statement of Law of Multiple Proportions
- Limitations of Law of Multiple Proportions
- Practice Problems
- Frequently Asked Questions - FAQs
Statement of Law of Multiple Proportions
John Dalton was the one who gave this law.
“As stated by this law, if two elements combine to form multiple compounds, then the masses of one element combined with another element's fixed mass bear a simple whole number ratio to one another”.
Let’s understand the definition in simple terms with a known example.
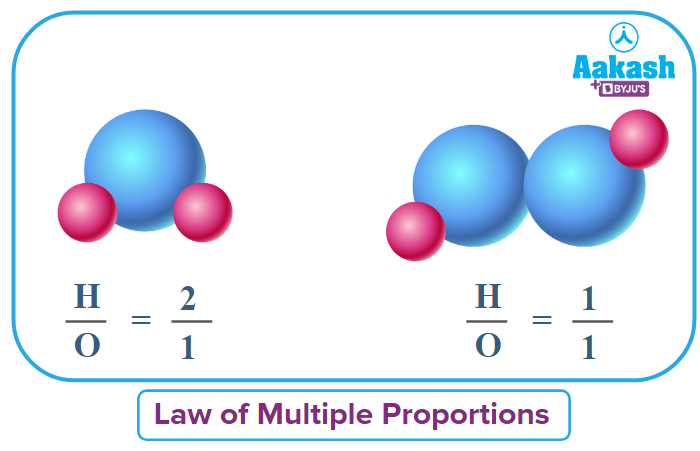
We have taken two elements: hydrogen and oxygen
H & O combines to give two compounds:
(i) H2O (water)
(ii) H2O2(Hydrogen peroxide)
|
Element |
H2O |
H2O2 |
|
H |
2g |
2g |
|
O |
16g |
32g |
In the above example you can see clearly that mass of H is fixed. Hence, according to the statement of the law of multiple proportions, the ratio of mass of oxygen in H2O and H2O2 should be a whole number. Let’s verify it,
Considering the mass of O in H2O=m1and O in H2O2=m2
The ratio comes out to be a simple whole number.
Let’s take another example.
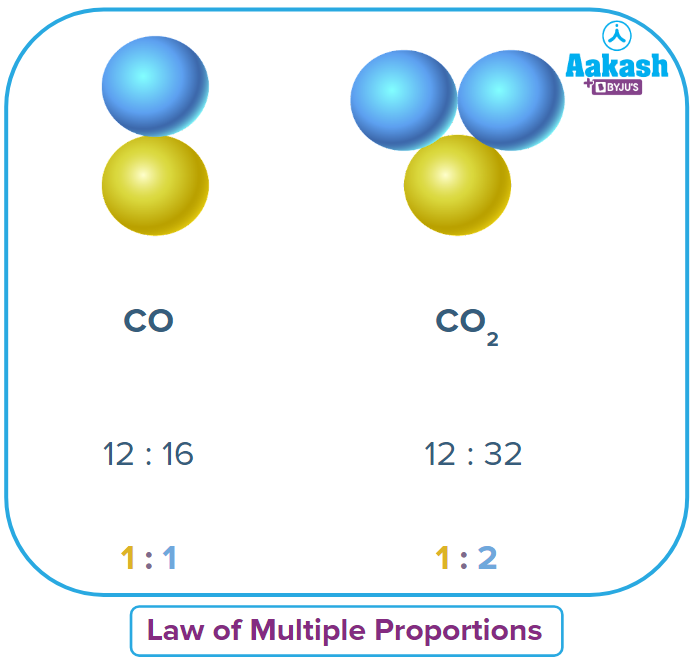
We have taken two elements: carbon and oxygen
C & O combines to give two compounds:
(i) CO2 (Carbon dioxide)
(ii) CO (Carbon monoxide)
|
Element |
CO2 |
CO |
|
C |
12 g |
12 g |
|
O |
32 g |
16 g |
In the above example you can see clearly that mass of C is fixed. Hence, according to the the statement of the law of multiple proportions, the ratio of mass of oxygen in CO2 and CO should be a whole number. Let’s verify it,
Considering the mass of O in CO2=m1 and O in CO=m2
The ratio comes out to be a simple whole number.
Limitations of Law of Multiple Proportions
The Law of Multiple Proportions holds true until all of an element's compounds contain only that one specific isotopic form of the element.
Practice Problems
Q1. Which of the following pairs illustrates the law of multiple proportions?
a. H2O & HCl
b. NO & NO2
c. H3PO4 & H2O
d. All of these
Answer: (B)
“As stated by this law, if two elements combine to form multiple compounds, then the masses of one element combined with another element's fixed mass bear a simple whole number ratio to one another”.
For example: Nitrogen and oxygen elements combine to form two oxides NO and NO2.
Case-1: In NO, 14 g of nitrogen is combined with 16 g of oxygen.
The mass ratio of
In NO2, 14 g of nitrogen is combined with 32 g of oxygen.
The mass ratios of
Thus, we can see that the mass of oxygen combined with a fixed mass of nitrogen (14 g) bears a simple ratio of 1632 or 12.
So, option B is the correct answer.
Q2. The pair of compounds below that best exemplifies the law of multiple proportions is:
a. NH3 & NCl3
b. H2S & SO2
c. CuO & Cu2O
d. CS2 & FeSO4
Answer: (C)
“As stated by this law, if two elements combine to form multiple compounds, then the masses of one element combined with another element's fixed mass bear a simple whole number ratio to one another”.
As a result, when copper is combined in a ratio of 1:2 with a fixed weight of oxygen, CuO and Cu2O form two distinct compounds.
Q3. Law of multiple proportions came into inception due to which of the following scientists?
a. John Dalton
b. Lavoisier
c. Gay Lussac
d. Prout
Answer: (A)
This law was given by John. Dalton.
“As stated by this law, if two elements combine to form multiple compounds, then the masses of one element combined with another element's fixed mass bear a simple whole number ratio to one another”.
Q4. Which of the following statements is incorrect?
a. Law of multiple proportions was given John Dalton
b. CO2 & CO follow the law of multiple proportions.
c. Heavy water and hydrogen peroxide follow law of multiple proportions.
d. None of the above
Answer: (C)
Solution: Law of multiple proportions was given by John Dalton.
C & O combines to give two compounds:
(i) CO2 (Carbon dioxide)
(ii) CO (Carbon monoxide)
|
Element |
CO2 |
CO |
|
C |
12 g |
12 g |
|
O |
32 g |
16 g |
In the above example you can see clearly that mass of C is fixed. Hence, according to the the statement of the law of multiple proportions, the ratio of mass of oxygen in CO2 and CO should be a whole number. Let’s verify it,
Considering the mass of O in CO2=m1 and O in CO=m2
The ratio comes out to be a simple whole number. Hence, CO2 & CO follow the law of multiple proportions.
Heavy water and hydrogen peroxide are compounds made from two different isotopes of hydrogen that are hydrogen and deuterium. Hence, it does not obey the law of multiple proportions as the masses of hydrogen and deuterium are different.
Frequently Asked Questions - FAQs
Q1. Mention a few examples of molecules that follow the law of multiple proportions.
Answer: According to the law of multiple proportions, if two elements combine to form multiple compounds, then the masses of one element combined with another element's fixed mass bear a simple whole number ratio to one another. A few examples following this law are: NO & NO2, SO2 & SO3, CO & CO2 etc.
Q2. How does the law of multiple proportions indicate that atoms exist?
Answer: One of Dalton's atomic theory's observations served as the foundation for the law of multiple proportions. When two or more elements combine together, multiple compounds can be prepared. If one of the element’s mass is fixed then the ratio of other elements mass in both compounds would be a simple whole number.
This law demonstrates that some constituents combine in a specific ratio. These elements could be atoms. Thus, the existence of atoms that combine to form molecules is demonstrated by the law of multiple proportions.
Q3. Does the law of multiple proportions apply to isotopes?
Answer: For isotopes, the law of multiple proportions does not apply. As the law of multiple proportions states that when two elements combine to form two or more chemical compounds, the masses of the combining elements have a simple whole number ratio.
For example :
CO2 & CO
The ratio of oxygen in both compound is 1:2, hence they obey law of multiple proportion but as we know isotopes of carbon exist as C14, C13 & C12. According to the statement of law of multiple proportions the mass of one element should be fixed. Since the atomic masses of the isotopes in this case differ, the ratio and the masses are not fixed.
Q4. What distinguishes the laws of multiple proportion and constant proportion from one another?
Answer: In contrast to the law of multiple proportions, which states that if two elements combine to form more than one chemical compound, the ratio between the masses of the second element that combines with a fixed mass of the first element has to be simple whole number, the law of constant composition states that the same proportion of samples always consists of the same proportion of elements by mass. Law of multiple proportions holds true if one of the element's mass is kept constant, which is not required in law of constant proportions.


