-
Call Now
1800-102-2727
Kolbe’s Electrolysis – Reaction, Mechanism, Practice Problems and FAQ
Your teachers would have regularly discussed the connections between the three distinct fields of chemistry with you. Let's discuss an example where we use an idea from physical chemistry to explain an idea from organic chemistry.
There are numerous ways to prepare hydrocarbons, right?
But did you know that electrochemistry may also be used to prepare hydrocarbons? It is intriguing that we can prepare hydrocarbons using the anode and cathode concepts from physical chemistry. This method is called Kolbe’s electrolysis.
Let’s get to know more about Kolbe’s electrolysis on this concept page.
TABLE OF CONTENTS
- Kolbe’s Electrolysis – Introduction
- Kolbe’s Electrolysis – Mechanism
- Kolbe’s Electrolysis – Limitations
- Kolbe’s Electrolysis – Examples
- Practice Problems
- Frequently Asked Questions – FAQ
Kolbe’s Electrolysis – Introduction
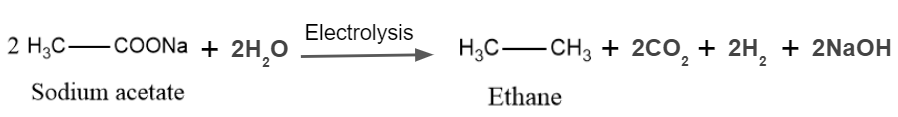
The organic reaction known as Kolbe's electrolysis is named after the illustrious chemist H. Kolbe. It is a general technique for producing substituted hydrocarbons from substituted carboxylic acids, and it produces carbon dioxide gas using an electric discharge.
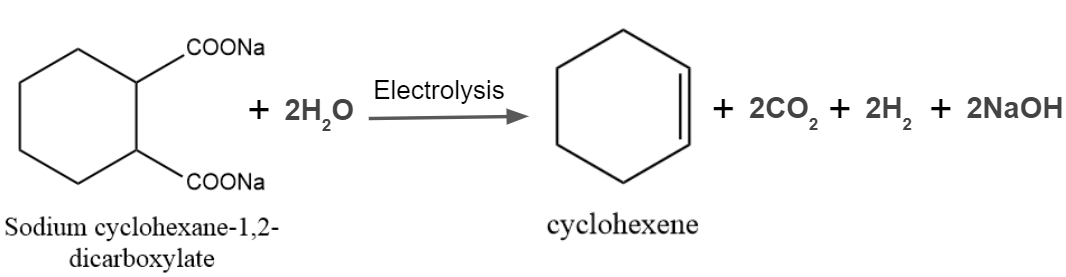
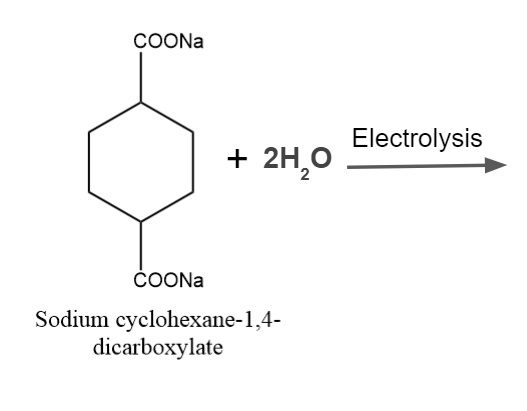
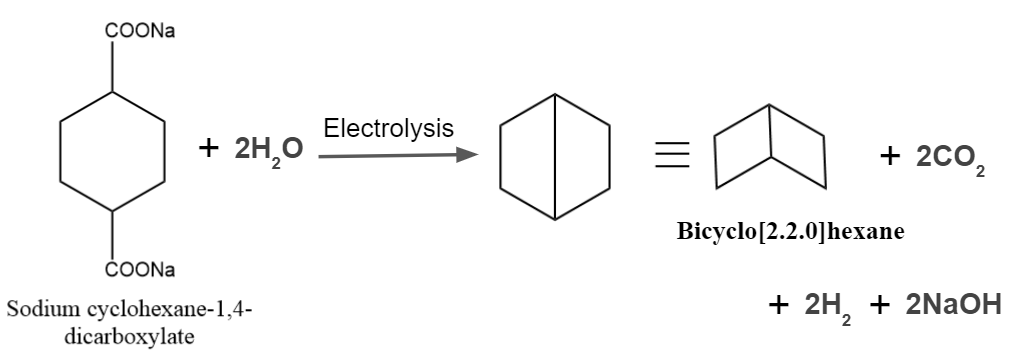
It is one of the most popular processes for creating substituted hydrocarbons and alkanes. Unsaturated hydrocarbons or cyclic rings may be produced if the same organic substance contains both carboxylic acid groups.
The Kolbe reaction, also known as the carboxylate ion reaction, is a decarboxylative dimerization reaction between two carboxylic acids. Decarboxylative dimerization is the word used to describe the direct combining of alkyl groups and the loss of carbon dioxide molecules. Electrons play a significant role in the reaction, which happens in the presence of an electric discharge. The overall reaction is as follows:
Kolbe’s Electrolysis – Mechanism
The reaction mechanism suggests that when there are electrons present in the solution, the carboxylate molecule loses carbon dioxide. Any acyl, aryl or alkyl group is represented by R. The mechanism is outlined as follows:
At anode (Oxidation)
The electrochemical decarboxylation step of the two-step radical process results in an unstable and extremely reactive radical intermediate. This is followed by radical recombination, when chemically active radicals reunite with one another through a covalent connection to create the molecule R-R.
At cathode (Reduction)
As the reaction proceeds, the pH of the solution increases due to the formation of OH- ions.
The Na+ ions will remain in the solution. This is because the Na+ and H+ ions formed by the electrolysis of water will move towards the cathode. But H+ ions will be preferentially reduced at the cathode because the reduction potential of H+ is more than that of Na+ ions. The H+ ions will be discharged to produce H2 gas liberated at the cathode.
The Na+ being in solution combine with OH- to produce
On using two different types of salt of acids, a mixture of three alkanes is obtained.
Kolbe’s Electrolysis – Limitations
- Alkanes with only an even number of carbon atoms can be prepared by this method.
- Methane cannot be prepared by this method.
Kolbe’s Electrolysis – Examples
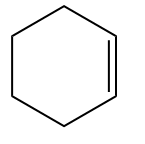
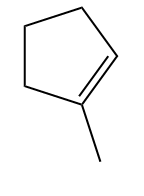
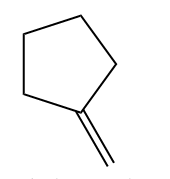
Example 1:
Example - 2:
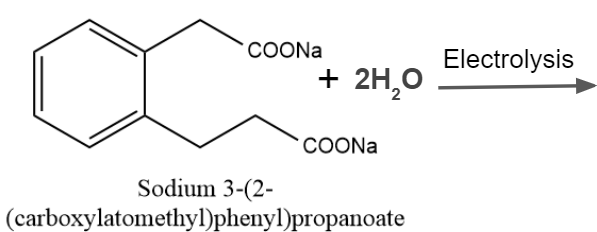
Practice Problems
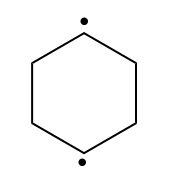
Q1. Predict the product of Kolbe’s electrolysis in the given reaction.
- None of these
Answer: B
Solution: Kolbe's electrolysis reaction is a decarboxylative dimerization of two carboxylic acids, or just carboxylate ions. Decarboxylative dimerisation is the direct linking of alkyl groups and the loss of carbon dioxide molecules. The process, which takes place in the presence of an electric discharge, depends heavily on electrons. The reaction can be described as
So, option B is the correct answer.
Q2. In the given reaction, predict the product of Kolbe's electrolysis.
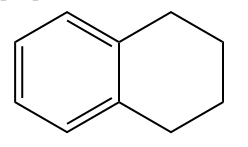
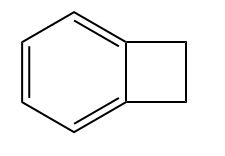
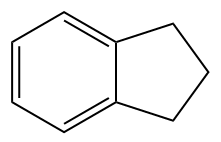
- None of these
Answer: C
Solution: The Kolbe reaction is a decarboxylative dimerisation of two carboxylic acids, or simply carboxylate ions. The term decarboxylative dimerisation refers to the loss of carbon dioxide molecules and the direct joining of alkyl groups. The reaction takes place in the presence of an electric discharge, and electrons play an important role. The reaction can be given as
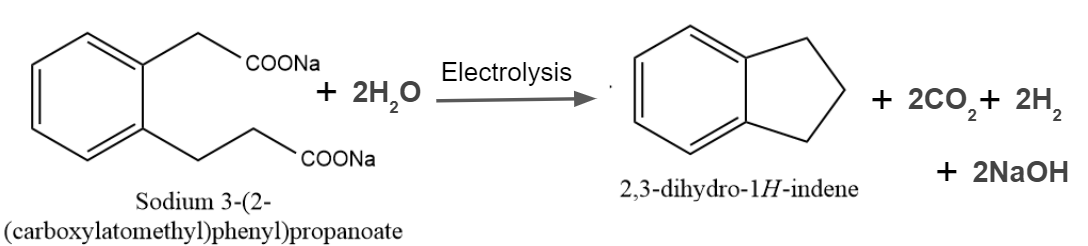
So, option C is the correct answer.
Q3. What is the product of the given reaction?
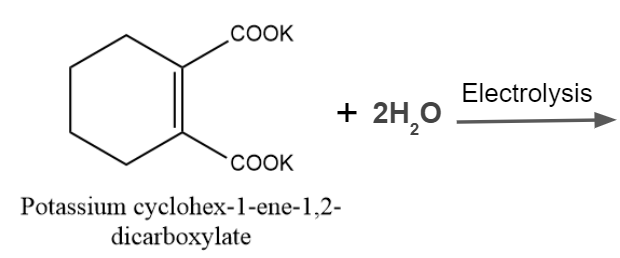
Answer: B
Solution: A decarboxylative dimerization of two carboxylic acids, also known as carboxylate ions, is the Kolbe reaction. Decarboxylative dimerization is the word used to describe the direct combining of alkyl groups and the loss of carbon dioxide molecules. Electrons play a significant role in the reaction, which happens in the presence of an electric discharge. The reaction might be provided as
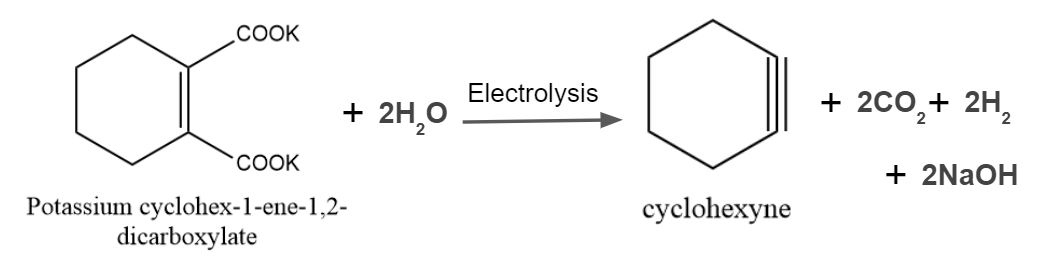
So, option B is the correct answer.
Q4. Predict the outcome of the given reaction's Kolbe's electrolysis.
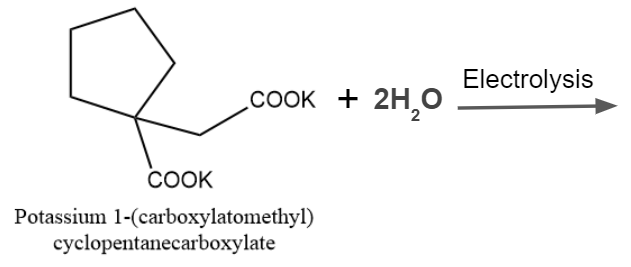
Answer: D
Solution: The Kolbe reaction, also known as carboxylate ions, is a decarboxylative dimerization of two carboxylic acids. The loss of carbon dioxide molecules and the direct linking of alkyl groups are referred to as "decarboxylative dimerization." Electrons are crucial to the reaction, which takes place in the presence of an electric discharge. The reaction could be expressed as
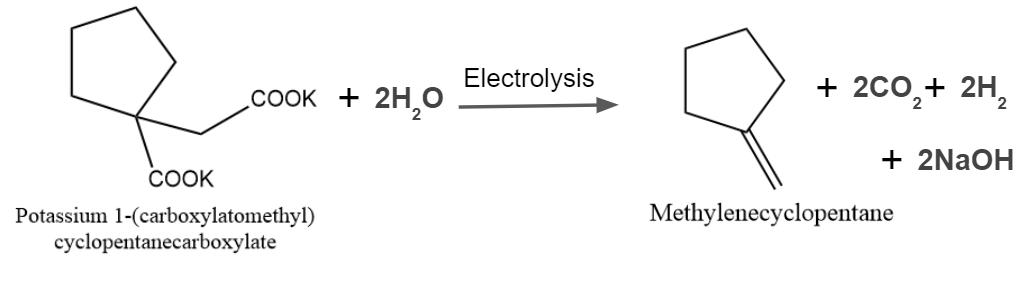
So, option C is the correct answer.
Frequently Asked Questions – FAQ
Q1. How can alkene be synthesised using Kolbe’s Electrolysis method?
Answer: When a potassium or sodium salt of a dicarboxylic acid is subjected to electrolysis, it gives an alkene. When a dibasic acid, having carboxylic acid groups on adjacent carbons is electrolysed in an aqueous solution, an alkene is formed.
Q2. Why does the pH of the solution increase in Kolbe’s electrolysis?
Answer: The Na+ being in the solution combines with OH- to produce
As the reaction proceeds, the pH of the solution increases due to the formation of OH- ions.
Q3. What are the drawbacks of Kolbe's electrolysis method?
Answer: There are two major constraints:
- Alkanes with only an even number of carbon atoms can be prepared by this method.
- Methane cannot be prepared by this method.
Q4. Why can not methane be prepared using Kolbe's electrolytic method?
Answer: For the creation of symmetrical alkanes, or alkanes with an even number of carbon atoms, Kolbe's electrolysis approach is suitable. Since methane only contains one carbon atom, Kolbe's electrolytic method cannot be used to create it.