-
Call Now
1800-102-2727
Ketone: Nomenclature, Preparations, Properties, Uses, Practice Problems and Frequently Asked Questions
Glucose is the primary source of fuel for the body. But whilst carbohydrates are low, ketogenesis turns into the number one fuel processes for maximum cells. These are the instances in which ketones function herbal via means of animal metabolism.
This is the way it happens:
In regular instances for energy the body breaks down glucose for energy. When the case is like that the glucose level in the body is low, then the body is pressured to interrupt down proteins or fat for energy and produces ketone via means of-merchandise which serves as the elements for power metabolism. These ketone bodies are acetone, aceto-acetic acid, and beta-hydroxy-butyric acid.
This is the equal case of what happens in case of starvation, ketone bodies deliver as much as 40-50% of the energy necessities for maximum body tissues, and as much as 60-70% of the power for the brain.
Table of content
- Structure of Ketones
- Nomenclature of ketones
- Preparation of ketones
- Properties of ketones
- Uses of ketones
- Practice problems
- Frequently asked questions
Structure of Ketones
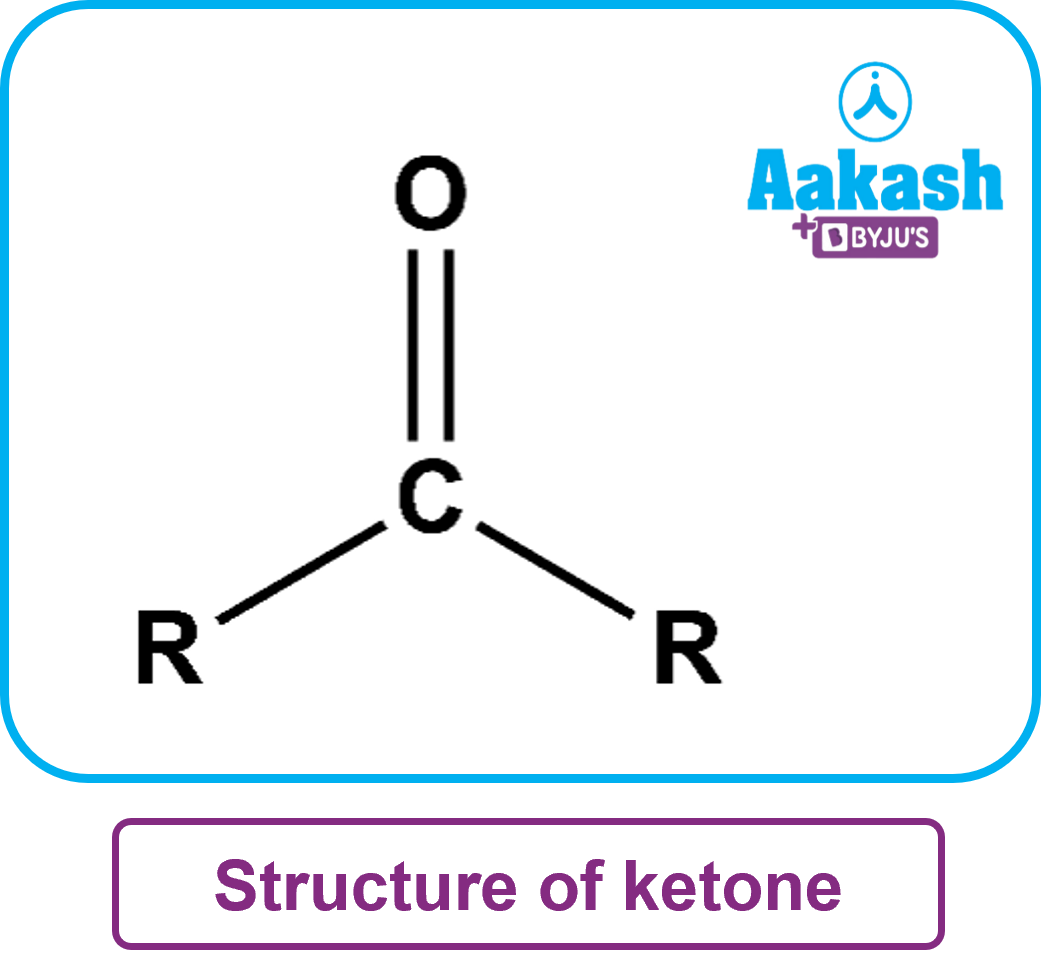
Ketones are organic compounds with a functional group C=O and a structure R (C=O) R'. Here, R and R’ can be alkyl or aryl groups. These carbonyl compounds have carbon-containing substituents on both sides of the carbon-oxygen double bond. The carbonyl carbon of the ketone group is an SP2 hybrid orbital. The structure of the ketone is a triangular planar. The bond angle of this structure is about 120oC. Because carbon-oxygen bonds polarize carbonyl groups (oxygen is more electron-withdrawing than carbon), ketones tend to be nucleophilic at the oxygen atom and electrophilic at the carbon atom.
Nomenclature of ketones:
Ketone is named after the parent alkane with the suffix "one". The position of the carbonyl group of a ketone is indicated by a number when naming the ketone. For example, CH3(CO)CH3 it is called 2-propanone. However, this compound is commonly referred to as acetone. Ketones are generally named by writing the name of the individual alkyl groups attached to the carbonyl carbon, then by writing "ketone" as the third word in the name. For example, butan-2-one can be written as ethyl methyl ketone.
|
Formula |
Common name |
IUPAC name |
|
CH3(CO)CH3 |
Acetone |
Propan-2-one |
|
CH3(CO)CH2CH3 |
Ethyl Methyl Ketone |
Butan-2-one |
|
CH3CH2(CO)CH2CH3 |
Diethyl ketone |
Pentan-3-one |
|
CH3CH(CH3)(CO)CH3 |
Isopropyl methyl ketone |
3-Methylbutan-2-one |
Preparations of ketones
Ketones are organic compounds that contain a carbonyl (C=O) group. The general formula for ketones is R (C=O) R'. Here, R and R’ can be alkyl or aryl groups. They fall into two categories based on substituents. Symmetric ketones (if two identical groups are attached to a carbonyl group) and asymmetric ketones (if two different groups are attached to a carbonyl group). There are many ways to produce ketones on an industrial scale and in the laboratory. Standard methods include oxidation of alcohols, hydrocarbons, etc. Some common methods of making ketones are described below.
1. Preparation of ketones from acyl chloride:
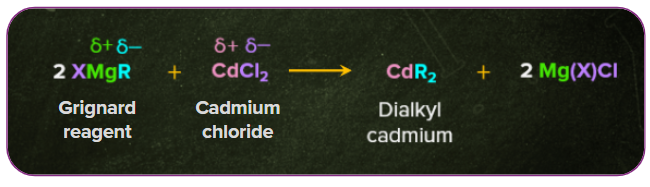
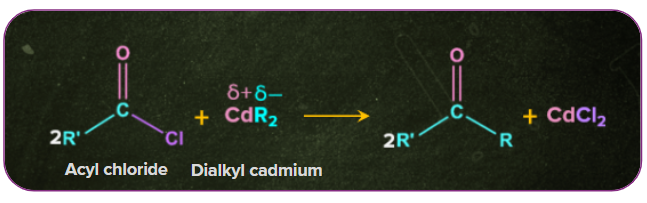
Acyl chlorides produce ketones when treated with dialkyl cadmium. Example: When cadmium chloride reacts with Grignard reagents, dialkyl cadmium is formed. The dialkyl cadmium thus further reacts with the acyl chloride to form a ketone.
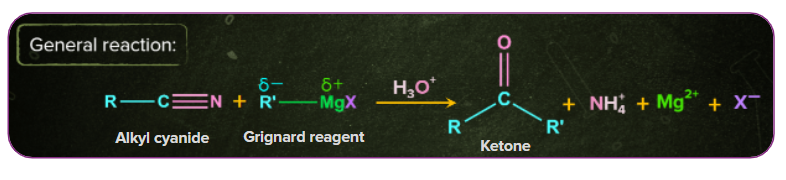
2. Preparation of Ketones from Nitriles:
Nitriles on treatment with Grignard reagent upon further hydrolysis yields ketones.
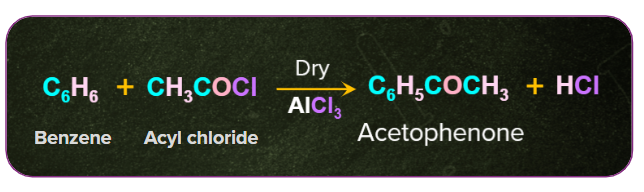
3. Preparation of ketones from benzene:
Ketones are formed by electrophilic aromatic substitution of the benzene ring with acid chloride in the presence of Lewis acids such as AlCl3. This reaction is commonly known as the Friedel-Crafts acylation reaction.
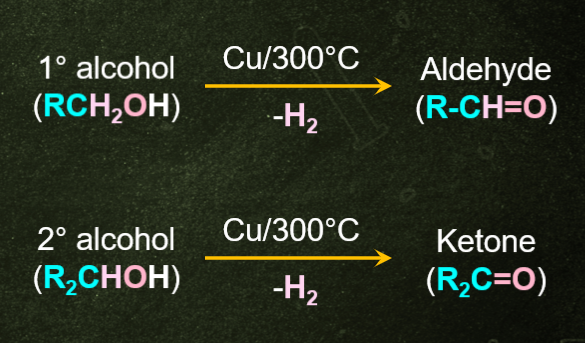
4. Ketone production by dehydrogenation of alcohol:
Dehydrogenation of alcohol is a reaction in which one hydrogen molecule is removed from one alcohol molecule during oxidation. During alcohol oxidation, both CO and OH bonds are cleaved to form C=O bonds. Secondary alcohols are dehydrated in the presence of strong oxidants to produce ketones.
Example: When the vapor of a secondary alcohol passes through copper heated to 573 Kdehydrogenation produces ketones. Tertiary alcohol does not dehydrate during oxidation but they will undergo dehydrogenation. Therefore, in the case of tertiary alcohol, alkenes are produced.
Physical Properties of Ketones
Boiling point: In general, the boiling points of aldehydes and ketones increases with increasing molecular weight. The boiling point depends on the strength of the intramolecular force.
- Van der Waals dispersion Forces: As the length of a molecule and the number of atoms increases, so the forces of attraction between them also increases. For both aldehydes and ketones, the boiling point rises as the number of carbon atoms increases.
- Van der Waals Dipole Dipole attractions: Due to the presence of carbon-oxygen double bonds, both aldehydes and ketones are inherently polar. There is an attractive force between a permanent dipole and a molecule close to it. This is why aldehydes and ketones have higher boiling points than hydrocarbons of similar size.
Solubility:
Lower aldehydes and ketones, such as methanal, propanone, and ethanal, can form hydrogen bonds with water and are miscible with water in any proportion.
Due to the long length of the non-polar alkyl chain, the water solubility drops sharply as the molar mass increases for the aldehydes and ketones. However, all aldehydes and ketones are soluble in organic solvents such as ethers and benzene.
Chemical properties of ketone
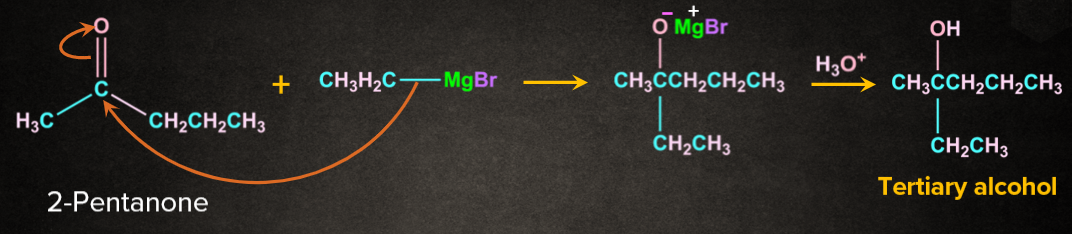
1. Reaction with grignard reagent:
Ketones on reaction with grignard reagent followed by hydrolysis gives tertiary alcohol.
For example 2-pentanone on reaction with ethyl magnesium bromide followed by hydrolysis gives tertiary alcohol.
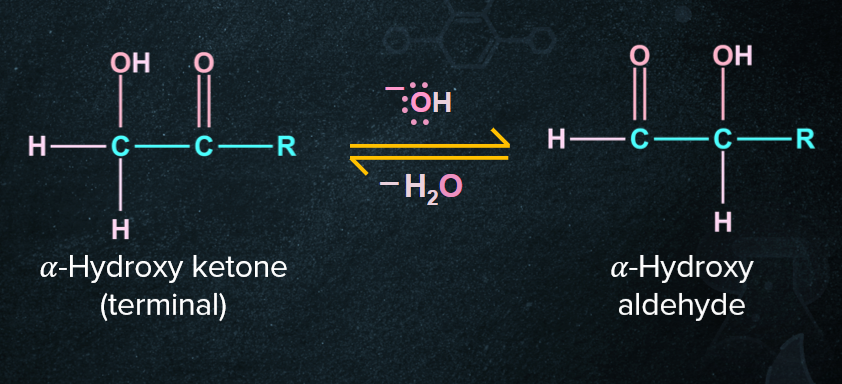
2. Tollens’ test of alpha hydroxy ketones:
Alpha-hydroxy ketones can give a positive Tollens’ test because α-Hydroxyketones have the ability to tautomerize to aldehydes and aldehydes give a Tollens’ test. Some non terminal alpha hydroxy ketones also give the positive Tollens’ test like benzoin.
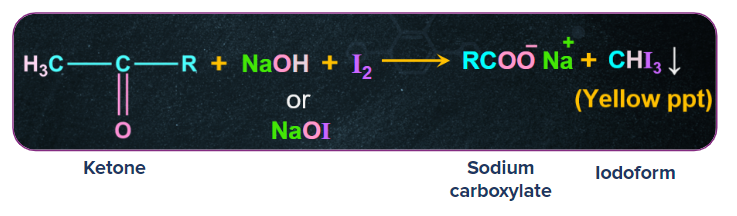
3. Haloform reaction:
Ketones having alpha -CH3 group on reaction with NaOH/X2 gives carboxylate ion and haloform. If we take chlorine as halogen it is called chloroform reaction, bromine as halogen it is called bromoform reaction and iodine as halogen it is called iodoform reaction.
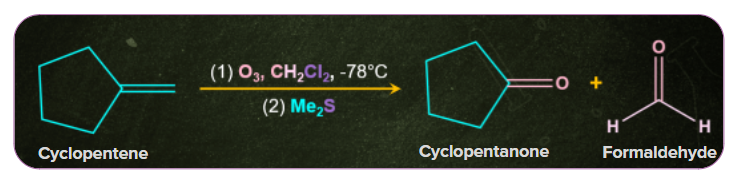
4. Ozonolysis:
Ketones are obtained by the ozonolysis of substituted alkenes followed by reacting the ozonolysis products with dimethyl sulfide at -78oC . A mixture of aldehydes and ketones is obtained depending upon the structure of the hydrocarbon.
Uses of ketones
- Propanone (acetone) is used in the production of polymers such as plexiglass.
- Ketones are used as a solvent and as a starting material for the synthesis of many organic compounds.
- Acetone and ethyl methyl ketone are mainly used as industrial solvents.
Practice problems
Q.1. What is the IUPAC name of acetone?
(A) 2-propanone
(B) 3-butanone
(C) 2-butanone
(D) 3-pentanone
Answer: (A)
Solution: The formula of acetone is CH3(CO) CH3 the IUPAC name is 2-propanone.
Q.2. Among the following which is more reactive in nucleophilic addition reaction?
(A) CH3(CO) CH3
(B) C6H5 (CO) CH3
(C) C6H5 (CO) C6H5
(D) All of these
Answer : (A)
Solution: Sterically, the presence of relatively large substituents in ketones hinders the approach of nucleophile to carbonyl carbon. In acetone the two alkyl groups are small so nucleophilic addition reaction is easy in acetone compared to other two compounds and also in other two compounds the electrophilicity of the carbonyl group will be less due to the presence of the phenyl ring.
Q.3. Acetone on reaction with methyl magnesium bromide followed by hydrolysis will give
(A) Primary alcohol
(B) Secondary alcohol
(C) Tertiary alcohol
(D) Quaternary alcohol
Answer: (C)
Solution: Acetone on reaction with methyl magnesium bromide followed by hydrolysis gives tertiary alcohol. The reaction is as follows
Q.4. Acetone on reaction with I2/NaOH gives
(A) CHI3
(B) CHCl3
(C) CHBr3
(D) None of the above
Answer: (A)
Solution: Reaction of acetone with I2/NaOH is called an Iodoform reaction. The product formed is the iodoform (CHI3) which is yellow precipitate.
Frequently asked questions
1. Why are aldehydes and ketones classified differently?
Answer: Both aldehydes and ketones have carbonyl groups, but they are classified differently because the group of atoms surrounding the carbonyl group is different in both. Therefore, aldehydes have one alkyl or aryl group and one hydrogen is attached to a carbonyl group, whereas ketones have two aryl or alkyl groups attached to both sides of the carbonyl group.
2. Why are the boiling points of ketones and aldehydes lower than those of alcohol?
Answer: Aldehydes and ketones do not have intermolecular hydrogen bonds like alcohols. Therefore, they have less forces of attraction between the molecules. Hence the boiling point of ketones and aldehydes are lower than alcohol.
3. Between aldehydes and ketones which are more reactive in nucleophilic addition reaction?
Answer: Due to steric and electronic reasons, aldehydes are more reactive than ketones in nucleophilic addition processes.
In ketones, the presence of two relatively big substituents makes it more difficult for a nucleophile to approach the carbonyl carbon than in aldehydes with only one such substituent.
Two alkyl groups lower the electrophilicity of the carbonyl carbon in ketones more effectively than in aldehydes.
As a result, Aldehydes are more reactive than Ketones in nucleophilic addition processes.
4. Are ketones acidic?
Answer: Ketones are acidic molecules, so an increased level of ketones can cause the blood to become more acidic which prevents the body's processes from working normally. By definition, ketone levels in DKA (diabetic ketoacidosis) are too high, causing the blood to become dangerously acidic.


